Immune checkpoint inhibition: comprehensive benefits, but not devoid of risks
Three-year findings in CheckMate 227
First-line nivolumab plus ipilimumab (NI) was shown to significantly prolong OS compared to chemotherapy in patients with advanced NSCLC irrespective of tumor PD-L1 expression in the randomized, phase III CheckMate 227 study [1]. At the ASCO Congress, Ramalingam et al. presented the updated 3-year efficacy and safety results from Part 1 of the trial [2]. Part 1 consisted of Part 1a that compared NI (n = 396) with chemotherapy (n = 397) and nivolumab monotherapy (n = 396), as well as Part 1b which assessed NI (n = 187) vs. chemotherapy (n = 186) and nivolumab plus chemotherapy (n = 177). Patients in Part 1a showed PD-L1 expression ≥ 1 %, while those in Part 1b tested PD-L1–negative (< 1 %).
At three years, first-line NI continued to provide long-term benefits compared to chemotherapy regardless of PD-L1 expression. In Part 1a, 3-year OS rates were 33 % vs. 22 % with the combination and chemotherapy, respectively (HR, 0.79). For Part 1b, these were 34 % vs. 15 % (HR, 0.64). More than one third of all responders remained in response after three years with NI (38 % and 34 % for PD-L1 expressors ≥ 1 % and < 1 %, respectively), while the respective rates in the chemotherapy group ranged below 5 %. Also, the combination showed lasting superiority over both nivolumab monotherapy and nivolumab plus chemotherapy independent of PD-L1 expression.
An exploratory landmark analysis assessed the impact of response at six months on long-term OS. This showed that among patients with PD-L1 ≥ 1 %, 70 % of those achieving complete or partial responses at six months with NI were alive at three years; in the chemotherapy arm, this applied only to 39 %. Similar results were observed for the group with PD-L1 < 1 % (82 % vs. 25 %). The extended safety follow-up over at least 36.3 months did not reveal any new signals for the combination. Treatment-related select AEs of NI affecting the skin, gastrointestinal tract, endocrine system and other areas decreased over time. The authors concluded that NI is a novel chemotherapy-sparing first-line treatment option for patients with advanced NSCLC. In May 2020, the regimen has received approval in the USA in this indication, while the European application was withdrawn four months earlier.
Two cycles of chemotherapy plus checkpoint blockade: CheckMate 9LA
The phase III, randomized CheckMate 9LA trial was conducted based on the assumption that adding a limited course of chemotherapy to first-line NI might provide rapid disease control while building on the durable OS benefit observed with NI in CheckMate 227. Among 719 patients with stage-IV or recurrent NSCLC, 361 received NI combined with two cycles of chemotherapy. In the control arm (n = 358), four cycles of chemotherapy were administered, followed by optional pemetrexed maintenance in patients with non-squamous tumors. OS was defined as the primary endpoint.
