Current perspectives in EGFR-targeted therapy
Global phase IIIb study assessing afatinib
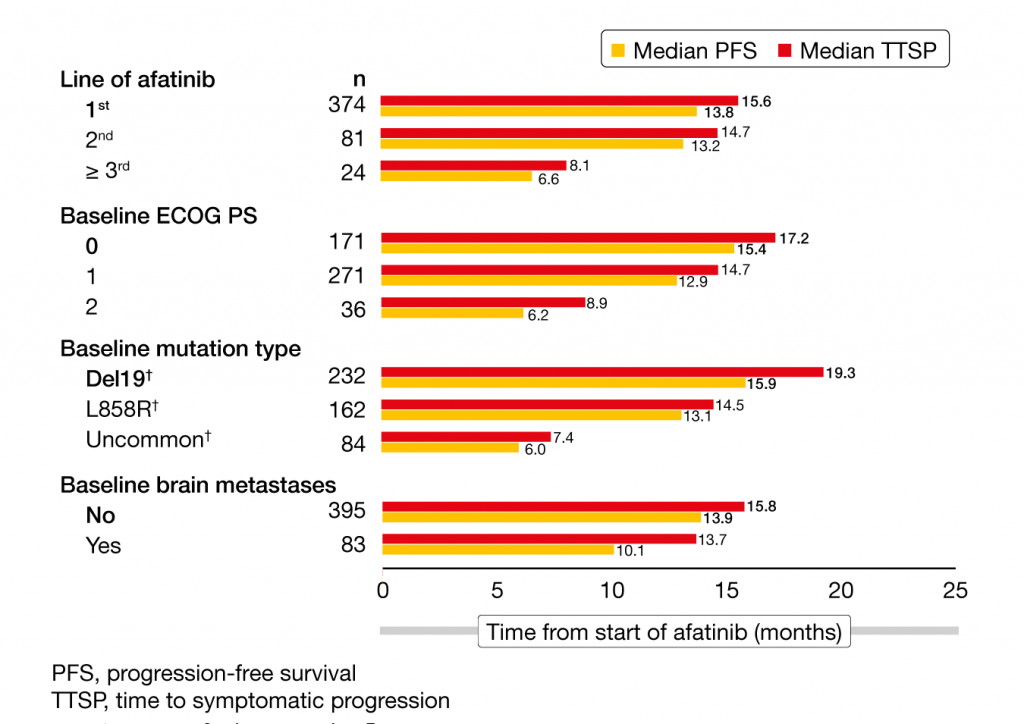
The second-generation, irreversible ErbB family blocker afatinib has been established as a first-line standard option in patients with EGFR-mutant NSCLC based on the phase III LUX-Lung 3 and 6 trials that revealed significant progression-free survival (PFS) and objective response rate (ORR) improvement compared to platinum-doublet chemotherapy [1, 2]. Moreover, the phase IIb LUX-Lung 7 study showed significant benefits regarding PFS, ORR and time to treatment failure with afatinib compared to the first-generation EGFR tyrosine kinase inhibitor (TKI) gefitinib [3]. An open-label, multicenter, phase IIIb trial conducted in Europe, Israel and Australia is currently assessing afatinib 40 mg/day until disease progression in the real-world setting. Two dose reductions to 30 mg or 20 mg/day can be performed based on individual tolerability. Asymptomatic brain metastases are allowed. A total of 479 patients were included in the interim analysis presented at ELCC 2019 [4]. Seventeen percent of these had brain metastases, 18 % had uncommon EGFR mutations, with exon 20 insertions representing the most frequent type, and the ECOG performance status was 1 and 2 in 57 % and 8 %, respectively. Seventy-eight percent and 17 % received afatinib as first- and second-line therapy, respectively. Thus, the population reflected real-life conditions. Safety was defined as the primary endpoint of the interim analysis, with PFS, time to symptomatic progression (TTSP), ORR and disease control rate (DCR) defined as efficacy endpoints. Overall, the results were consistent with those observed for afatinib in the LUX-Lung 3, 6 and 7 trials. Afatinib showed a predictable and manageable safety profile. Diarrhea and rash were the most common adverse events (AEs) and the main reasons for dose reductions, which became necessary in 54 %. In 8 %, patients had AEs leading to treatment discontinuation, most commonly diarrhea. Each of the other AEs causing discontinuation accounted for < 1 % of these cases. Also, the interim analysis revealed encouraging efficacy findings, with median PFS of 13.37 months and TTSP of 14.91 months. The activity of afatinib in patients with brain metastases was confirmed; this group had median PFS and TTSP of 10.1 and 13.7 months, respectively (Figure 1).
