Anti-EGFR treatment: real-world experience and clinical trial insights
Factors determining treatment selection
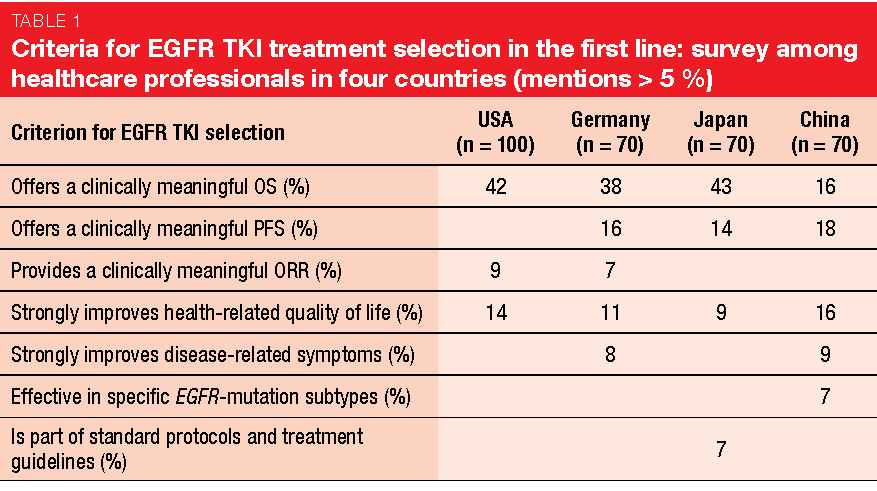
As the treatment landscape for EGFR-mutant stage IIIB/IV NSCLC has significantly changed over the past years, Hirsh et al. assessed current attitudes among physicians towards decision making for EGFR tyrosine kinase inhibitor (TKI) sequencing [1]. Between April and May 2018, the investigators conducted a representative online survey of 310 healthcare professionals including oncologists, pulmonologists, thoracic surgeons and internal respiratory specialists across the USA, Germany, Japan and China.
Irrespective of treatment line, the physicians’ most important treatment ambitions when prescribing TKIs included increasing OS, followed by improvement in quality of life. While clinically meaningful OS stood out for the US, Germany and Japan, offering a clinically meaningful PFS, OS and improved health-related quality of life appeared to be of equal importance to Chinese doctors (Table 1). Predictability of treatment outcome in the first-line setting was another important factor influencing treatment choices. In terms of sequencing of TKIs, 55 % of participants strongly preferred a treatment sequence offering the patients maximum time on targeted therapies.
Physicians across all countries expressed a strong need for information on potential resistance mutations before changing their current treatment practice. Thirty-six percent of all health care professionals agreed that they do not feel they have all the data required to make informed decisions on how to sequence EGFR-targeted NSCLC treatments.
Real-world data on afatinib and feasibility in elderly patients
The irreversible ErbB family inhibitor afatinib is approved in several countries worldwide for the treatment of patients with EGFR-mutant NSCLC. Real-world data presented at the WCLC 2018 corroborated the efficacy of afatinib in everyday clinical practice. A multicentre retrospective study of 128 patients conducted at five institutions in Japan revealed comparable or even better efficacy compared to previous clinical trials [2]. In the first-line and re-challenge settings, median PFS was 17.8 and 8.0 months, respectively. Median OS for first-line patients amounted to 39.5 months. Dose reductions did not diminish treatment efficacy, but even significantly prolonged PFS compared to patients without dose reductions (18.5 vs. 7.9 months; p = 0.018). Another retrospective observational study conducted in 22 patients showed a median PFS of 13.1 months, an ORR of 86.3 % and a disease control rate of 95.5 % [3]. At 12 months, 81.8 % of patients were alive.
An open-label, multicentre, single-arm phase II study identified first-line afatinib at a daily dose of 30 mg as a preferable treatment option in the elderly [4]. Substantial activity of this regimen was observed in 40 patients aged 70 years or older, with an ORR of 72.5 % and a 100 % disease control rate. Twenty-eight patients (70 %) achieved partial responses, and complete response occurred in one case (2.5 %). PFS and OS were 15.2 and 30.2 months, respectively. Most of the AEs observed in the study were rated as low-grade.
