Treatment of lung cancer: status quo & news from ASCO 2016
Robert Pirker, MD, Department of Internal Medicine I, Medical University of Vienna, Austria, discussed evidence from clinical trials for the treatment of lung cancer, with a focus on the findings presented at the ASCO 2016 Congress. As Dr. Pirker emphasised, a variety of topics was discussed at the conference, although none of the clinical trial results reported on this year have heralded fundamental changes in clinical practice.
Adjuvant chemotherapy of completely resected NSCLC
The implementation of adjuvant chemotherapy in lung cancer marks one of the major achievements over the last decades. One of the current goals is the identification of predictive biomarkers for patient selection. However, this approach has not been successful to date. “The IALT-Bio trial tried to establish biomarkers, and LACE-Bio aimed to validate them, but the validation has failed,” Dr. Pirker reported. At present, no predictive biomarkers are available. Another approach is customised chemotherapy, which is being tested in an Italian trial (NCT01784549). “Customised chemotherapy is complex, and it remains to be seen if this concept works,” Dr. Pirker said.
Thus far, the integration of targeted therapies into the adjuvant setting has also failed. This applies to the addition of bevacizumab to chemotherapy, to the MAGE-A3 vaccine, and to EGFR tyrosine kinase inhibitor (TKI) therapy in patients unselected for EGFR mutations. Two phase III trials are assessing adjuvant gefitinib compared to chemotherapy in patients with EGFR-mutation-positive tumours: ADJUVANT (NCT01405079), and WJOG6410L [1]. Disease-free survival is the endpoint in both studies. “These results will be available soon,” Dr. Pirker noted.
Also, the ongoing ADAURA study is investigating the third-generation EGFR TKI osimertinib versus placebo in patients with completely resected stage IB-IIIA NSCLC and exon 19 deletion or L858R mutation (NCT02511106). Patients with or without adjuvant chemotherapy are eligible. The ALCHEMIST trial aims at the identification of molecular markers in the adjuvant setting (NCT02194738) and evaluates adjuvant therapy with erlotinib in EGFR-mutation-positive patients, as well as treatment with crizotinib in ALK-positive patients.
Therapeutic options in stage III NSCLC
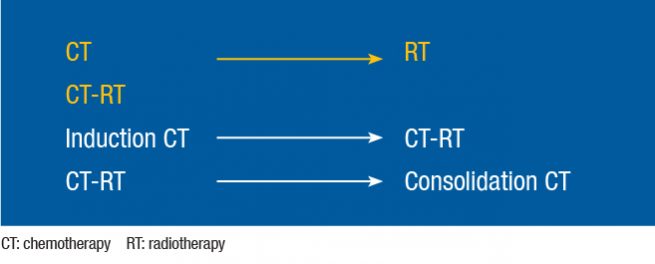
Locally advanced (stage III) NSCLC is a heterogeneous disease, the treatment of which usually involves radiotherapy and chemotherapy. These two strategies can be applied either sequentially or concomitantly (Figure 1). Induction chemotherapy can precede chemoradiotherapy, or a combined schedule can be followed by consolidation chemotherapy. Surgery is included for selected patients.
Figure 1: Administration of chemotherapy and radiotherapy in stage III NSCLC
Optimisation of stage III therapy has been attempted in different areas, with limited success. Prophylactic brain irradiation was tested in a trial based on the rationale that the central nervous system is frequently the first metastatic site. “This study had to be stopped because the patients declined receiving another treatment after completion of chemoradiotherapy,” Dr. Pirker reported. Vaccination did not meet expectations, either. The randomised, placebo-controlled, phase III START study revealed no overall survival (OS) advantage of tecemotide maintenance after chemoradiation [2].
A new concept is proton therapy, which allows for circumscribed application of irradiation doses, thus sparing other organs such as the heart. At the ASCO Congress, Liao et al. presented a Bayesian randomised trial comparing intensity-modulated radiation therapy and passively scattered proton therapy for locally advanced NSCLC [3]. Time to treatment failure as the primary endpoint did not differ between the two techniques. “A major issue with proton therapy is its costs,” Dr. Pirker stated. “Accordingly, the patients who resort to proton therapy are mostly wealthy, better educated, and live in urban areas that provide access to this method.”
