EGFR-targeted therapy: at the right time in the right patient
Approximately 11 % of Caucasian patients with NSCLC have tumours that harbour EGFR mutations [1], which occur in exons 18, 19, 20 and 21 of the EGFR gene. Common mutations include exon 19 in-frame deletions and the exon 21 Leu858Arg point mutation (L858R) [2]. Exon 20 insertions are known to mediate resistance [3]. Little data are available for the other more uncommon mutations.
The activating EGFR mutations sensitise lung tumours to EGFR tyrosine kinase inhibitor (TKI) therapies. The irreversible ErbB family blocker afatinib and the reversible EGFR TKIs gefitinib and erlotinib have been approved for first-line therapy of patients with advanced, EGFR-mutation positive NSCLC. The phase IIb LUX-Lung 7 study is the first prospective, global, randomised trial to compare two EGFR-directed therapies (afatinib, gefitinib) in a head-to-head manner in this setting. A total of 319 patients with EGFR-mutated, stage IIIB/IV adenocarcinoma of the lung who had not received any prior treatments for advanced or metastatic disease were randomised to either afatinib 40 mg/day or gefitinib 250 mg/ day. According to the primary analysis, when compared to gefitinib, afatinib significantly improved the co-primary endpoints of PFS (hazard ratio [HR], 0.73; p = 0.017) and time to treatment failure (TTF; HR, 0.73; p = 0.007) [4]. Correspondingly, ORR was significantly superior in the afatinib arm (p = 0.008).
LUX-Lung 7: OS analysis and other updated outcomes
At the ESMO Congress, Paz-Ares et al. presented the primary OS analysis of LUX-Lung 7, which yielded a difference of 3 months between these two TKIs in favour of afatinib (27.9 vs. 24.5 months), although this difference did not reach statistical significance (HR, 0.86; p = 0.2580) [5]. Most of the prespecified subgroups derived greater OS benefit from afatinib than gefitinib. The median OS for afatinib was similar across the age subgroups. Also, the OS analyses by mutation subtype favoured afatinib in patients with both deletion 19 and the L858R mutation, although without reaching statistical significance. Forty-six percent in the afatinib arm and 56 % in the gefitinib arm received subsequent EGFR TKI therapy. In those treated with a subsequent thirdgeneration EGFR TKI, median OS was
similar for afatinib and gefitinib.
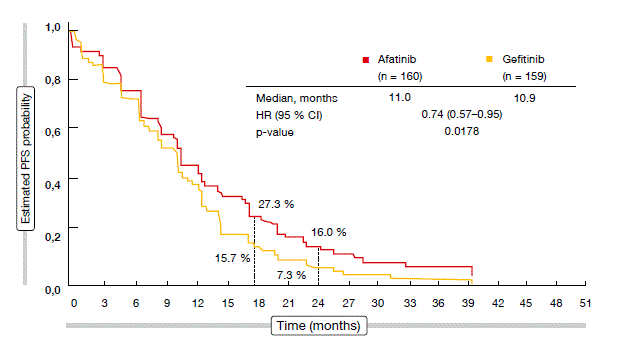
The updated results on PFS and ORR were consistent with the initial data. Relative to gefitinib, afatinib significantly improved PFS (11.0 vs. 10.9 months; HR, 0.74; p = 0.0178; Figure) and ORR (73 % vs. 56 %; p = 0.002). At 24 months, PFS rates were 16.0 % versus 7.3 %. In patients with the L858R mutation, ORR significantly favoured afatinib (69 % vs. 42 %; p = 0.003), while in those with deletion 19, afatinib gave rise to a numerical ORR advantage (75 % vs. 66 %; p = 0.150). AEs were predictable and manageable, with both TKIs showing equally low rates of treatment discontinuation. Updated quality-of-life data remained similar between the arms. The investigators concluded that the overall data, which were largely positive across multiple clinically relevant endpoints, suggest that afatinib is a more effective treatment option than gefitinib in the first-line setting.