RealGiDo
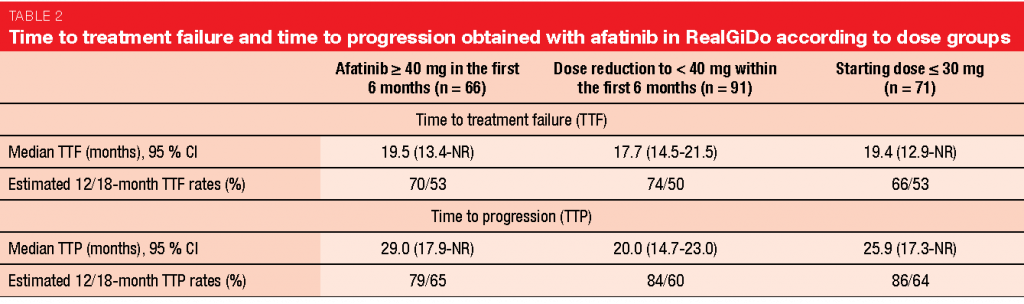
In the LUX-Lung clinical trials assessing afatinib in patients with EGFR-mutation–positive NSCLC, the incidence and severity of AEs was reduced by the use of tolerability-guided dose adjustments, which did not compromise efficacy [5, 6]. Real-world data from the non-interventional, observational RealGiDo study confirmed that outcomes in afatinib-treated NSCLC patients can be optimised by tailoring afatinib doses based on individual patient characteristics and adverse drug reactions (ADRs) [7]. Dose adjustments reduced the frequency and intensity of ARDs without impacting treatment efficacy. RealGiDo was conducted at 29 sites across 13 countries worldwide and included 228 patients. Compared to the pivotal LUX-Lung 3 trial [8], the cohort contained more patients with deletion 19 (78 % vs. 49 %), fewer Asian patients (44 % vs. 72 %) and a greater proportion of individuals with poor performance status, as 12 % had ECOG PS 2-3 compared to none in LUX-Lung 3.
Overall, 78 % of patients had a dose modification in the course of RealGiDo. Thirty-one percent received a starting dose of < 40 mg. In 20 % of these, dose increases were implemented during the study. ADRs constituted the main reason for dose modifications. Consistent with LUX-Lung 3, most dose reductions in patients who started on a dose of 40 mg occurred within the first 6 months of treatment. The rate of dose reductions was numerically higher in RealGiDo (67 % vs. 53 % in LUX-Lung 3).
Nevertheless, afatinib demonstrated consistent activity regardless of dose reductions or modification of starting doses. Time to treatment failure (TTF) was 18.7 months in the total population and did not differ significantly across the groups with and without dose modifications (Table 2). This also applied to time to progression, which amounted to 20.8 months in the total population (Table 2). In patients starting on doses ≤ 30 mg, the overall ADR incidence was similar to that in patients using starting doses ≥ 40 mg, with fewer grade-3 and no grade-4 ADRs.
Mechanisms of afatinib resistance
Approximately half of the patients progressing on treatment with first- or second-generation EGFR TKIs are expected to have EGFR T790M resistance mutations. For afatinib, data are lacking even though it is assumed that resistance mechanisms might differ from those found in first-generation EGFR TKIs due to the irreversible and pan-HER nature of inhibition.
Nakamura et al. analysed 20 patients with acquired afatinib resistance, including resistance during EGFR TKI re-challenge [9]. As with first-generation EGFR TKIs, T790M mutation was associated with acquired resistance to afatinib, albeit with somewhat lower frequency. Among the patients who were T790M-negative before the start of afatinib treatment, 40 % became positive at the time of progression. C797S emerged in 3 patients, although with very low allele frequency. While BRAF V600E mutation was detected in 1 patient, no MET amplification occurred.
In their prospective assessment of 25 previously afatinib-treated patients, Iwama et al. identified T790M as a putative mechanism of resistance in 44 % [10]. The cause was unknown in another 40 %. Apart from these groups, only 1 patient each showed other mutations or combined aberrations that have also been reported in the setting of other-generation EGFR TKIs (i.e., MET copy number gains [CNGs], NRAS CNGs, T790M plus EGFR CNGs, T790M plus PIK3CA CNGs plus PIK3CA E545K).
