CNS disease does not preclude successful treatment
Neurological symptom burden at diagnosis affects survival
Brain metastases occur in approximately 35 % of patients with metastatic NSCLC and are associated with a variety of neurological symptoms, as well as poor prognosis [1]. However, little is known about the prognostic impact of the symptomatic burden of CNS lesions at the time of diagnosis. This was assessed by an analysis based on a real-life cohort of 1,608 NSCLC patients from the Vienna Brain Metastasis Registry with newly diagnosed brain metastases [2]. Neurological symptoms were evident in 73.8 %. Symptoms included neurological deficits (61.3 %), signs of increased intracranial pressure (30.3 %), epileptic seizures (13.6 %), and neuropsychological symptoms (14.5 %).
According to this analysis, oligosymptomatic or asymptomatic patients, compared to symptomatic patients, experienced significantly longer median OS after their diagnosis of brain metastases (11 vs. 7 months; p < 0.001). Interestingly, signs of increased intracranial pressure showed a significant correlation with prolonged survival (8 vs. 6 months in patients without increased intracranial pressure; p = 0.032). A multivariate analysis identified an independent association between the presence of neurological symptoms and survival from the time of diagnosis. Overall, this study highlights the importance of integrating the neurological symptom burden into the prognostic assessment of patients with NSCLC and brain metastases.
Outcomes according to CNS disease: single-agent pembrolizumab…
The KEYNOTE-001, 010, 024, and 042 trials compared pembrolizumab monotherapy with chemotherapy in NSCLC patients [3-6]. A pooled analysis of these four studies was performed to investigate the effects of pembrolizumab in PD-L1–positive disease according to the presence of brain metastases at baseline [7]. Exploratory subgroup analyses for patients with CNS lesions had been prespecified in all trials. Mansfield et al. presented data for a total of 3,170 individuals 293 of whom had brain metastases (199 and 94 of these received pembrolizumab and chemotherapy, respectively), while 2,877 showed no cerebral disease (pembrolizumab: n = 1,754; chemotherapy: n = 1,123). Approximately half of the patients in each treatment group had a tumor proportion score (TPS) ≥ 50 %.
The clinical benefit of pembrolizumab in patients with TPS ≥ 50 % was similar irrespective of the presence of brain metastases at baseline. Median OS was 19.7 vs. 9.7 months for the pembrolizumab and chemotherapy arms in patients with CNS lesions (HR, 0.78), and 19.4 vs. 11.7 months in those without (HR, 0.66). Comparable risk reductions resulted for PFS, with 4.1 vs. 4.6 months in patients with brain metastases (HR, 0.70) and 6.5 vs. 6.1 months in those without (HR, 0.69). Likewise, in the group with TPS ≥ 1 %, pembrolizumab gave rise to similar OS and PFS effects.
Response rates measured at all tumor sites were higher with pembrolizumab than with chemotherapy for patients with and without brain metastases. In the group with cerebral lesions, median duration of response had not been reached yet for pembrolizumab-treated patients with both TPS ≥ 50 % and TPS ≥ 1 %, while it was 7.6 and 8.3 months, respectively, with chemotherapy. Pembrolizumab monotherapy showed a manageable safety profile irrespective of the presence of brain metastases. In their conclusion, the authors emphasized that prembrolizumab monotherapy is a standard-of-care therapy for patients with PD-L1–positive advanced NSCLC including those with treated, stable brain metastases.