Figure 1: Median PFS and TTSP with afatinib in real-world practice according to line of treatment, performance status, mutation type, and presence of brain metastases
GIDEON: real-world evidence from Germany
The prospective, non-interventional GIDEON study investigated the efficacy and tolerability of first-line afatinib when used in routine clinical practice in Germany. GIDEON enrolled a high proportion of patients aged ≥ 70 years, thus providing an opportunity to study the real-world use of afatinib in older individuals. PFS at 12 months constituted the primary endpoint, while PFS, overall survival (OS), ORR and DCR were secondary outcomes. Overall, 151 patients were treated, 67 (44 %) of whom were ≥ 70 years old. Among these elderly patients, 22 % had brain metastases at inclusion, and rare mutations were present in 18 %. Brückl et al. reported the results from a post-hoc analysis of older patients included in the first interim analysis of the study [5]. These findings supported the use of afatinib in the elderly, as the outcomes even exceeded those obtained in the overall study population. The 12-month PFS rate amounted to 62 % (12-month PFS in the entire population, 54.6 %), with median PFS of 17.3 months (vs. 12.9 months). ORR and DCR were 78 % and 93 %, respectively (vs. 73 % and 90 %, respectively). Furthermore, the safety profile of afatinib in elderly patients was comparable to that seen in the younger subgroup. Grade ≥ 3 treatment-emergent AEs were similar in patients aged ≥ 70 years and < 70 years, with diarrhea occurring most commonly. There was a trend towards lower starting doses in older patients, with 40 mg used as initial treatment in 62 % vs. 83 % for the younger cohort, although the percentages of patients requiring dose reductions appeared comparable across age groups (55 % vs. 58 %).
First-line outcomes for osimertinib
Two phase I expansion cohorts of the AURA study evaluating first-line treatment with the third-generation, irreversible EGFR TKI osimertinib have yielded robust ORR and prolonged PFS in patients with locally advanced or metastatic EGFR-mutant NSCLC [6]. Sequential patient cohorts of 30 patients each received osimertinib 80 mg or 160 mg/day until progression. The final efficacy and safety data from these expansion cohorts were presented by Yang et al. at ELCC 2019 [7]. Osimertinib 80 mg showed durable clinical activity and manageable tolerability. Confirmed ORRs were 67 % and 87 % for the 80 mg and 160 mg cohorts, respectively. Median duration of response was 19.3 and 16.7 months, respectively, and median PFS 22.1 and 19.3 months, respectively. The safety profile of osimertinib 80 mg was consistent with previous reports. AEs prompting dose reductions occurred in 35 % of patients overall and were more common in the 160 mg cohort than in the 80 mg cohort (53 % vs. 17 %). In total, 12 % of patients discontinued treatment due to AEs. The investigators concluded that the final results support the use of osimertinib as first-line therapy for patients with EGFR-mutant NSCLC. The 80 mg dose was confirmed as the optimal and therefore recommended dose.