Figure: Progression-free survival according to independent data review with afatinib versus gefitinib in LUX-Lung 7
TTF in LUX-Lung 7
Schuler et al. reported results for the coprimary endpoint of time to treatment failure (TTF), which was chosen to reflect real-world clinical practice and treatment guidelines [6]. TTF was defined as the time from randomisation to the time of treatment discontinuation for any reason, including disease progression, treatment toxicity, and death. Patients could remain on treatment beyond progression if deemed beneficial by the physician. Thirty-five percent of patients in the afatinib arm and 29.6 % of those in the gefitinib arm who obtained clinical benefit continued their TKI treatment beyond radiological progression, for median durations of 2.7 and 2.0 months, respectively.
In the overall LUX-Lung 7 population, afatinib provided superior TTF compared to gefitinib (13.7 vs. 11.5 months; HR, 0.73; p = 0.0073). These TTF benefits were generally consistent across the prespecified subgroups (i.e., type of EGFR mutation, presence of brain metastases, baseline ECOG performance status, gender, age, ethnicity, smoking history). Significantly greater percentages of patients in the afatinib arm were free of treatment failure at 24 months (25 % vs. 13 %) and at 30 months (15 % vs. 5 %).”
These results are complementary to the PFS and ORR findings in LUX-Lung 7. According to the authors, improved TTF with afatinib testifies to its general tolerability and the manageability of the associated AEs, and suggests that this drug can confer additional clinical benefit in patients who continue treatment beyond radiological disease progression.
VeriStrat® stratification of afatinib-treated patients
The phase III, global, open-label, LUXLung 8 study compared afatinib with erlotinib in patients with stage IIIB/IV squamous-cell NSCLC who had progressed after first-line platinum-doublet chemotherapy. In this trial, afatinib significantly improved OS, PFS and disease control rate (DCR) compared to erlotinib [7].
Goss et al. evaluated the predictive value of the VeriStrat® serum protein test in LUX-Lung 8, using OS as the primary efficacy variable [8]. VeriStrat® can be used to identify patient responses to their tumour by measuring several acute-phase reactant proteins in the blood. This test categorises patients according to a distinct classification algorithm that distinguishes between ‘good’ (VS-G) and ‘poor’ (VS-P). Clinical outcomes were analysed with respect to the patient VeriStrat® status in the overall population and in the pre-defined subgroups. Results were obtained for 675 patient samples. Of these, 412 and 263 fell into the VS-G and VS-P categories, respectively.
The VeriStrat® test was shown to have a strong independent stratification effect with these afatinib-treated patients. In the VS-G group, as compared to erlotinib, afatinib gave rise to significant improvements in OS (11.5 vs. 8.9 months; HR, 0.79) and PFS (3.3 vs. 2.0 months; HR, 0.73). In the VS-P group, on the other hand, the OS and PFS did not differ significantly between these two TKIs. The patients who received afatinib experienced significant OS and PFS benefits based on the VS-G group versus the VS-P group (p < 0.0001 for each). Multivariate analysis showed that VeriStrat® was an independent predictor of OS and PFS in these afatinib-treated patients, regardless of ECOG performance status, best response to first-line therapy, ethnicity, and age. However, there was no significant interaction between the VeriStrat ® classification and treatment group for OS or PFS.