ASTRIS: osimertinib in everyday clinical practice
Wu et al. reported results from the second interim analysis of the ongoing ASTRIS trial, which is the largest international, real-world treatment study investigating the third-generation EGFR TKI osimertinib in EGFR T790M-positive, locally advanced or metastatic NSCLC [11]. Before receiving osimertinib 80 mg daily, the patients had been treated with at least one prior EGFR TKI. Asymptomatic stable CNS metastases were allowed. The patients were identified using a wide range of clinically employed molecular tests and specimens.
According to the findings, the clinical activity and safety of osimertinib as assessed in this real-world population were in line with the results observed in the AURA clinical trial programme. In the overall population of the ASTRIS trial, the response rate was 56.1 %, and median PFS amounted to 11.0 months. Time to treatment discontinuation was 12.6 months. OS data were immature at the time of the analysis, with an 18-month OS rate of 63.4 %.
Subgroup analyses showed consistent efficacy in patients aged ≥ 75 years and < 75 years; here, PFS was 11.8 and 10.9 months, respectively. Moreover, osimertinib provided clinical benefit in patients with poor performance status (PS 2), although this group experienced shorter PFS than the population with PS 0 or 1 (6.9 vs. 11.1 months for PS 2 and PS 0/1, respectively), which is not surprising. Also, the data corroborated the CNS activity of osimertinib. Median PFS results for patients with and without brain metastases were 9.7 and 11.9 months, respectively.
Uncommon mutations: activity of osimertinib
Approximately 10 % of EGFR mutants harbour uncommon mutations, which represent a heterogeneous group of rare molecular alterations within exons 18 to 21. There has been a paucity of data regarding the sensitivity of these tumours to EGFR TKI therapies [12].
According to a phase II, open-label, single-arm, multicentre study, osimertinib is active in NSCLC with uncommon mutations [13]. Thirty-five EGFR-TKI–naïve patients with stage IV NSCLC and activating EGFR mutations other than exon 19 deletion, L858R mutation, T790M mutation, and exon 20 insertion were included. G719X mutation was most prevalent in this population (54 %), followed by L861Q (26 %) and S768I (23 %). When treated with osimertinib 80 mg daily, 51 % of patients experienced objective responses, and disease control occurred in 89 %. Median PFS and duration of response were 8.2 and 9.8 months, respectively.
A separate analysis according to the most frequent uncommon mutations revealed the highest response rate in the presence of the L861Q mutation (77.8 %) compared to G719X and S768I (57.9 % and 37.5 %, respectively). Activity was also observed in some patients with CNS metastases. Nine patients had brain lesions at baseline. After initiation of osimertinib treatment, one of these achieved complete intracranial response, and two had partial responses. Osimertinib demonstrated a manageable safety profile consistent with previous reports.
Mutation-independent effects of afatinib
Likewise, results obtained from a global named-patient-use programme underscore the efficacy of afatinib in patients with advanced NSCLC harbouring uncommon mutations [14]. Almost all of 2,242 patients included in 10 Asian countries had received previous lines of treatment (median, 3) that involved erlotinib or gefitinib. Ninety-seven percent of patients with known tumour mutation status were EGFR-positive. Among those with specified mutations, 93.9 % had common mutations, while 10.6 % had uncommon mutations (e.g., exon 20 insertion, G719X, L861Q, S768I, T790M). In some cases, multiple mutations were observed. Twelve patients had HER2 mutations, but no EGFR mutations.
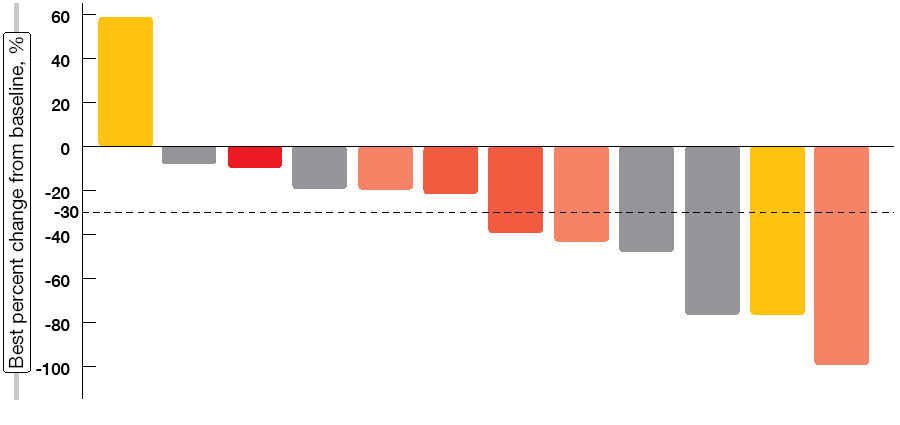
Overall, 24.4 % of patients responded to the afatinib treatment, with 77.7 % achieving disease control. Activity of afatinib was evident in patients with both common and uncommon EGFR mutations (Figure 1). Response rates for these two groups were 27.4 % and 28.1 %, respectively. When analysed according to the type of uncommon mutation, ORRs amounted to 42.9 % in patients with G719X, L861Q and S768I mutations, and to 20 % in those with EGFR exon 20 insertion. Patients with HER2 mutation responded in 14.2 %.