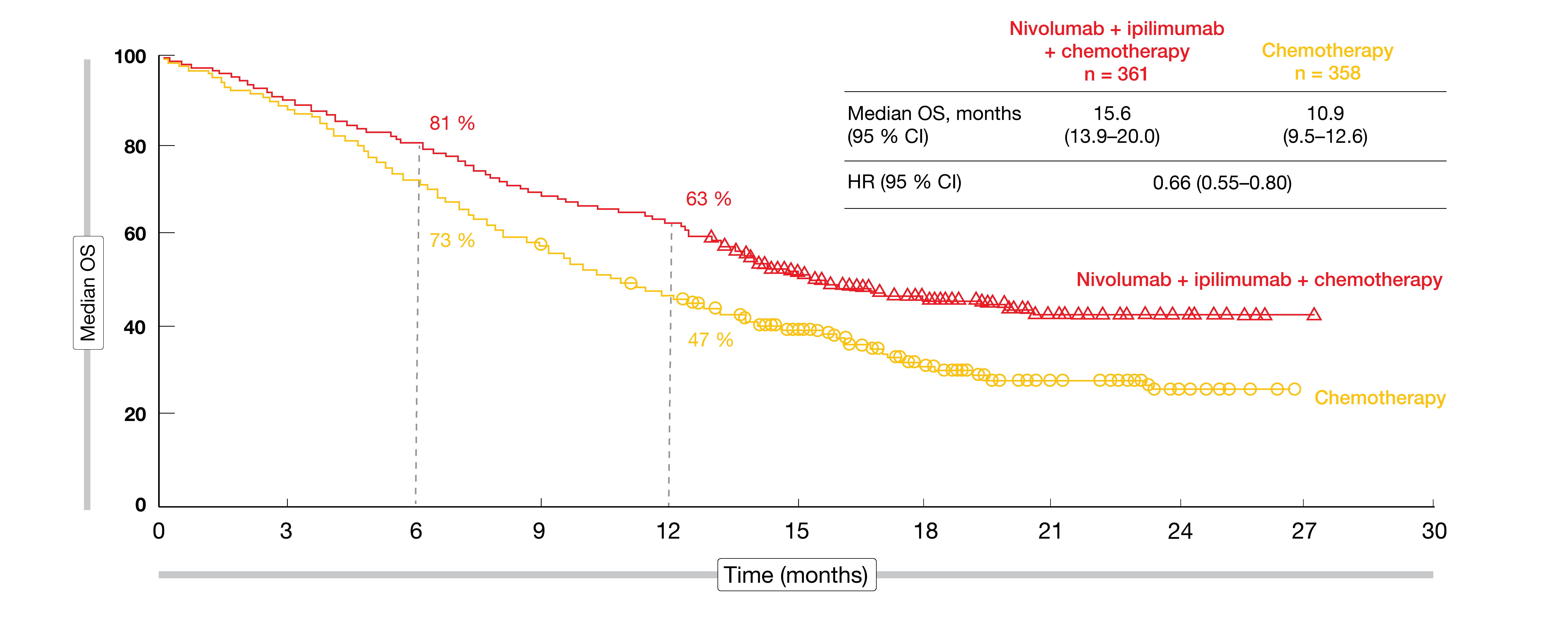
After a minimum follow-up of 8.1 months, CheckMate 9LA met its primary endpoint at the time of the pre-planned interim OS analysis, with a statistically significant benefit for the immunotherapy-based regimen compared to chemotherapy only (14.1 vs. 10.7 months; HR, 0.69; p = 0.0006) [3]. The OS advantage increased over time; according to updated results obtained after a follow-up of 12.7 months, median OS was 15.6 vs. 10.9 months (HR, 0.66; Figure 1). Patients fared better regarding survival with the immunotherapy-based approach regardless of histology (squamous vs. non-squamous) and PD-L1 expression (< 1 %, ≥ 1 %, 1-49 %, ≥ 50 %). At 12 months, PFS rates were 33 % vs. 18 % (HR, 0.68), and 49 % vs. 24 % patients responded to treatment.
The combination did not induce any new AEs; any-grade treatment-related AEs mainly included nausea, anemia, asthenia, and diarrhea. Immune-related AEs were mostly grades 1 and 2. Overall, the CheckMate 9LA study demonstrated that NI plus a limited course of chemotherapy should be considered as a new first-line treatment option for patients with advanced NSCLC. Indeed, approval in the USA has been granted by the Food and Drug Administration in May 2020.
Figure 1: CheckMate 9LA: sustained overall survival benefit of nivolumab plus ipilimumab and chemotherapy versus chemotherapy
Durvalumab/tremelimumab ± chemotherapy
Another trial to assess the combined first-line approach of dual checkpoint inhibition and chemotherapy in stage IV NSCLC is the international, randomized phase II CCTG BR.34 study [4]. Patients were allocated to either durvalumab plus tremelimumab followed by durvalumab maintenance (n = 150) or the same regimen plus platinum doublet chemotherapy followed by durvalumab alone or combined with pemetrexed, depending on histology (n = 151). Although the addition of chemotherapy did not prolong OS (HR, 0.88), the combined strategy led to significant benefits compared to the immunotherapy-only approach with respect to PFS (7.7 vs. 3.2 months; HR, 0.67; p = 0.0035) and ORR (p = 0.033).
The effect of the addition of chemotherapy appeared to be greater in patients with blood tumor mutational burden (bTMB) < 20 mutations/Mb; however, the interaction test was negative. According to the investigators, this finding warrants further evaluation in randomized studies. Irrespective of the type of treatment, patients with bTMB ≥ 20 mut/Mb had longer OS and PFS than those with bTMB < 20 mut/Mb, which suggested a prognostic (rather than a predictive) effect. PD-L1 levels were not associated with differential benefit from the addition of chemotherapy. More patients experienced serious AEs in the experimental arm, although the incidence of immune-related AEs was similar between the two groups. Analyses of quality of life, plasma genomics and cost in this trial are ongoing.