Anti-EGFR agents in untreated patients with advanced NSCLC
EGFR mutations are present in 30 % to 60 % of Asian patients with adenocarcinomas, and in 10 % to 15 % of their Caucasian counterparts. For ALK rearrangement, these percentages range from 3 % to 5 %. While all EGFR TKIs prolong PFS, afatinib is the only EGFR TKI that has shown improved survival in patients with advanced EGFR-mutated NSCLC, compared to chemotherapy. The combined analysis of the LUX-Lung 3 and 6 trials yielded median survival times for patients with deletion 19 of 31.7 vs. 20.7 months (HR, 0.59; p = 0.0001; Figure 2) [4]. In contrast, for those with L858R mutations, the median survival times were 22.1 vs. 26.9 months (HR, 1.25; p = 0.16).
Figure 2: Highly significant overall survival improvement with first-line afatinib over chemotherapy according to the combined analysis of LUX-Lung 3 and LUX-Lung 6 [4]
The fully human anti-EGFR monoclonal antibody necitumumab was investigated in the SQUIRE trial in 1,093 patients with advanced squamous NSCLC [5]. This study compared necitumumab added to cisplatin plus gemcitabine with cisplatin plus gemcitabine alone. “OS was significantly prolonged in the necitumumab arm,” Dr. Pirker pointed out (11.5 vs. 9.9 months; stratified HR, 0.84; p = 0.01; Figure 3). Based on this pivotal trial, necitumumab was approved as first-line therapy for squamous EGFR-expressing NSCLC in combination with gemcitabine and cisplatin. Non-squamous patients were included in the INSPIRE trial that investigated cisplatin plus pemetrexed with or without necitumumab [6]. However, enrolment was prematurely stopped due to a lack of survival benefit of the combination, in addition to an increased rate of severe adverse events.
Figure 3: SQUIRE trial: overall survival benefit with necitumumab plus gemcitabine and cisplatin versus gemcitabine plus cisplatin alone [5]
Later lines and EGFR mutation positivity
The LUX-Lung 8 trial compared afatinib with erlotinib in patients with advanced squamous NSCLC who had progressed after at least four cycles of a first-line platinum-based doublet. Afatinib therapy led to improved survival compared to erlotinib (median, 7.9 vs. 6.8 months; HR, 0.81; p = 0.0077) [7].
Patients with EGFR-mutation-positive tumours who undergo EGFR TKI therapy will ultimately develop resistance. At that time, re-biopsy is recommended. Treatment options at the time of resistance to the first-generation and second-generation TKIs include a switch to chemotherapy, possibly followed by TKI re-challenge, use of third-generation EGFR TKIs, and continuation of the original TKI therapy. Local interventions can also be added to the TKI treatment. Another option is combined administration of afatinib and cetuximab.
At present, the third-generation EGFR TKIs are osimertinib, rociletinib and olmutinib. These agents target EGFR-activating mutations and the T790M mutation, while sparing wild-type EGFR. “The sparing of wild-type EGFR ensures improved tolerability due to decreased side effects, such as rash or diarrhoea,” Dr. Pirker explained. Osimertinib has already been approved in locally advanced or metastatic T790M-positive disease. The AURA3 trial is comparing osimertinib with platinum-based chemotherapy after progression on EGFR TKI therapy in T790M-positive disease (NCT02151981). “The OS results will be available soon, and these will provide information on the choice between TKI therapy and chemotherapy after disease progression,” Dr. Pirker noted.
Encouraging results have been obtained with olmutinib in a phase I/II trial in Korean patients [8]. “Tumour responses in T790M-positive patients occurred in 61 %, and disease control was achieved in 90 %.” In the meantime, the clinical development of rociletinib has been stopped due to toxicity and modest efficacy.
Next-generation agents in ALK-positive disease
Alectinib is an ALK inhibitor with activity against ALK-resistance mutations. In the Japanese J-ALEX study, it gave rise to improved PFS compared to crizotinib as first-line treatment in patients with ALK-positive disease (not reached vs. 10.2 months; HR, 0.34; p > 0.0001) [9]. “We are waiting for the results of the global ALEX trial, which is comparing alectinib with crizotinib.” Dr. Pirker explained (NCT02075840). “These findings will shed light on the optimal first-line strategy in ALK-positive NSCLC.”