Real-world evidence for afatinib in later lines
The named patient use (NPU) programme, which was initiated in May
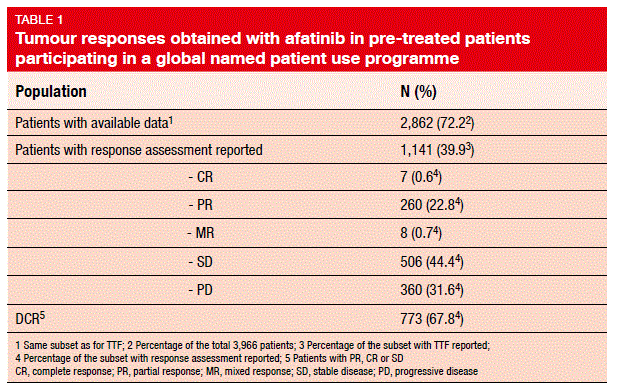
2010, provides real-world evidence of afatinib use in global clinical practice [9]. Eligibility criteria included advanced or metastatic NSCLC, progression after clinical benefit on erlotinib/gefitinib and/or presence of an activating EGFR/HER2 mutation, exhaustion of all other treatment options, and ineligibility for actively recruiting afatinib trials. As of January 2016, data were available for 3,966 NSCLC patients from 41 countries across six continents. The patients were heavily pre-treated. Approximately 50 % received afatinib as their fourth or later lines.
Median TTF was 4.4 months for all of these patients for whom the data were available. Similar TTF findings were seen for patients with any EGFR mutation, common or uncommon EGFR mutations, and HER2 mutations. The ORR was 23.4 % for all of the patients, with a DCR of 67.8 % (Table 1). Of note, the patients with any EGFR mutation and those with common and uncommon EGFR mutations showed similar ORRs. Response rates of 19 % and 35 % were seen for the patients with NSCLC harbouring the T790M and exon 20 insertion mutations, respectively. No new or unexpected safety findings were observed in the NPU programme.
Effects in patients with leptomeningeal disease
The central nervous system (CNS) is a common site of recurrence in patients with NSCLC, probably owing to the low penetration of agents into the CNS. Cerebrospinal fluid (CSF) concentration rates of the EGFR TKIs gefitinib and erlotinib were found to be low [10]. However, the combined analysis of patients with brain metastases in the LUX-Lung 3 and 6 trials suggests that afatinib works in the brain [11].
As Tamiya et al. noted in their prospective multi-centre trial including 11 patients with EGFR-positive NSCLC and leptomeningeal carcinomatosis [12], treatment with afatinib at the recommended daily dose of 40 mg shows a higher median cerebrospinal fluid (CSF) penetration rate than previously reported. The median blood and CSF levels were 88.2 ng/mL and 1.4 ng/mL, respectively, and the median CSF penetration rate was 1.65 %. Overall, median PFS and OS were 2.0 and 3.8 months, respectively. Particularly patients harbouring uncommon EGFR mutations appeared to benefit from afatinib with regard to clinical outcomes.
IMPRESS: gefitinib continuation has detrimental effects
After progression on EGFR TKI therapy, the continuation of this treatment in combination with platinum-based doublet chemotherapy was suggested to be beneficial because of potential tumour heterogeneity at the time of resistance.
This prompted the design of the IMPRESS trial. IMPRESS was the first randomised, phase III, multi-national study that investigated continued application of gefitinib plus chemotherapy versus chemotherapy alone in patients with EGFR-mutated advanced NSCLC who had acquired resistance to first-line gefitinib treatment after initial response. The primary analysis had already shown no statistically significant differences between the two strategies for PFS, ORR and DCR [13].
In agreement with the preliminary OS analysis, the final OS data demonstrated significant inferiority of the gefitinib combination [14]. Median OS was 13.4 months in the gefitinib arm and 19.5 months in the control arm, which translated into an increase in mortality risk of almost 50 % with the addition of gefitinib (HR, 1.44; p = 0.016). Furthermore, all of the subgroup categories favoured the chemotherapy-only treatment.
A larger proportion of the patients in the chemotherapy-only arm received EGFR TKI therapy after discontinuation, which might have contributed to their longer survival.
Exploratory plasma biomarker analyses suggested that this OS decrease associated with gefitinib continuation is driven by T790M-positive status. In the T790M-positive subgroup, median OS was 10.8 and 14.1 months with gefitinib and chemotherapy only, respectively (HR, 1.49). Results for the T790M-negative subgroup were inconclusive, however. Caution must be exercised here, because T790M status according to circulating tumour DNA is not always informative. The authors concluded that this first-generation EGFR TKI therapy needs to be stopped at the time of radiological disease progression, due to the risk of survival deterioration seen here for treatment continuation. Patients with T790M-positive status should be considered for third-generation EGFR TKI treatment.
