Further defining the optimal use of immune checkpoint inhibitors
Neoadjuvant evaluation of nivolumab
As the anti-PD-1 antibody nivolumab is known to induce deep and durable responses in a subset of lung cancer patients, this agent was investigated in the neoadjuvant setting, which is an area of unmet need. There have been no advances in systemic treatment of resectable lung cancer since 2004. Chaft et al. hypothesised that neoadjuvant nivolumab treatment might induce immunity against micrometastases [1]. Newly diagnosed patients with resectable stage I (> 2 cm)/II/IIIA NSCLC received two doses of nivolumab 3 mg/kg, on days 14 and 28, followed by surgical resection. In the post-operative setting, standard-of-care treatment was administered. Safety and feasibility constituted the primary endpoints of this study. Out of 22 patients enrolled, 21 received neoadjuvant treatment, and tumour resection was performed for 20, as one patient was nonresectable due to tracheal invasion.
For the primary endpoint of feasibility, this trial demonstrated that nivolumab treatment did not delay or interfere with surgery in any of these patients. No unexpected safety signals occurred. Drug-related adverse events were restricted to grades 1 and 2, with the exception of one case of pneumonia that led to cancellation of the second dose of nivolumab. However, surgery was not delayed in this patient. One death in the postoperative safety period was unrelated to the study drug (sequelae of a traumatic fall).
Induction of T cells specific for mutation-associated neoantigen
Four weeks after neoadjuvant nivolumab treatment, radiographic evaluation per RECIST v1.1 showed that out of 21 patients, two (10 %) and 18 (85 %) obtained partial responses and stable disease, respectively. Only one (5 %) developed progression. Assessment of pathological responses in the surgery specimens revealed major pathological response (MPR; defined as ≤ 10 % viable tumour cells) in 9 out of 21 cases (43 %). PD-L1 positivity prior to treatment did not correlate with MPR. To date, median postoperative followup is 12 months. Two of 20 resected patients have recurred (one solitary brain metastasis, one systemic relapse), but none of the patients with MPR experienced relapse. One patient who was not resected died of lung cancer.
Correlative studies were conducted in a subset of tumours. These showed that mutation burden and neoantigen density are associated with pathological response to the neoadjuvant treatment. T cells specific for dominant mutationassociated neoantigen (MANA) were identified in the blood and the tumour. They expanded in the blood upon neoadjuvant administration of nivolumab. The authors concluded that temporal increases in MANA-specific T-cell receptors in the peripheral blood after nivolumab treatment might be a biomarker of nivolumab response.
Nivolumab plus ipilimumab: two-year update of CheckMate 012
The multicohort CheckMate 012 trial evaluated nivolumab alone or in combination with other agents, including ipilimumab, as first-line treatment of advanced NSCLC. It demonstrated encouraging clinical activity; for instance, patients experienced high response rate and durable responses [2, 3]. Goldman et al. presented the 2-year OS results and other up-dated findings from the nivolumab plus ipilimumab combination cohorts [4]. In these arms, patients received nivolumab 3 mg/kg every 2 weeks plus ipilimumab 1 mg/kg either every 6 weeks (n = 39) or every 12 weeks (n = 38).
The pooled results from the two cohorts showed continued clinical benefit with nivolumab plus ipilimumab in all of the patients and in those with ≥ 1 % and ≥ 50 % PD-L1 expression. For all of the treated patients, the 2-year OS rate was 49 %, and for those with ≥ 1 % PD-L1 expression, 58 %. The 2-year PFS rates for these groups were 29 % and 38 %, respectively. A total of 34 (44 %) patients in the cohorts receiving nivolumab plus ipilimumab every 6 and 12 weeks lived for at least 2 years. This was achieved in patients with diverse histology, smoking status, EGFR mutation status, PD-L1 expression, and best overall response to treatment. However, efficacy was enhanced with increasing PD-L1 expression. It was noted that a subset of patients who discontinued therapy had sustained responses in the absence of treatment. Nivolumab plus ipilimumab remained tolerable, and most treatment-related AEs were manageable. No new safety concerns occurred with longer follow-up.
Three-year survival with pembrolizumab as a single agent
The effects of the PD-1 antibody pembrolizumab were first demonstrated in the large multicohort phase Ib KEYNOTE- 001 study that assessed pembrolizumab monotherapy for previously treated and treatment-naive patients with melanoma and advanced NSCLC [5, 6]. Overall, 550 NSCLC patients were enrolled. Of these, 101 were treatmentnaive, and 449 had received previous therapy.