Brigantinib, another next-generation ALK inhibitor with broad inhibitory activity, was tested at two doses in the ALTA trial [10]. Patients who had ex-
perienced progression on crizotinib received either 180 mg or 90 mg brigantinib. The objective response rates were 54 % and 45 %, respectively; disease control was achieved in 86 % and 82 %, respectively. In the 180 mg dose group, the median PFS exceeded 1 year (12.9 months).
Ramucirumab and nintedanib in second-line treatment of advanced NSCLC
In the second-line setting of advanced NSCLC, treatments with ramucirumab, the triple angiokinase inhibitor nintedanib, the irreversible ErbB family blocker afatinib, and immune checkpoint inhibitors constitute major recent advances. Ramucirumab was tested in addition to docetaxel in the REVEL trial [11]. As compared to the control arm, where the patients received docetaxel only, the combination gave rise to a 14 % reduction in mortality risk. Nintedanib was assessed in two phase III trials. The LUME-Lung 1 study investigated docetaxel with or without nintedanib in patients with all histological subtypes [12]. “While the addition of nintedanib significantly improved PFS in the total cohort, a significant OS benefit occurred only in the subgroup with adenocarcinoma,” Dr. Pirker reported (10.9 vs. 7.9 months; HR, 0.75; p = 0.0073).
LUME-Lung 2 compared nintedanib plus pemetrexed with pemetrexed alone in patients with non-squamous NSCLC [13]. The results favoured the combination in terms of both progression-free survival (PFS; 4.4 vs. 3.6 months; HR, 0.83; p = 0.04) and disease control (61 % vs. 53 %; p = 0.039), whereas OS did not differ between the two arms. “Nintedanib has been approved in Europe for second-line treatment of locally advanced or metastatic adenocarcinoma in combination with docetaxel,” Dr. Pirker said.
Immune checkpoint inhibitors in advanced NSCLC
Immune checkpoint inhibitors have demonstrated superior efficacy compared to docetaxel in second-line treatment of patients with advanced NSCLC. Side effects include immune-related pneumonitis, colitis, hepatitis, nephritis, endocrinopathies and infusion-related events. “Approval and reimbursement of nivolumab and pembrolizumab have been implemented in many countries,” Dr. Pirker said.
The anti-PD-1 antibody nivolumab has shown OS benefits compared to docetaxel in advanced squamous NSCLC (CheckMate 017) [14] and adenocarcinomas (CheckMate 057) [15]. The degree of PD-L1 expression appears to predict the benefit of immune checkpoint inhibitors. The anti-PD-1 antibody pembrolizumab showed superiority over docetaxel in the KEYNOTE-010 trial, which enrolled patients with PD-L1 expression on at least 1 % of tumour cells [16]. OS was significantly longer for both doses of pembrolizumab (HRs, 0.71 and 0.61, for 2 mg/kg and 10 mg/kg, respectively). “This difference was even more pronounced in patients with at least 50 % of tumour cells expressing PD-L1,” Dr. Pirker noted.
Similarly, the efficacy of the anti-PD-L1 antibody atezolizumab is greatest in the biomarker-enriched population, according to the POPLAR trial [17]. Here, baseline PD-L1 expression on tumour cells and tumour-infiltrating immune cells was assessed using immunohistochemistry (IHC). Atezolizumab significantly improved OS compared with docetaxel. “The survival advantage correlated with PD-L1 assessment that considered expression on both cell types,” Dr. Pirker reported.
Phase III trials on first-line treatment with immune checkpoint inhibitors are ongoing in advanced NSCLC. KEYNOTE-024, which includes 305 patients with PD-L1 expression of ≥ 50 %, is comparing pembrolizumab with platinum-based chemotherapy (NCT02142738). “According to a press release issued in June 2016, pembrolizumab treatment has demonstrated superior PFS and OS,” Dr. Pirker said [18]. KEYNOTE-042 is also testing pembrolizumab versus chemotherapy, although in patients with a broader range of PD-L1 expression (≥ 1 %) (NCT02220894). The primary outcome is OS.