Median TTF was 7.6 months in the total cohort and 7.2 months in the EGFR-positive group. Again, patients with uncommon mutations fared at least as well as those with common mutations; TTF was 8.4 and 6.4 months for these two groups, respectively. Patients with exon 20 insertions even showed a median TTF of 18.9 months. In the HER2-positive patient population, this was 12.2 months.
Figure 1: Afatinib in common and uncommon mutations: response rates (%)
Poziotinib in exon 20-positive patients
The pan-epidermal growth factor receptor inhibitor poziotinib was designed as a potent and selective inhibitor of EGFR and HER2 exon 20 mutations. Targeting this rare, difficult-to-treat mutation became possible because the size and shape of pozitinib enable the molecule to fit into the sterically hindered exon 20 binding pocket.
An open-label, phase II trial presented at the WCLC 2018 demonstrated pronounced anti-tumour activity for poziotinib in metastatic, heavily pre-treated EGFR exon 20-mutant NSCLC [15]. The ORR achieved in 50 patients was 55 %, with 43 % having been confirmed to date. This compares favourably to historical ORR rates obtained with approved EGFR TKIs and standard-of-care second-line agents including docetaxel and immune checkpoint inhibitors. Durable responses were observed. At the time of the analysis, 6 patients had been receiving treatment for > 1 year. Median PFS was 5.5 months.
Moreover, the trial contained 13 patients with HER2 exon 20-mutant NSCLC who also derived significant benefits. Initial responses occurred in 50 % of evaluable patients (n = 12), and median PFS was 5.1 months. EGFR-related toxicity proved manageable. Dose reductions became necessary in 60 %, but discontinuations due to AEs were rare at 3 %. A confirmatory, international, multicentre study evaluating poziotinib in EGFR and HER2 exon 20-mutant NSCLC patients is currently enrolling (NCT03318939).
Rationale for combinations with immunotherapy
Experimental data support the combination of EGFR-targeted therapy with immune checkpoint inhibitors [16]. EGFR TKI treatment was found to alter the tumour microenvironment by way of indirect effects on immune cells. A phase I study examined the immunomodulatory effects of combining afatinib 40 mg daily with pembrolizumab 200 mg Q3W in 11 patients with advanced, EGFR-mutant NSCLC that had progressed on front-line EGFR TKI therapy [17]. The combination showed modest activity with an ORR of 18 % and a median PFS > 6 months in 4 patients. It induced dynamic changes in the immune microenvironment in patients who showed clinical benefit, such as increases in CD3-positive T cell counts and decreases in tumour Ki-67.
All patients with partial responses and/or PFS > 6 months experienced immune-related AEs (irAEs) that necessitated eventual discontinuation of pembrolizumab. Patients who derived clinical benefit but developed irAEs had a distinct circulating and tumour immune cell profile including increased CD4/CD8 T cell ratio and increased soluble B- and T-lymphocyte attenuator. Larger data sets are needed to further support these findings.
Promising activity of third-generation TKI lazertinib
The potent, highly mutant-selective and irreversible third-generation EGFR TKI lazertinib targets both the T790M mutation and activating EGFR mutations while sparing wild-type EGFR. Lazertinib is able to penetrate the blood-brain barrier. In an open-label, multicentre, phase I/II study, patients with locally advanced or metastatic NSCLC and acquired resistance to prior EGFR TKI treatment received lazertinib once daily. Cho et al. presented the findings obtained in the dose escalation and dose expansion parts of the trial; these included a total of 127 patients [18].
