Rare driver mutations: BRAF- and HER2-mutant NSCLC
First-line dabrafenib plus trametinib
BRAF driver mutations in NSCLC are rare at 2 % [1, 2], but tumours with BRAFV600E mutations have histological features suggestive of aggressive biology [3]. When treated with platinum-based chemotherapy, these patients showed less favourable outcomes [3, 4].
The multi-cohort, non-randomised, phase II BRF113928 study investigated a targeted approach using the BRAF inhibitor dabrafenib and the MEK inhibitor trametinib in patients with advanced BRAFV600E-mutated NSCLC. This trial contained a dabrafenib monotherapy arm (Cohort A) as well as two combination arms (Cohorts B and C) that received dabrafenib 150 mg twice daily plus trametinib 2 mg daily. Cohort B consisted of pre-treated patients, while Cohort C contained a treatment-naïve population. The primary endpoint for each cohort was investigator-assessed ORR. An independent review committee (IRC) reviewed responses according to RECIST.
The indirect comparison across Cohorts A and B demonstrated higher ORR and longer median PFS with the combined administration of dabrafenib and trametinib than with dabrafenib monotherapy in pre-treated patients. [5, 6]. At the ESMO 2017 Congress, Planchard et al. presented findings obtained in the treatment-naïve Cohort C that comprised 36 patients with stage IV NSCLC [7]. This is the first study of combined BRAF and MEK inhibition as first-line therapy in patients with BRAFV600E-mutant metastatic NSCLC.
Fifty-one percent alive at 2 years
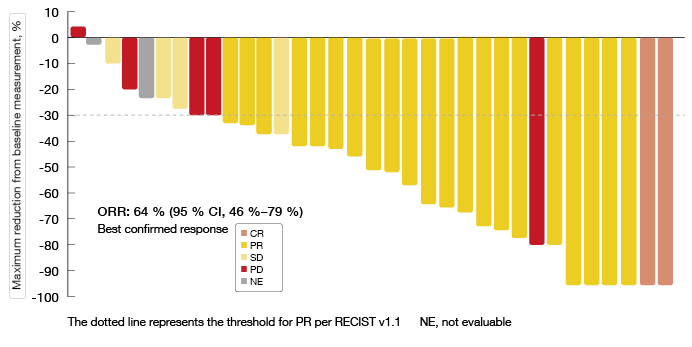
The dabrafenib plus trametinib regimen gave rise to substantial anti-tumour activity and durable responses. According to both investigator and IRC assessment, ORR was 64 %. Together with the proportion of patients demonstrating SD, this resulted in a DCR of 75 % and 72 % as per investigator and IRC review, respectively. Two patients experienced CR, and 21 achieved PR (Figure). Duration of response was 10.4 and 15.2 months according to investigator and IRC assessment, respectively. Median PFS was 10.9 and 14.6 months, respectively. At 6 months, 72 % and 69 % of patients, respectively, were progression-free. The combination gave rise to a preliminary median OS of 24.6 months. Fifty-one percent of patients were alive at 2 years. ORR, duration of response and PFS resembled those reported for the previously treated cohort receiving combination therapy in BRF113928 [5, 6].
The safety profile proved manageable and was similar to previous experience with the combination. AEs of all grades necessitated treatment discontinuation in 19 % and dose reductions in 31 %. Pyrexia, nausea and diarrhoea constituted the most common AEs. No new safety signals were observed. Based on these results, dabrafenib plus trametinib was recently approved by the EMA and the FDA for use in patients with metastatic NSCLC expressing the BRAFV600E mutation, regardless of prior treatment history.