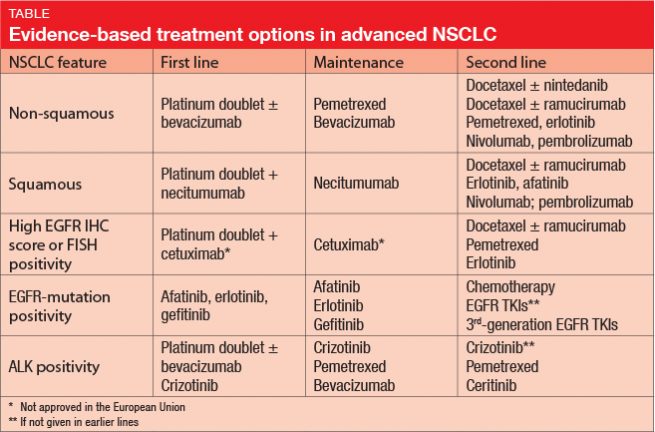
Suggested algorithm for advanced NSCLC
Dr. Pirker presented his algorithm for first-line, maintenance and second-line treatments of advanced NSCLC (Table). The choice of treatment varies according to histological features (non-squamous, squamous) and the presence of EGFR IHC score or FISH positivity, EGFR-mutation or ALK positivity. As Dr. Pirker pointed out, the choices presented in the Table are evidence based, although some have not been approved in the European Union.
Small-cell lung cancer (SCLC)
The management of SCLC poses specific challenges. “Available treatments are often not being administered to patients,” Dr. Pirker stressed. “According to the SEER data, only 50 % of patients with SCLC receive chemotherapy.”
SCLC is radiosensitive, but the ideal radiotherapy is under discussion. Here, the CONVERT trial that was presented at the ASCO Congress has shown that once-daily thoracic irradiation with 66 Gy resulted in similar survival compared to twice-daily radiotherapy with 45 Gy [19]. The authors concluded that both regimens can be used.
Immunotherapy has found its way into the treatment of SCLC. The CheckMate 032 trial has established the activity of nivolumab plus ipilimumab in the second-line setting [20]. CheckMate 451 is currently assessing this combination as maintenance therapy in extensive-stage SCLC after first-line platinum-based doublet chemotherapy (NCT02538666).
Promising results have been obtained with the DLL3-targeted antibody–drug conjugate rovalpituzumab tesirine (Rova-TTM). In the SCRX16-001 study, clinical benefit rates were 68 % and 89 % in the total cohort and in the biomarker-selected group (DLL3 expression of ≥ 50 %), respectively [21]. Thirty-two percent of patients with pronounced DLL3 expression were alive at 1 year.
Tiseo et al. showed that the addition of bevacizumab to cisplatin and etoposide in first-line treatment of extensive SCLC gives rise to significantly improved PFS compared to the chemotherapy regimen alone, but OS did not differ between the two arms [22].
REFERENCES
- Tada H et al., Vinorelbine plus cisplatin versus gefitinib in resected non-small cell lung cancer harboring activating EGFR mutation (WJOG6410L). J Clin Oncol 30, 2012 (suppl; abstr TPS7110)
- Butts C et al., Tecemotide (L-BLP25) versus placebo after chemoradiotherapy for stage III non-small-cell lung cancer (START): a randomised, double-blind, phase 3 trial. Lancet Oncol 2014; 15(1): 59-68
- Liao ZX et al., Bayesian randomized trial comparing intensity modulated radiation therapy versus passively scattered proton therapy for locally advanced non-small cell lung cancer. J Clin Oncol 34, 2016 (suppl; abstr 8500)
- Yang JC et al., Afatinib versus cisplatin-based chemotherapy for EGFR mutation-positive lung adenocarcinoma (LUX-Lung 3 and LUX-Lung 6): analysis of overall survival data from two randomised, phase 3 trials. Lancet Oncol 2015; 16(2): 141-151
- Thatcher N et al., Necitumumab plus gemcitabine and cisplatin versus gemcitabine and cisplatin alone as first-line therapy in patients with stage IV squamous non-small-cell lung cancer (SQUIRE): an open-label, randomised, controlled phase 3 trial. Lancet Oncol 2015; 16: 763-774
- Paz-Ares L et al., Necitumumab plus pemetrexed and cisplatin as first-line therapy in patients with stage IV non-squamous non-small-cell lung cancer (INSPIRE): an open-label, randomised, controlled phase 3 study. Lancet Oncol 2015; 16(3): 328-337
- Soria J-C et al., Afatinib versus erlotinib as second-line treatment of patients with advanced squamous cell carcinoma of the lung (LUX-Lung 8): an open-label randomised controlled phase 3 trial. Lancet Oncol 2015; 16: 897-907