… and pembrolizumab plus chemotherapy
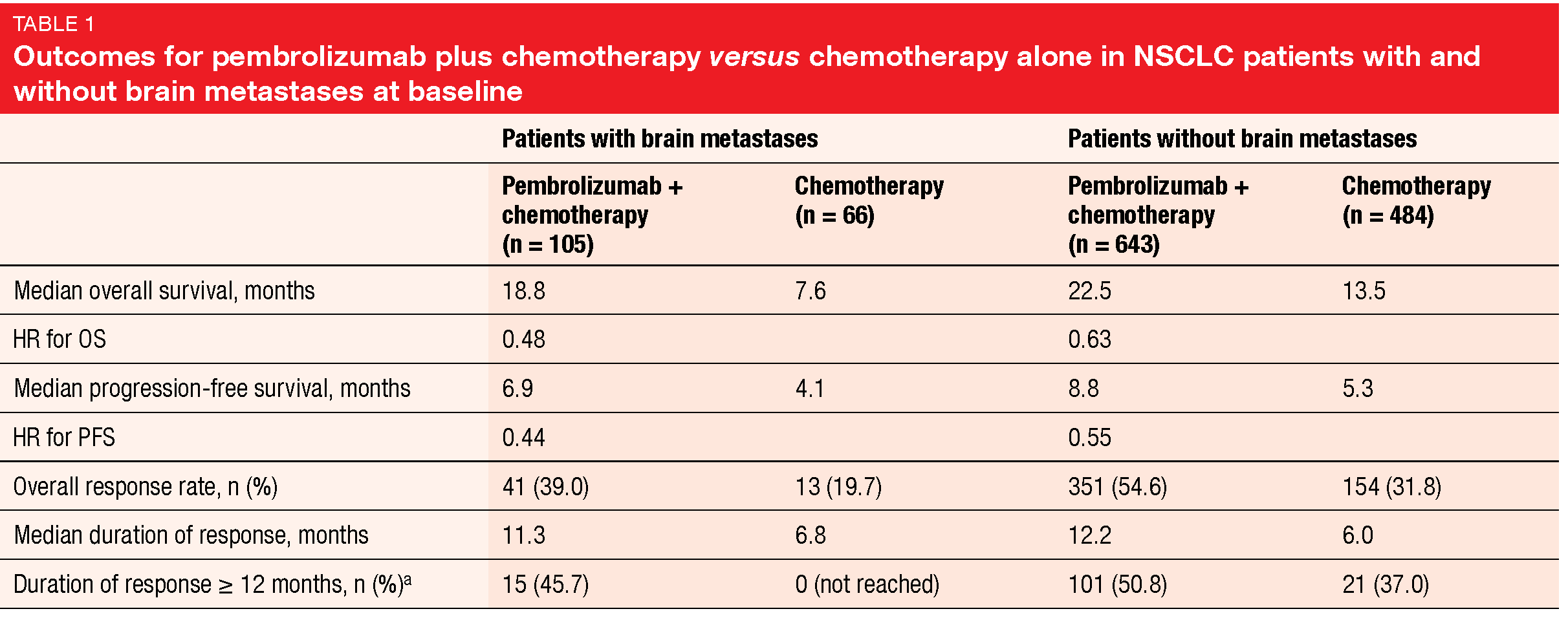
In similar vein, Powell et al. reported an exploratory pooled analysis of the KEYNOTE-021, 189, and 407 trials that evaluated the outcomes in NSCLC patients with and without stable brain metastases at baseline [8]. These studies had tested first-line pembrolizumab plus platinum-based chemotherapy compared to chemotherapy [9-12]. Among a total of 1,298 patients, 171 had brain metastases at the time of study inclusion (pembrolizumab plus chemotherapy: n = 105; chemotherapy: n = 66) and 1,127 did not (pembrolizumab plus chemotherapy: n = 643; chemotherapy: n = 484).
Pembrolizumab plus chemotherapy improved outcomes over chemotherapy alone in both groups (Table 1). This was true for OS, PFS, ORR, and duration of response. The combination had a manageable safety profile in patients with and without brain metastases. In the experimental arm, the presence of cerebral lesions was not associated with an increased rate of adverse events affecting the CNS. The authors noted that pembrolizumab plus chemotherapy is a standard-of-care strategy for patients with advanced NSCLC including those with treated and untreated asymptomatic brain metastases.