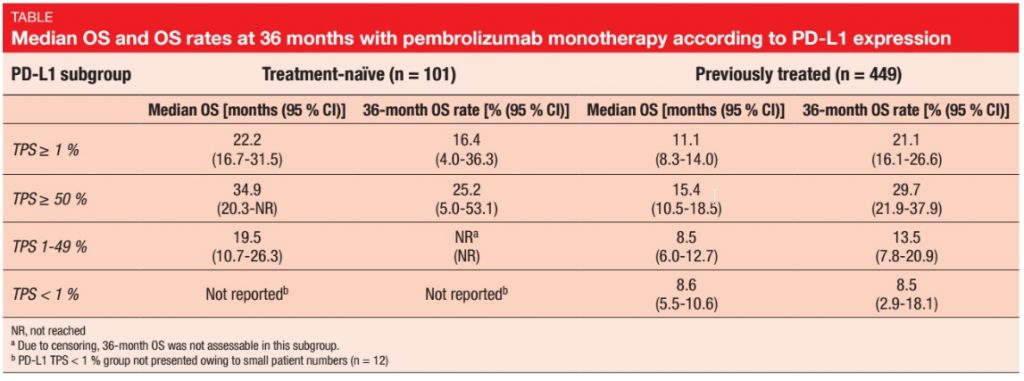
As the 3-year analysis of KEYNOTE- 001 showed, pembrolizumab at 2 mg/kg or 10 mg/kg every 3 weeks or 10 mg/kg every 2 weeks provided longterm OS benefit for both first-line and pre-treated patients with advanced NSCLC expressing PD-L1 [7]. At 36 months, 26.4 % and 19.0 % of first-line and pre-treated patients, respectively, were alive. For PD-L1 expression status, the subgroup with tumour proportion score (TPS) ≥ 50 % derived greater benefits from treatment than the cohorts with lower PD-L1 expression (Table). Pembrolizumab therapy had favourable effects across subgroups, as defined by various baseline clinical characteristics (i.e., smoking history, histology, EGFR mutation status, prior radiation). The long-term findings did not suggest any cumulative immune-mediated toxicity or late-onset grade 3 to 5 AEs. These data represent the longest efficacy and safety follow-up for patients with advanced NSCLC who have received pembrolizumab treatment.
PFS2 in KEYNOTE-024
The KEYNOTE-024 trial tested fixeddose pembrolizumab (200 mg every 3 weeks for 2 years) in patients with untreated stage IV NSCLC and a PD-L1 TPS ≥ 50 %, compared to platinum-doublet chemotherapy. Overall, 305 patients were randomised. According to the primary analysis that was conducted after a median follow-up of 11.2 months, both PFS and OS were highly significantly in favour of the immunotherapeutic agent, with HRs of 0.50 and 0.60, respectively (p < 0.001, p = 0.005, respectively) [8].