- Park K et al., Olmutinib (BI 1482694; HM61713), an EGFR mutant-specific inhibitor, in T790M+ NSCLC: efficacy and safety at the RP2D. J Clin Oncol 34, 2016 (suppl; abstr 9055)
- Nokihara H et al., Alectinib versus crizotinib in ALK inhibitor naïve ALK-positive non-small cell lung cancer: primary results from the J-ALEX study. J Clin Oncol 34, 2016 (suppl; abstr 9008)
- Kim D-W et al., Brigatinib in patients with crizotinib-refractory ALK+ non-small cell lung cancer: first report of efficacy and safety from a pivotal randomized phase 2 trial (ALTA). J Clin Oncol 34, 2016 (suppl; abstr 9007)
- Garon EB et al., Ramucirumab plus docetaxel versus placebo plus docetaxel for second-line treatment of stage IV non-small-cell lung cancer after disease progression on platinum-based therapy (REVEL): a multicentre, double-blind, randomised phase 3 trial. Lancet 2014; 384(9944): 665-673
- Reck M et al., Docetaxel plus nintedanib versus docetaxel plus placebo in patients with previously treated non-small-cell lung cancer (LUME-Lung 1): a phase 3, double-blind, randomised controlled trial. Lancet Oncol 2014; 15(2): 143-155
- Hanna NH et al., Lume-lung 2: A multicenter, randomized, double-blind, phase III study of nintedanib plus pemetrexed versus placebo plus pemetrexed in patients with advanced nonsquamous non-small cell lung cancer (NSCLC) after failure of first-line chemotherapy. J Clin Oncol 31, 2013 (suppl; abstr 8034)
- Brahmer J et al., Nivolumab versus docetaxel in advanced squamous-cell non-small-cell lung cancer. N Engl J Med 2015; 373(17):
123-135 - Borghaei H et al., Nivolumab versus docetaxel in advanced nonsquamous non-small-cell lung cancer. N Engl J Med 2015; 373(17): 1627-1639
- Herbst RS et al., Pembrolizumab versus docetaxel for previously treated, PD-L1-positive, advanced non-small-cell lung cancer (KEYNOTE-010): a randomised controlled trial. Lancet 2016; 387(10027): 1540-1550
- Fehrenbacher L et al., Atezolizumab versus docetaxel for patients with previously treated non-small-cell lung cancer (POPLAR): a multicentre, open-label, phase 2 randomised controlled trial. Lancet 2016; 387(10030): 1837-1846
- www.mercknewsroom.com, Press release, Thursday, June 16, 2016 6:45 am EDT
- Faivre-Finn C et al., Concurrent ONce-daily Versus twice-daily RadioTherapy: a 2-arm randomised controlled trial of concurrent chemo-radiotherapy comparing twice-daily and once-daily radiotherapy schedules in patients with limited-stage small cell lung cancer and good performance status. J Clin Oncol 34, 2016 (suppl; abstr 8504)
- Antonia SJ et al., CheckMate 032: nivolumab alone or in combination with ipilimumab for the treatment of recurrent small cell lung cancer. J Clin Oncol 34, 2016 (suppl; abstr 100)
- Rudin CM et al., Safety and efficacy of single agent rovalpituzumab tesirine (SC16LD6.5), a delta-like protein 3 (DLL3)-targeted antibody-drug conjugate (ADC) in recurrent or refractory small cell lung cancer (SCLC). J Clin Oncol 34, 2016 (suppl; abstr LBA8505)
- Tiseo M et al., Italian multicenter phase III randomized study of cisplatin-etoposide with or without bevacizumab as first-line treatment in extensive stage small cell lung cancer (SCLC): GOIRC-AIFA FARM6PMFJM trial. J Clin Oncol 34, 2016 (suppl; abstr 8513)


![Figure 2: Highly significant overall survival improvement with first-line afatinib over chemotherapy according to the combined analysis of LUX-Lung 3 and LUX-Lung 6 [4]](https://memoinoncology.com/wp-content/uploads/2020/04/Grafik-10-preceptorship-vienna-en.jpg)
![Figure 3: SQUIRE trial: overall survival benefit with necitumumab plus gemcitabine and cisplatin versus gemcitabine plus cisplatin alone [5]](https://memoinoncology.com/wp-content/uploads/2020/04/Grafik-11-preceptorship-vienna-en.jpg)