Activity of gefitinib and erlotinib in uncommon mutations
A meta-analysis investigated the efficacy of the first-generation EGFR-TKIs gefitinib and erlotinib in patients with uncommon EGFR mutations (S768I, L861Q, G719X, R705K, and others) [15]. Out of 6,404 patients from the 13 trials included, 466 (7.3 %) were diagnosed as having uncommon EGFR mutations. These patients had received gefitinib and erlotinib as any line of treatment.
In single-arm synthesis, the overall ORRs for uncommon and common mutations were 34 % and 71 %, respectively. Direct comparisons indicated significantly lower responses in patients with uncommon mutations (odds ratio, 0.30). Also, they showed an inferior 6-month PFS rate (odds ratio, 0.44; p < 0.001). ORRs were still considerable, however, particularly in complex mutations, i.e., mutations at two or more uncommon mutant sites (64.2 %). The authors therefore stated that first-generation EGFRTKIs remain an option for the treatment of patients with uncommon mutations, but the decision-making needs to be cautious. The specific efficacies related to each mutation site merit future studies with larger sample sizes.
EGFR mutations: characteristics in a large French cohort
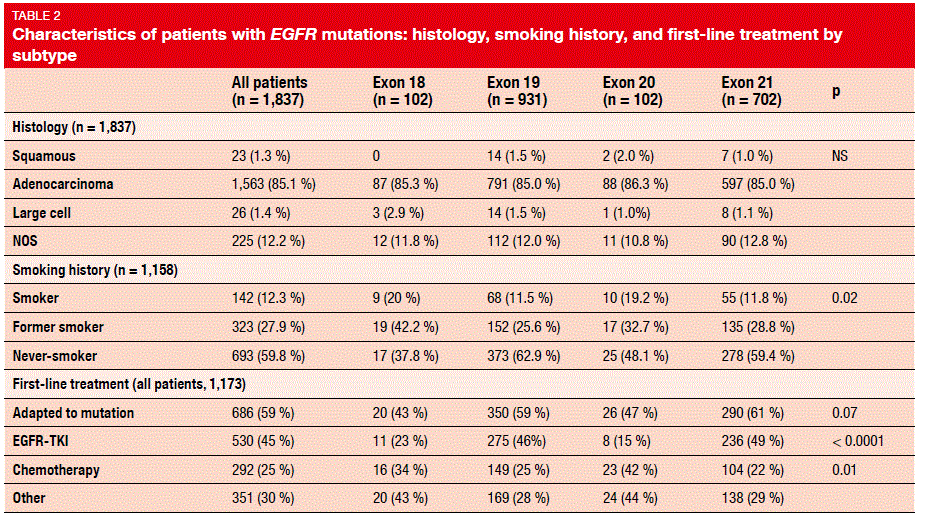
As different molecular properties of EGFR mutation subtypes might affect responses to EGFR TKIs and patient outcomes, an observational ancillary study of the French nationwide programme “Biomarkers France” assessed the characteristics of non-small cell lung tumours harbouring EGFR mutations on the basis of 18,679 analyses that represented 17,664 patients [16]. After exclusion of EGFR wild-type and the T790M mutation, a total of 1,837 patients with EGFR mutations were analysed.
Fifty-two percent and 38 % had exon 19 and exon 21 mutations, respectively. Exon 18 and exon 20 mutations were found in 5 % each. Two thirds of the patients were female, and 85.1 % had adenocarcinoma (Table 2). Sixty percent of those with exon 19 mutations and exon 21 mutations were never-smokers, while these proportions were smaller in the populations with uncommon mutations. Patients with exon 19 mutations and exon 21 mutations were more likely to receive first-line EGFR TKI therapy than the other two groups.