Final analysis of KEYNOTE-189
In the randomized, double-blind, phase III KEYNOTE-189 trial, pembrolizumab plus platinum-based chemotherapy first demonstrated significantly improved OS and PFS over placebo plus chemotherapy in untreated patients with metastatic non-squamous NSCLC regardless of PD-L1 expression [5, 6]. At ASCO 2020, Rodriguez-Abreu et al. presented the protocol-specified final analysis of KEYNOTE-189 after a median of 31.0 months from randomization to data cut-off [7].
With long-term follow-up, pembrolizumab plus pemetrexed/platinum continued to improve the efficacy outcomes over chemotherapy alone. Median OS was approximately twice as long in the experimental arm as in the control arm (22.0 vs. 10.6 months; HR, 0.56). This also applied to PFS (9.0 vs. 4.9 months; HR, 0.49) and PFS2, i.e. PFS after the next line of therapy (17.0 vs. 9.0 months; HR; 0.50). Objective responses resulted in 48.3 % vs. 19.9 %. PD-L1 expression did not affect any of these outcomes. ORR was high at 85.7 % in the group of patients in the experimental arm who completed 35 cycles of pembrolizumab (n = 56); this included four complete responders. Median OS had not been reached in this cohort yet.
The authors concluded that pembrolizumab plus pemetrexed/platinum is a standard-of-care therapy for patients with newly diagnosed metastatic non-squamous NSCLC.
Anti-TIGIT antibody tiragolumab
The co-inhibitory receptor TIGIT is expressed on multiple immune cells and inhibits T cells as well as NK cells by binding to its ligand on tumor and antigen-presenting cells [8-10]. It was hypothesized that anti-TIGIT antibodies preventing this binding reaction might restore the anti-tumor response and complement the activity of anti-PD-(L)1 antibodies. Indeed, the anti-TIGIT monoclonal antibody tiragolumab showed activity in combination with atezolizumab in the phase I GO30103 trial (NCT02794571). Based on these observations, the randomized, double-blind, phase II CITYSCAPE study assessed tiragolumab plus atezolizumab (n = 67) compared to placebo plus atezolizumab (n = 68) in the first-line treatment of patients with stage IV, PD-L1–expressing NSCLC [11].
ORR and PFS were defined as the co-primary endpoints. After a median follow-up of 10.9 months, tiragolumab plus atezolizumab, as compared to placebo and atezolizumab, induced clinically meaningful improvements in ORR (37 % vs. 21 %) and PFS (5.55 vs. 3.88 months; HR, 0.58) in the ITT population. Of note, both ORR and PFS benefits were observed in patients with PD-L1 tumor proportion scores (TPS) ≥ 50 %, but not in those with TPS 1-49 % (Table). Duration of response and OS are not yet mature.
Tiragolumab plus atezolizumab was well tolerated, with a safety profile similar to that of the comparator regimen. Although immune-mediated toxicity occurred more frequently in the experimental arm, these AEs were primarily grades 1 or 2 and were manageable. SKYSCRAPER-01, an ongoing phase III study, is aiming to confirm the observed activity and safety of tiragolumab plus atezolizumab in untreated patients with PD-L1–expressing (TPS ≥ 50 %) NSCLC.
Pneumonitis as an underreported AE
Although immunotherapy is of increasing importance in the treatment of patients with advanced NSCLC, potentially life-threatening AEs such as checkpoint inhibitor pneumonitis (CIP) need to be dealt with and might deserve more consideration. Spieler et al. hypothesized that CIP might be underreported in patients with advanced NSCLC receiving nivolumab monotherapy and that radiomics features can identify CIP which has been clinically misclassified [12]. Within an Institutional Review Board-approved database, 9 of 159 nivolumab-treated NSCLC patients (5 %) had been diagnosed with any-grade CIP. Forty additional patients without a CIP diagnosis were randomly selected from the same population by the investigators. In all 49 cases, uninvolved lung in the last pre-immunotherapy CT imaging study was segmented, delineated, and analyzed for radiomics features associated with CIP. A logistic regression model incorporating radiomics assigned a CIP probability score to each patient.
Six radiomics features were shown to correlate with CIP. The radiomics-based probability model assigned seven out of 40 patients (17.5 %) without a clinical diagnosis of CIP a > 50 % probability of CIP. The chart review revealed that six of these seven patients exhibited symptoms or radiographic features highly suggestive of CIP. Therefore, it appears that the incidence of CIP is underreported and radiomics features can help to identify cases that have been clinically misclassified. Future directions for research include expansion of this study across the full database, correlation of radiomics features with blood biomarkers, and the inclusion of tumor burden and radiotherapy as additional covariates in the analysis.