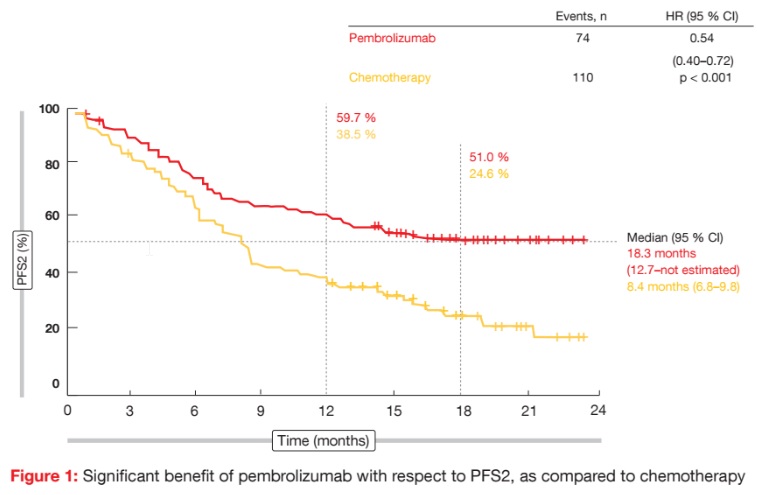
The analysis presented at the ASCO Congress related to the PFS in the second line (PFS2); i.e., the time from randomisation to progression of disease (PD) per investigator review after the start of second-line therapy or death, whichever occurred first [9]. Patients who were alive without PD on secondline therapy were censored at the time of the last known survival without PD, while those who died without PD and those who discontinued the secondline therapy were counted as events. In KEYNOTE-024, 79 chemotherapy-treated patients crossed over to pembrolizumab on study, and 12 received anti-PD-1 treatment outside of the crossover, which made for a 60.3 % effective crossover rate. Forty-eight and 97 patients in the pembrolizumab and chemotherapy arms, respectively, received subsequent therapy of any type. Median duration of second-line therapy was 3.6 and 3.5 months, respectively. Pembrolizumab-treated patients experienced significantly better outcomes, with a median PFS2 of 18.3 months (vs. 8.4 months; HR, 0.54; p < 0.001; Figure 1). At 18 months, PFS2 rates were 51.0 % and 24.6 %, respectively. This means that patients with PD-L1 expression ≥ 50 % have better survival if the treatment is started with pembrolizumab rather than with a platinumdoublet chemotherapy. The analysis also included updated OS outcomes. Here, pembrolizumab continued to show significantly improved results (median OS, not reached vs. 14.5 months; HR, 0.63; p = 0.003). At 18 months, 61.2 % versus 43.0 % of patients were alive in the two treatment arms. As was noted, a high degree of separation of the OS curves was maintained despite the effective crossover rate of 60 %. Along with a favourable safety profile, the findings of the current analysis support pembrolizumab as a standard-ofcare for first-line treatment of NSCLC with PD-L1 TPS ≥ 50 %.
Incorporating pembrolizumab into first-line chemotherapy
The addition of pembrolizumab to firstline chemotherapy with pemetrexed and carboplatin showed favourable ORR and PFS when compared with chemotherapy alone in cohort G of the open-label, randomised, phase I/II KEYNOTE-021 trial [10]. At the time of the primary analysis, the HR for OS was 0.90.
Based on 5 months of additional follow- up, pembrolizumab plus chemotherapy continued to be more effective than standard chemotherapy in patients with treatment-naive, advanced, nonsquamous NSCLC, irrespective of PDL1 expression [11]. ORR was almost doubled (56.7 % vs. 30.2 %; p = 0.0016), and the risk of progression or death was halved (not reached vs. 8.9 months; HR, 0.50; p = 0.0038). Furthermore, a trend towards a greater OS benefit emerged with longer follow-up in spite of the high crossover rate of 75 %. The reduction in mortality risk due to the addition of pembrolizumab was 31 % (HR, 0.69; p = 0.13), and the 12-month OS rates amounted to 76.0 % and 69.3 %. The combination proved tolerable, with readily manageable safety profile.
According to the investigators, pembrolizumab plus chemotherapy with pemetrexed and carboplatin represents an effective and tolerable treatment option as initial therapy for patients with advanced non-squamous NSCLC. Consequently, this combination has been granted accelerated approval by the US Food and Drug Administration.
OAK: atezolizumab beyond disease progression
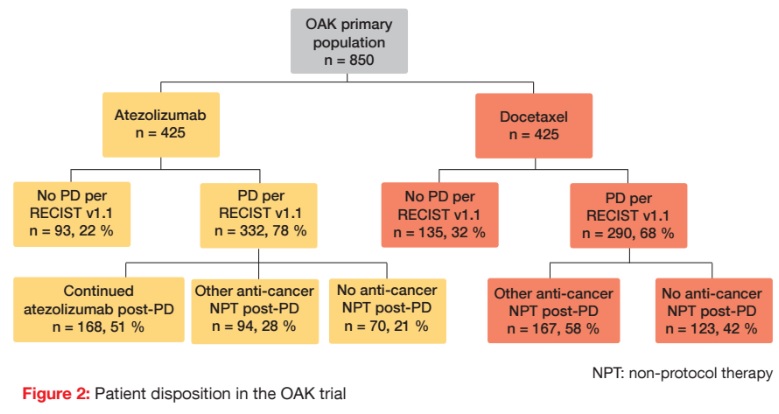
The randomised phase III OAK study evaluated the PD-L1 antibody atezolizumab in the second-line setting [12]. Patients with locally advanced or metastatic NSCLC after one to two lines of chemotherapy that included at least one platinum-based regimen and any PDL1 status received either atezolizumab (n = 425) or docetaxel (n = 425). Treatment beyond progression was allowed in the atezolizumab arm, as long as the patients were deriving clinical benefit, based on the protocol-defined criteria. The objective of the analysis presented at the ASCO Congress was the determination of the benefit–risk profile of atezolizumab treatment beyond progression according to RECIST v1.1 [13]. This was based on the consideration that RECIST v1.1-based endpoints such as ORR and PFS tend to underestimate the potential OS benefit of checkpoint inhibitors. There had been discordance regarding endpoints in the OAK trial, which demonstrated OS benefit of atezolizumab, but no improvements in ORR or PFS. The investigators hypothesised that immunotherapy might alter tumour biology in a way that extends survival benefit beyond radiographic progression. Overall, 78 % and 68 % of patients treated in the experimental and control arms, respectively, experienced progression per RECIST (Figure 2).