Four-drug activity after EGFR TKI failure
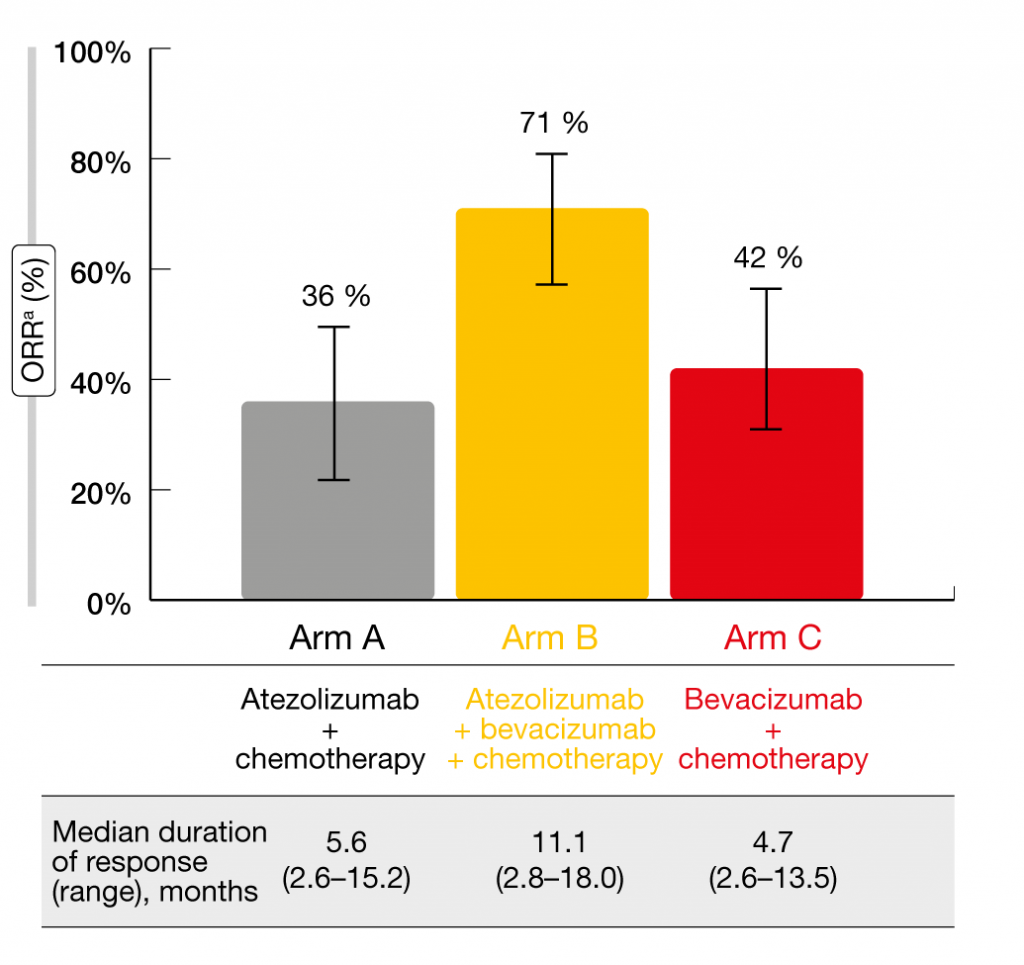
There is an urgent unmet need after failure of first-line EGFR TKIs, as these patients have limited treatment options. A potential approach is the concomitant use of anti-angiogenic agents and immunotherapy in addition to chemotherapy. The randomized, three-arm IMpower150 trial assessed the combination of the anti-PD-L1 antibody atezolizumab, carboplatin/paclitaxel and the anti-VEGF antibody bevacizumab (Arm B) compared to atezolizumab plus chemotherapy (Arm A) and chemotherapy plus bevacizumab (Arm C). IMpower150 was conducted in an all-comer population with stage IV or recurrent metastatic non-squamous NSCLC (n = 1,202) and showed significant benefits for both PFS and OS in Arm B vs. Arm C [8, 9]. The study included patients with EGFR mutations who had experienced progression or intolerance to treatment with at least one approved targeted agent (n = 124). Out of these, 45, 34, and 45 were treated in Arms A, B, and C, respectively. At ELCC 2019, Reck et al. presented an exploratory analysis of efficacy outcomes in EGFR-positive patients [10]. This showed that the four-drug combination of atezolizumab, carboplatin, paclitaxel and bevacizumab improved clinical outcomes in the EGFR-mutant group. Arm B experienced prolonged PFS compared to Arm C (10.2 vs. 6.9 months), which also applied to OS (median OS, not reached vs. 18.7 months). The addition of atezolizumab to bevacizumab and chemotherapy increased both PFS and OS benefits across all subgroups of EGFR-positive patients. Moreover, patients in Arm B fared best with respect to ORR (71 % vs. 36 % and 42 % in Arms A and C, respectively; Figure 2) and duration of response (11.1 vs. 5.6 and 4.7 months, respectively). In contrast, comparisons across Arms A and C did not yield any significant differences regarding PFS or OS. The safety profile observed in the EGFR-positive patients was comparable to that seen in the ITT population, which was also true for immune-related AEs. Treatment-related AEs occurred slightly more often in Arm B compared to Arms A and C. According to these results, the combination of atezolizumab, bevacizumab and platinum-based chemotherapy might represent a potential new option for patients with EGFR-mutant lung cancer in whom EGFR TKI treatment has failed.