GIDEON: real-world experience with afatinib
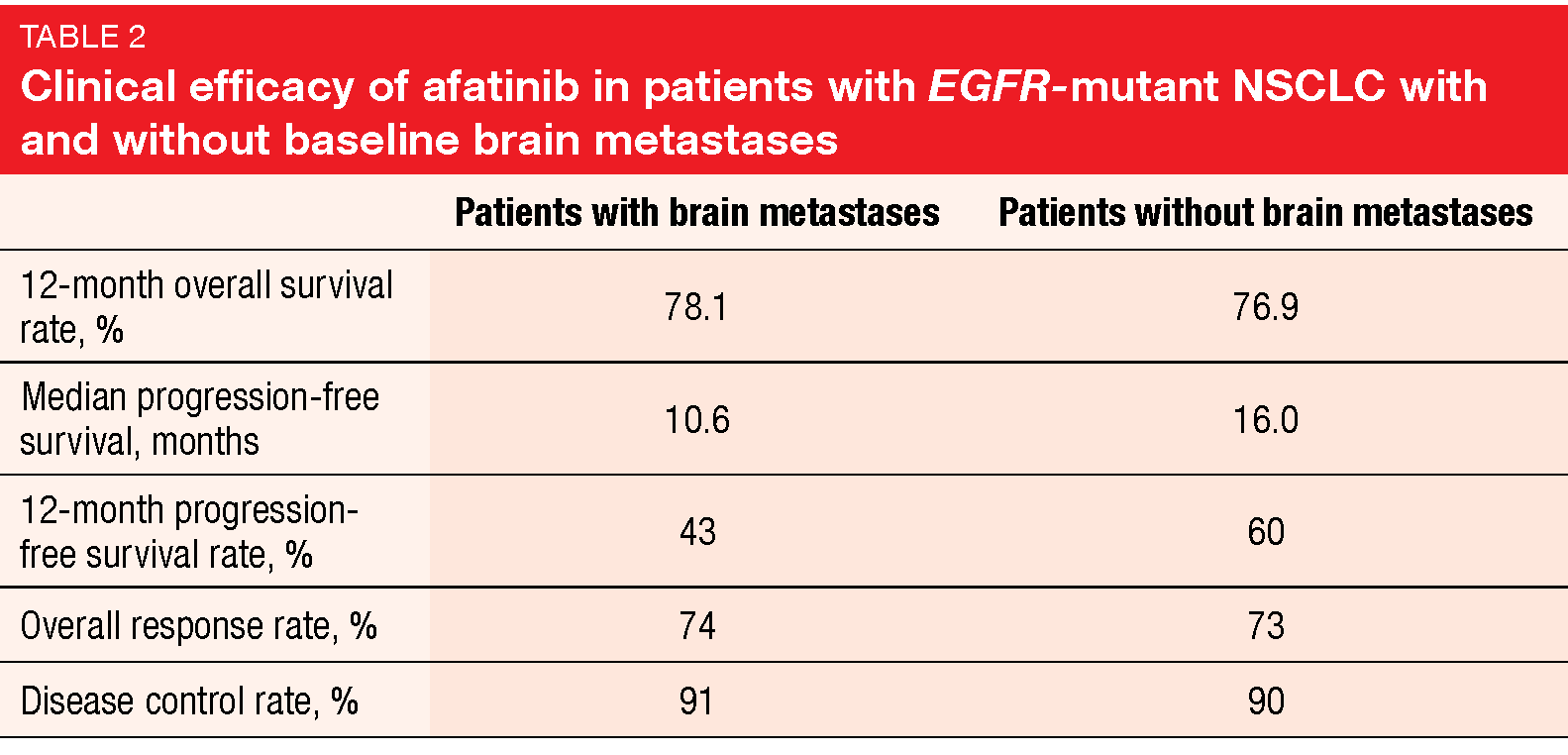
In patients with EGFR mutations, the proportion of NSCLC patients who develop brain metastases is as high as 40 % to 60 % [13-15]. A subanalysis of the non-interventional German GIDEON study that prospectively assessed the real-world use of afatinib in the setting of EGFR-mutant advanced NSCLC demonstrated activity of this treatment in patients with baseline brain metastases [16]. GIDEON included 49 patients with cerebral lesions (32.5% of the total population). Notably, this proportion is higher than in the randomized controlled trials conducted with afatinib [17, 18].
The presence or absence of brain metastases had no influence on ORR or DCR (Table 2). In line with the negative prognostic impact of CNS lesions, median PFS was shorter in patients with brain metastases than in those without (10.6 vs. 16.0 months). Median OS had not been reached yet at the time of the analysis. The safety results were consistent with those reported in the pivotal LUX-Lung clinical trials [18-20], and adverse events occurring during afatinib treatment did not differ across patients with and without brain metastases. Taken together, these data underline the efficacy and safety of afatinib in patients with CNS lesions, thus supporting the use of afatinib in this treatment setting.