In the dose escalation part, no dose-limiting toxicity was observed up to a dose of 320 mg. The most common AEs comprised pruritus, rash, constipation, and decreased appetite. Across the entire dose range, the confirmed ORR amounted to 61 %, and disease control resulted in 89 %. For patients with T790M mutation, these were 66 % and 93 %, respectively. Lazertinib also showed activity in patients with measurable brain metastases, with an intracranial ORR of 50 % (Figure 2). Responses lasted for extended periods, with the longest duration of response exceeding 12.5 months. At the time of the data cut-off, median PFS had not yet been calculated.
As the authors noted, lazertinib demonstrated promising anti-tumour activity in patients with acquired resistance to prior EGFR TKI treatment. Based on the risk-benefit profile and pharmacokinetics, 240 mg is the recommended phase II dose. A global phase III clinical trial investigating first-line lazertinib will commence in 2019.
Figure 2: Intracranial responses to lazertinib in patients with measureable brain metastases
REFERENCES
- Hirsh V et al., Oncologist treatment considerations and selection in EGFR M+ NSCLC. WCLC 2018, P3.01-108
- Tanaka H et al., Real world study of afatinib in first-line or re-challenge setting for patients with EGFR mutant non-small cell lung cancer. WCLC 2018, P3.13-22
- Liam CK et al., Real-world experience of first-line afatinib treatment in patients with EGFR mutant advanced non-small cell lung cancer. WCLC 2018, P3.15-21
- Suzuki K et al., A phase II study of afatinib treatment for elderly patients with previously untreated advanced non-small cell lung cancer harboring EGFR mutations. WCLC 2018, P1.01-92
- Yang JC et al., Effect of dose adjustment on the safety and efficacy of afatinib for EGFR mutation-positive lung adenocarcinoma: post hoc analyses of the randomized LUX-Lung 3 and 6 trials. Ann Oncol 2016; 27(11): 2103-2110
- Hirsh V et al., First-line afatinib (A) vs gefitinib (G) for patients (pts) with EGFR mutation positive (EGFRm+) NSCLC (LUX-Lung 7): Patient-reported outcomes (PROs) and impact of dose modifications on efficacy and adverse events (AEs). J Clin Oncol 2016; 34 (15_suppl): 9046-9046
- Halmos B et al., Impact of afatinib dosing on safety and effectiveness in real-world patients with EGFR mutation-positive advanced NSCLC (RealGiDo). WCLC 2018, P1.01-28
- Sequist LV et al., Phase III study of afatinib or cisplatin plus pemetrexed in patients with metastatic lung adenocarcinoma with EGFR mutations. J Clin Oncol 2013; 31(27): 3327-3334
- Nakamura T et al., Mechanisms of acquired resistance to afatinib clarified with liquid biopsy. WCLC 2018, P3.13-18
- Iwama E et al., A multicenter prospective biomarker study to explore mechanisms of afatinib resistance based on digital PCR and next generation sequencing. WCLC 2018, P2.13-18
- Wu YL et al., ASTRIS: A real world treatment study of osimertinib in patients with EGFR T790M mutation-positive NSCLC. WCLC 2018, MA02.03
- Beau-Faller M et al., Rare EGFR exon 18 and exon 20 mutations in non-small-cell lung cancer on 10 117 patients: a multicentre observational study by the French ERMETIC-IFCT network. Ann Oncol 2014; 25(1): 126-131
- Cho JH et al., An open-label, multicenter, phase II single arm trial of osimertinib in non-small-cell lung cancer patients with uncommon EGFR mutation (KCSG-LU15-09). WCLC 2018, OA10.05
- Chang GC et al., Named patient use program for afatinib in advanced NSCLC with progression on prior therapy: experience from Asian centers. WCLC 2018, P1.01-11
- Heymach JV et al., Phase II trial of poziotinib for EGFR and HER2 exon 20 mutant NSCLC. WCLC 2018, OA02.06
- Jia Y et al., EGFR-targeted therapy alters the tumor microenvironment in EGFR-driven lung tumors: rationale for combination therapies. WCLC 2018, MA27.10
- Riess JW et al., Immunomodulatory effects of afatinib and pembrolizumab in EGFR-mutant NSCLC with progression on prior EGFR TKI. WCLC 2018, P1.04-09
- Cho BC et al., Lazertinib, a third generation EGFR TKI, in patients with EGFR-TKI-resistant NSCLC: updated results of a phase I/II study. WCLC 2018, MA26.09