Figure: Investigator-assessed maximum change in target lesions with dabrafenib plus trametinib combination treatment
Afatinib in heavily pre-treated patients with HER2 mutation
Approximately 1–4 % of adenocarcinomas of the lung harbour HER2 mutations [8], but approved targeted treatments are still lacking for these patients. Afatinib works by irreversibly inhibiting signalling from all ErbB family receptor homodimers and heterodimers, including HER2 [9, 10]. A global named patient use programme initiated in 2010 is providing real-world data on the use of afatinib in global clinical practice for NSCLC patients with no established therapeutic option. Peters et al. reported treatment outcomes for the cohort with HER2-mutant NSCLC [11].
The patients treated in the named patient use programme received afatinib 50 mg daily; lower starting doses were allowed at the discretion of the physician. As of April 2017, data were available for 28 patients. More than half of them had been treated with three or more systemic lines. First-generation EGFR TKI monotherapy had been administered in 36 %. Seven patients had already received HER2-directed drugs. All specified HER2 mutations were identified in exon 20, with the most common mutation type being a 2325/YVMA insertion (n = 8).
The analysis suggested clinically meaningful efficacy of afatinib. Median time to treatment failure (TTF) was 2.9 months; here, patients showing a 2325/YVMA insertion experienced markedly improved TTF (9.9 months) compared to those with other specified HER2 mutations (1.9 months). In the overall population, 32 % of patients had a TTF of > 1 year. Among 16 patients with available response data, ORR and DCR were 19 % and 69 %, respectively. This is in line with findings from another international, multi-centre study of afatinib in HER2-mutant NSCLC [12]. Patients with a 2325/YVMA insertion obtained disease control in 100 %. No unexpected AEs occurred. Based on these results, the evaluation of afatinib in earlier treatment lines in HER2-mutant NSCLC patients might be warranted.
References
- Barlesi F et al., Routine molecular profiling of patients with advanced non-small-cell lung cancer: results of a 1-year nationwide programme of the French Cooperative Thoracic Intergroup (IFCT). Lancet 2016; 387: 1415-1426
- Kris MG et al., Using multiplexed assays of oncogenic drivers in lung cancers to select targeted drugs. JAMA 2014; 311: 1998-2006
- Marchetti A et al., Clinical features and outcome of patients with non-small-cell lung cancer harboring BRAF mutations. J Clin Oncol 2011; 29: 3574-3579
- Cardarella S et al., Clinical, pathologic, and biologic features associated with BRAF mutations in non-small cell lung cancer. Clin Cancer Res 2013; 19: 4532-4540
- Planchard D et al., Dabrafenib in patients with BRAF(V600E)-positive advanced non-small-cell lung cancer: a single-arm, multicentre, open-label, phase 2 trial. Lancet Oncol 2016; 17: 642-650
- Planchard D et al., Updated survival of patients (pts) with previously treated BRAF V600E–mutant advanced non-small cell lung cancer (NSCLC) who received dabrafenib (D) or D + trametinib (T) in the phase II BRF113928 study. J Clin Oncol 2017; 35(suppl) abstract 9075
- Planchard D et al., Phase 2 trial (BRF113928) of dabrafenib plus trametinib in patients with previously untreated BRAFV600E-mutant metastatic non-small cell lung cancer. ESMO 2017, abstract LBA51
- Peters S & Zimmermann S, Targeted therapy in NSCLC driven by HER2 insertions. Transl Lung Cancer Res 2014; 3(2): 84-88
- Solca F et al., Target binding properties and cellular activity of afatinib (BIBW 2992), an irreversible ErbB family blocker. J Pharmacol Exp Ther 2012; 343(2): 342-350
- Li D et al., BIBW2992, an irreversible EGFR/HER2 inhibitor highly effective in preclinical lung cancer models. Oncogene 2008; 27(34): 4702-4711
- Peters S et al., Activity of afatinib in heavily pre-treated patients with HER2 mutation-positive advanced NSCLC: findings from a global NPU programme. ESMO 2017, abstract 1355P
- Lai W-C V et al., Afatinib in patients with metastatic HER2-mutant lung cancers: An international multicenter study. J Clin Oncol 2017; 35(suppl) abstr 9071