Prolongation of post-PD OS
This analysis, which included the first treatment-beyond-progression OS from a phase III study of immunotherapy in advanced NSCLC, indicated clinical benefit of continued atezolizumab administration. Among atezolizumab- treated patients experiencing PD per RECIST, 51 % (n = 168) received atezolizumab therapy beyond progression, while 28 % (n = 94) were treated with other anticancer therapies, and 21 % (n = 70) with no anticancer therapy (Figure 2). According to the analysis, 7 % of patients who continued to receive atezolizumab had subsequent responses in target lesions (i. e., ≥ 30 % reduction post-PD), and 49 % had stable target lesions (i.e., best change between + 20 % and – 30 %). Post-PD tumour reduction or stability was observed across all of the PD-L1 expression subgroups. Atezolizumab treatment beyond progression showed a tolerable safety profile.
Within the group of patients with PD, those who continued to receive atezolizumab fared best with regard to post-PD OS. At 18 months, their OS rate was 37 % compared to 20 % in the group undergoing other anti-cancer therapies and 9 % in the patients who were not treated with anti-cancer agents. Median OS for these 3 groups was 12.7 months, 8.8 months and 2.2 months, respectively. In the docetaxel arm, patients who had post-PD immunotherapy showed better OS outcomes than those without immunotherapy (median, 17.3 vs. 7.5 months). Here, at 18 months, the OS rates were 42 % versus 12 %.
The researchers concluded that these findings support the concept of postprogression prolongation of survival and they highlight the inadequacy of RECIST v1.1 to capture the full clinical benefit of cancer immunotherapy. As these findings might be biased, they are no more than hypothesis generating, and confirmation in a randomised clinical trial is needed.
Determinants of response to immune checkpoint inhibitors
As anti-PD-(L)1 therapies are revolutionising treatment and outcomes for lung cancer patients, determinants of response and resistance are eagerly sought, with the objective to improve patient selection. Hellmann et al. used targeted next-generation sequencing with MSK-IMPACT to generate molecular profiling data for 240 patients [14]. These showed that tumour mutation burden correlated with improved benefit through anti-PD-(L)1 agents, particularly at higher thresholds. Moreover, the fraction of genome altered (i. e., quantified normalised percentage of genes with copy number loss or amplification) showed an inverse association with therapeutic benefit. This also applied to variants in individual genes, such as EGFR and STK11. Both tumour mutation burden and fraction of genome altered were reasonably estimated by the MSK-IMPACT test, while whole exome sequencing appeared to be better suited for the assessment of molecular signatures and various other molecular features.