REFERENCES
- Gibson AJW et al., Impact of number versus location of metastases on survival in stage IV M1b non-small cell lung cancer. Med Oncol 2018; 35(9): 117
- Steindl A et al., Impact of neurological symptom burden on the survival prognosis in a real-life cohort of patients with non-small cell lung cancer brain metastases. ESMO 2019, abstract 402PD
- Garon EB et al., Pembrolizumab for the treatment of non-small-cell lung cancer. N Engl J Med 2015; 372: 2018-2028
- Herbst RS et al., Pembrolizumab versus docetaxel for previously treated, PD-L1-positive, advanced non-small-cell lung cancer (KEYNOTE-010): a randomised controlled trial. Lancet 2016; 387: 1540-1550
- Reck M et al., Pembrolizumab versus chemotherapy for PD-L1-positive non-small-cell lung cancer. N Engl J Med 2016; 375: 1823-1833
- Mok TSK et al., Pembrolizumab versus chemotherapy for previously untreated, PD-L1-expressing, locally advanced or metastatic non-small-cell lung cancer (KEYNOTE-042): a randomised, open-label, controlled, phase 3 trial. Lancet 2019; 393: 1819-1830
- Mansfield AS et al., Outcomes with pembrolizumab monotherapy in patients with PD-L1-positive NSCLC with brain metastases: pooled analysis of KEYNOTE-001, 010, 024, and 042. ESMO 2019, abstract 1482O
- Powell SF et al., Pembrolizumab plus platinum-based chemotherapy in NSCLC with brain metastases: pooled analysis of KEYNOTE-021, 189, and 407. ESMO 2019, abstract 1483PD
- Gandhi L et al., Pembrolizumab plus chemotherapy in metastatic non-small-cell lung cancer. N Engl J Med 2018; 378(22): 2078-2092
- Langer CJ et al., Carboplatin and pemetrexed with or without pembrolizumab for advanced, non-squamous non-small-cell lung cancer: a randomised, phase 2 cohort of the open-label KEYNOTE-021 study. Lancet Oncol 2016; 17(11): 1497-1508
- Paz-Ares L et al., Pembrolizumab plus chemotherapy for squamous non-small-cell lung cancer. N Engl J Med 2018; 379(21): 2040-2051
- Borghaei H et al., 24-month overall survival from KEYNOTE-021 Cohort G: pemetrexed and carboplatin with or without pembrolizumab as first-line therapy for advanced nonsquamous non-small cell lung cancer. J Thorac Oncol 2019; 14(1): 124-129
- Iuchi T et al., Frequency of brain metastases in non-small-cell lung cancer, and their association with epidermal growth factor receptor mutations. Int J Clin Oncol 2015; 20: 674-679
- Hendriks LE et al., EGFR mutated non-small cell lung cancer patients: more prone to development of bone and brain metastases? Lung Cancer 2014; 84: 86-91
- Bhatt VR et al., Brain metastasis in patients with non-small-cell lung cancer and epidermal growth factor receptor mutations. J Clin Oncol 2013; 31: 3162-3164
- Laack E et al., Patients with brain metastases treated with afatinib in clinical practice – results from the prospective non-interventional study GIDEON. ESMO 2019, abstract 1536PD
- Schuler M et al., First-line afatinib versus chemotherapy in patients with non-small cell lung cancer and common epidermal growth factor receptor gene mutations and brain metastases. J Thorac Oncol 2016: 11; 380-390
- Park K et al., Afatinib versus gefitinib as first-line treatment of patients with EGFR mutation-positive non-small-cell lung cancer (LUX-Lung 7): a phase 2B, open-label, randomised controlled trial. Lancet Oncol 2016; 17(5): 577-589
- Wu Y-L et al., Afatinib versus cisplatin plus gemcitabine for first-line treatment of Asian patients with advanced non-small-cell lung cancer harbouring EGFR mutations (LUX-Lung 6): an open-label, randomised phase 3 trial. Lancet Oncol 2014; 15: 213-222
- Sequist LV et al., Phase III study of afatinib or cisplatin plus pemetrexed in patients with metastatic lung adenocarcinoma with EGFR mutations. J Clin Oncol 2013; 31: 3327-3334