Durvalumab rechallenge after pneumonitis
Saito et al. investigated the timing, clinical course, severity, management, and clinical outcomes of pneumonitis/radiation pneumonitis among patients with locally advanced NSCLC who received chemoradiotherapy after the approval of durvalumab in the real-world setting [13]. This retrospective study conducted at 17 Japanese centers involved consecutive patients who started concurrent chemoradiotherapy between May 2018 and May 2019. The analysis population comprised 275 individuals. Durvalumab consolidation was performed in 74.2 % of cases.
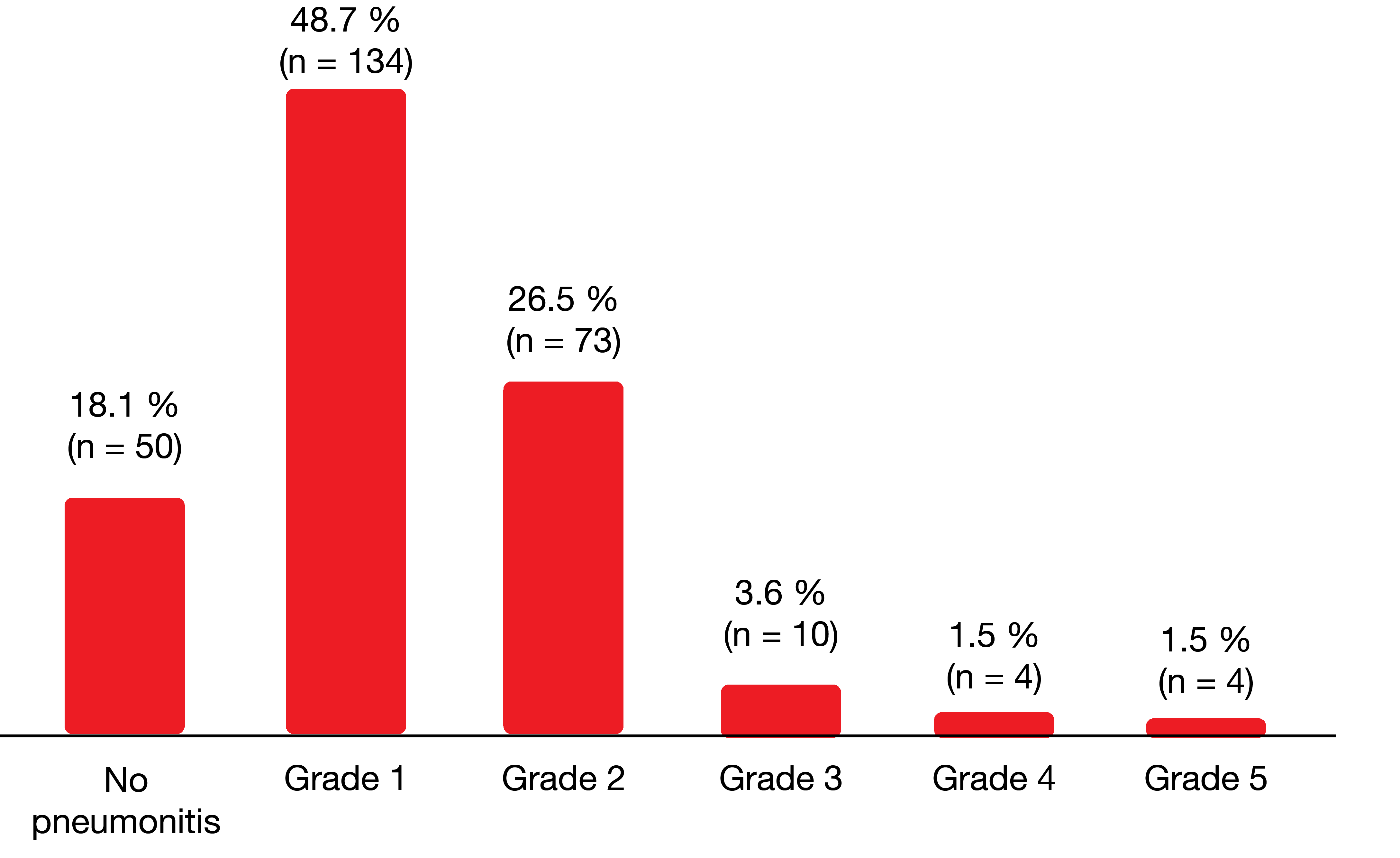
More than 80 % of patients developed pneumonitis that was mostly grades 1 (48.7 %) and 2 (26.5 %), although in 1.5 %, pneumonitis was fatal (Figure 2). Thirty-three percent of patients experienced pneumonitis with any symptoms (≥ grade 2); here, the lung volume receiving doses of ≥ 20 Gy (V20) was identified as an independent risk factor. In 6 %, grade ≥ 3 pneumonitis occurred. Thirteen patients (5 %) required home oxygen therapy after pneumonitis. Median onset of pneumonitis was 14 weeks after the initiation of chemoradiotherapy and approximately four to seven weeks after the initiation of durvalumab treatment.