Figure 2: Doubling of response rates and duration of response with atezolizumab plus bevacizumab and chemotherapy compared to the other treatment arms
Mechanisms of resistance
PD-L1 not only negatively regulates T cell function, but also leads to acquired resistance to EGFR TKIs in the NSCLC setting. Zhang et al. evaluated the mechanistic role of PD-L1 in primary resistance to EGFR TKI treatment [11]. Based on in-vitro studies and a xenograft mouse model, the researchers found that PD-L1 is responsible for primary resistance to gefitinib. Overexpression of PD-L1 attenuated sensitivity to gefitinib in vivo. In all, it appeared that PD-L1 contributes to primary resistance to EGFR TKIs in EGFR-mutant NSCLC, which might be mediated through induction of epithelial-mesenchymal transition. This data suggested that PD-L1–targeting immunotherapies are a promising strategy to restore sensitivity to EGFR TKI treatment in primary resistance. Anlotinib is a novel multi-target receptor TKI that shows activity in the inhibition of tumor angiogenesis and growth. A subset of patients with advanced NSCLC who are refractory to EGFR TKIs are sensitive to anlotinib treatment. Using different types of lung cancer cell lines and a xenograft model, Lian et al. elucidated the mechanisms underlying this effect [12]. They found that FGFR1 contributes to acquired resistance, while anlotinib suppresses proliferation, apoptosis and cell cycles by inhibiting the FGFR1 signaling pathway. FGFR1 might therefore be a potential therapeutic target in patients with EGFR-mutant NSCLC and acquired resistance.
Resistance to third-generation treatment
Fassunke et al. provided new insights into acquired resistance mechanisms to third-generation EGFR TKI therapy [13]. The investigators analyzed pre- and post-treatment samples of 124 patients who had developed the T790M resistance on early-generation EGFR TKI therapy and were treated with third-generation TKIs. There was a high prevalence of additional genetic aberrations potentially mediating innate and acquired resistance to third-generation agents. Co-occurring aberrations were found in 75 % of the samples. TP53 mutations were most frequent among these but had no impact on third-generation TKI treatment. Genetic changes in the samples with acquired resistance to third-generation EGFR TKIs emerged in the EGFR gene (e.g., T790M loss, acquisition of C797S) and in other genes (e.g., MET amplification, KRAS mutations). Amplification of MET showed a strong association with primary treatment failure and was thus the strongest factor of innate resistance. Loss of T790M and MET amplification represented the most common aberrations after third-generation TKI treatment. A new EGFR resistance mutation after third-generation TKI therapy is G724S. The authors noted that acquired resistance due to G724S can possibly be overcome using second-generation EGFR tyrosine kinase inhibitors.
REFERENCES
- Sequist LV et al., Phase III study of afatinib or cisplatin plus pemetrexed in patients with metastatic lung adenocarcinoma with EGFR mutations. J Clin Oncol 2012; 31(27): 3327-3334
- Wu YL et al., Afatinib versus cisplatin plus gemcitabine for first-line treatment of Asian patients with advanced non-small-cell lung cancer harbouring EGFR mutations (LUX-Lung 6): an open-label, randomised phase 3 trial. Lancet Oncol 2014; 15(2): 213-222
- Park K et al., Afatinib versus gefitinib as first-line treatment of patients with EGFR mutation-positive non-small-cell lung cancer (LUX-Lung 7): a phase 2B, open-label, randomised controlled trial. Lancet Oncol 2016; 17(5): 577-589
- Passaro A et al., Afatinib in EGFR TKI-naïve patients with locally advanced/metastatic NSCLC harbouring EGFR mutations: an interim analysis of a phase IIIB trial. ELCC 2019, abstract 1150
- Brückl WM et al., Elderly patients treated with afatinib in clinical practice – results from the prospective non-interventional study GIDEON. ELCC 2019, abstract 125P
- Ramalingam SS et al., Osimertinib as first-line treatment of EGFR mutation-positive advanced non-small-cell lung cancer. J Clin Oncol 2018; 36(9): 841-849
- Yang JCH et al., Osimertinib as first-line (1L) treatment for epidermal growth factor receptor (EGFR) mutation-positive advanced non-small cell lung cancer (NSCLC): final efficacy and safety results from two phase I expansion cohorts. ELCC 2019, abstract 122P
- Reck M et al., Primary PFS and safety analyses of a randomised phase III study of carboplatin + paclitaxel ± bevacizumab, with or without atezolizumab in 1L non-squamous metastatic NSCLC (IMpower150). ESMO 2017, LBA1_PR
- https://www.roche.com/media/releases/med-cor-2018-03-26.htm
- Reck M et al., IMpower150: an exploratory analysis of efficacy outcomes in patients with EGFR mutations. ELCC 2019, abstract 1040
- Zhang Y et al., PD-L1 confers primary resistance to EGFR TKI in EGFR mutant non-small cell lung cancer via inducing EMT phenotype. ELCC 2019, abstract 138P
- Lian Z et al., Anlotinib can overcome acquired resistance to EGFR-TKIs via FGFR1 signaling in non-small cell lung cancer. ELCC 2019, abstract 129P
- Fassunke J et al., New insights into acquired resistance mechanisms to third-generation EGFR tyrosine kinase inhibitor therapy in lung cancer. ELCC 2019, abstract 132P