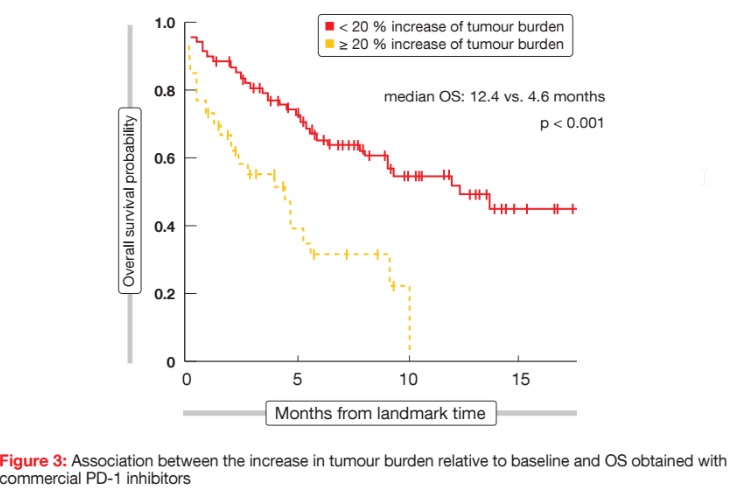
Nishino et al. evaluated tumour burden dynamics in 160 patients with advanced NSCLC who received nivolumab or pembrolizumab monotherapy, with the purpose being to identify imaging markers for clinical benefit of this treatment [15]. Here, 25 % of patients achieved objective responses or durable disease control. Using an 8-week landmark analysis, the researchers demonstrated that patients with < 20 % tumour burden increase from baseline had longer OS than patients with increases of ≥ 20 % (Figure 3). According to Cox models, patients whose tumour burden stayed below a 20 % increase from base- line throughout therapy had significantly reduced mortality risk (HR, 0.24; p < 0.0001), after adjusting for smoking and baseline tumour burden. Tumour burden increase of < 20 % might therefore be a practical marker of clinical benefit that can be validated prospectively in a larger cohort.
References:
- Chaft JE et al., Neoadjuvant nivolumab in early-stage, resectable non-small cell lung cancers. ASCO 2017, abstract 8508
- Hellmann MD et al., Nivolumab plus ipilimumab as first-line treatment for advanced nonsmall- cell lung cancer (CheckMate 012): results of an open-label, phase 1, multicohort study. Lancet Oncol 2017; 18: 31-41
- Gettinger S et al., First-line nivolumab monotherapy and nivolumab plus ipilimumab in patients with advanced NSCLC: long-term outcomes from CheckMate 012. WCLC 2016, abstract OA3.01
- Goldman JW et al., Nivolumab plus ipilimumab as first-line treatment for advanced NSCLC: 2-yr OS and long-term outcomes from CheckMate 012. ASCO 2017, abstract 9093
- Garon EB et al., Pembrolizumab for the treatment of non-small-cell lung cancer. N Engl J Med 2015; 372(21): 2018-2028
- Chatterjee M et al., Systematic evaluation of pembrolizumab dosing in patients with advanced non-small-cell lung cancer. Ann Oncol 2016; 27(7): 1291-1298
- Leighl NB et al., KEYNOTE-001: 3-year overall survival for patients with advanced NSCLC treated with pembrolizumab. ASCO 2017, abstract 9011
- Reck M et al., Pembrolizumab versus chemotherapy for PD-L1-positive non-small-cell lung cancer. N Engl J Med 2016; 375: 1823-1833
- Brahmer J et al., Progression after the next line of therapy (PFS2) and updated OS among patients (pts) with advanced NSCLC and PD-L1 tumor proportion score (TPS) ≥50% enrolled in KEYNOTE-024. ASCO 2017, abstract 9000
- Langer CJ et al., Carboplatin and pemetrexed with or without pembrolizumab for advanced, non-squamous non-small-cell lung cancer: a randomised, phase 2 cohort of the open-label KEYNOTE-021 study. Lancet Oncol 2016; 17(11): 1497-1508
- Papadimitrakopoulou VA et al., First-line carboplatin and pemetrexed with or without pembrolizumab for advanced nonsquamous NSCLC: Updated results of KEYNOTE-021 cohort G. ASCO 2017, abstract 9094
- Barlesi F et al., Primary analysis from OAK, a randomized phase III study comparing atezolizumab with docetaxel in 2L/3L NSCLC. ESMO 2016, abstract LBA44_PR
- Gandara DR et al., Atezolizumab treatment beyond disease progression in advanced NSCLC: results from the randomized phase III OAK study. ASCO 2017, abstract 9001
- Hellmann MD et al., Molecular determinants of response and resistance to anti-PD-(L)1 blockade in patients with NSCLC profiled with targeted next-generation sequencing (NGS). ASCO 2017, abstract 9015
- Nishino M et al., Tumor response dynamics of advanced non-small-cell lung cancer (NSCLC) patients (pts) treated with commercial PD-1 inhibitors in the clinical setting. ASCO 2017, abstract 9087