Among patients who developed pneumonitis during durvalumab consolidation, steroid therapy was administered in 25 %. In this group, a durvalumab rechallenge was performed in 41 %, with the majority of patients experiencing no relapse of pneumonitis. Half of those who did relapse did not require treatment interruption and had no fatal relapse or chronic respiratory failure. The authors noted that with careful consideration, durvalumab rechallenge might be an option after corticosteroid therapy for pneumonitis.
Figure 2: Incidence and severity of pneumonitis in patients with stage III NSCLC treated with chemoradiation and durvalumab consolidation
REFERENCES
- Hellmann MD et al., Nivolumab plus ipilimumab in advanced non-small-cell lung cancer. N Engl J Med 2019; 381: 2020-2031
- Ramalingam SS et al., Nivolumab + ipilimumab versus platinum-doublet chemotherapy as first-line treatment for advanced non-small cell lung cancer: Three-year update from CheckMate 227 Part 1. J Clin Oncol 38: 2020 (suppl; abstr 9500
- Reck M et al., Nivolumab + ipilimumab + 2 cycles of platinum-doublet chemotherapy vs. 4 cycles chemo as first-line treatment for stage IV/recurrent non-small cell lung cancer (NSCLC): CheckMate 9LA. J Clin Oncol 38: 2020 (suppl; abstr 9501)
- Leighl NB et al., CCTG BR.34: A randomized trial of durvalumab and tremelimumab +/- platinum-based chemotherapy in patients with metastatic (stage IV) squamous or nonsquamous non-small cell lung cancer (NSCLC). J Clin Oncol 38: 2020 (suppl; abstr 9502)
- Gandhi L et al., Pembrolizumab plus chemotherapy in metastatic non-small-cell lung cancer. N Engl J Med 2018; 378(22): 2078-2092
- Gadgeel S et al., Updated analysis from KEYNOTE-189: Pembrolizumab or placebo plus pemetrexed and platinum for previously untreated metastatic nonsquamous non-small-cell lung cancer. J Clin Oncol 2020; 38(14): 1505-1517
- Rodriguez-Abreu D et al., Protocol-specified final analysis of KEYNOTE-189: pemetrexed-platinum chemotherapy with or without pembrolizumab in patients with previously untreated metastatic nonsquamous NSCLC. J Clin Oncol 38: 2020 (suppl; abstr 9582)
- Manieri NA et al., TIGIT: A key inhibitor of the cancer immunity cycle. Trends Immunol 2017; 38(1): 20-28
- Rotte A et al., Mechanistic overview of immune checkpoints to support the rational design of their combinations in cancer immunotherapy. Ann Oncol 2018; 29(1): 71-83
- Yu X et al., The surface protein TIGIT suppresses T cell activation by promoting the generation of mature immunoregulatory dendritic cells. Nat Immunol 2009; 10: 48-57
- Rodriguez-Abreu D et al., CITYSCAPE: Primary analysis of a randomized, double-blind, phase II study of the anti-TIGIT antibody tiragolumab plus atezolizumab versus placebo plus atezo as first-line treatment in patients with PD-L1-selected NSCLC. J Clin Oncol 38: 2020 (suppl; abstr 9503)
- Spieler B et al., Is checkpoint inhibitor pneumonitis underreported in patients with advanced non-small cell lung cancer (NSCLC) on PD-1 inhibitor monotherapy? J Clin Oncol 38: 2020 (suppl; abstr 9579)
- Saito G et al., Real-world survey of pneumonitis/radiation pneumonitis among locally advanced NSCLC with chemoradiotherapy after the approval of durvalumab: A multicenter retrospective cohort study (HOPE-005/CRIMSON). J Clin Oncol 38: 2020 (suppl; abstr 9039)