The investigators concluded that EGFR mutations should be screened regardless of smoking status. Precision of the specific sequence of the mutation at diagnosis is crucial for the selection of the appropriate treatment. In the subgroup with uncommon mutations, results differed considerably: while patients with exon 18 mutations derived benefit from first-line EGFR TKIs (median PFS, 7.8 months; DCR, 80 %), this did not apply to those whose tumours harboured exon 20 mutations, which were equivalent to EGFR wild-type in this respect (median PFS, 2.7 months; DCR, 20 %). For exon 21 mutations, OS and PFS were longer with EGFR-TKI therapy for L858R compared to the other mutations.
PFS and OS obtained with TKI treatment for patients with exon 19 mutations were superior to all of the other mutations, and the median OS was significantly longer even when compared to patients with exon 21 mutations (p = 0.045). EGFR-TKIs should therefore be a part of the treatment plan, although not necessarily the first-line strategy.
REFERENCES
- Barlesi F et al., Routine molecular profiling of patients with advanced non-small-cell lung cancer: results of a 1-year nationwide programme of the French Cooperative Thoracic Intergroup (IFCT). Lancet 2016; 387(10026): 1415-1426
- Riely GJ et al., Update on epidermal growth factor receptor mutations in non-small cell lung cancer. Clin Cancer Res 2006; 12(24): 7232- 7241
- Beau-Faller M et al., Rare EGFR exon 18 and exon 20 mutations in non-small-cell lung cancer on 10 117 patients: a multicentre observational study by the French ERMETIC-IFCT network. Ann Oncol 2014; 25(1): 126-131
- Park K et al., Afatinib versus gefitinib as firstline treatment of patients with EGFR mutationpositive non-small-cell lung cancer (LUX-Lung 7): a phase 2B, open-label, randomised controlled trial. Lancet Oncol 2016; 17: 577-589
- Paz-Ares L et al., Afatinib versus gefitinib in patients with EGFR mutation-positive NSCLC: overall survival data from the phase IIb trial LUXLUNG 7. ESMO 2016, LBA43
- Schuler M et al., Time-to-treatment failure with first-line afatinib versus gefitinib in patients with EGFR mutation-positive advanced NSCLC: randomised phase IIb LUX-Lung 7 trial. ESMO 2016, abstract 1230P
- Soria JC et al., Afatinib versus erlotinib as second-line treatment of patients with advanced squamous cell carcinoma of the lung (LUX-Lung 8): an open-label randomised controlled phase 3 trial. Lancet Oncol 2015; 16: 897-907
- Goss GD et al., Evaluation of VeriStrat®, a serum proteomic test, in the randomised, open-label, phase III LUX-Lung 8 trial of afatinib versus erlotinib for the second-line treatment of advanced squamous cell carcinoma of the lung. ESMO 2016, abstract 1238P
- Cappuzzo D et al., Global named patient use programme of afatinib, an oral ErB family blocker, in heavily pretreated advanced NSCLC patients who progressed following prior therapies, including erlotinib or gefitinib. ESMO 2016, abstract 1236P
- Soria JC et al., Gefitinib plus chemotherapy versus placebo plus chemotherapy in EGFRmutation- positive non-small-cell lung cancer after progression on first-line gefitinib (IMPRESS): a phase 3 randomised trial. Lancet Oncol 2015; 16(8): 990-998
- Soria JC et al., Gefitinib/chemotherapy versus chemotherapy in EGFR mutation-positive NSCLC after progression on first-line gefitinib (IMPRESS study): final overall survival analysis. ESMO 2016, abstract 1201O
- Zhang Y et al., Efficacy of first-generation EGFR-TKIs in patients with NSCLC harbouring EGFR uncommon mutations: a pooled analysis. ESMO 2016, abstract 1231P
- Leduc C et al., Clinical and molecular characteristics of non-small cell lung cancer (NSCLC) harbouring EGFR mutations. ESMO 2016, abstract 1202O
More posts
SCLC: genomic alterations pave the way to targeted approaches
Rapid growth and early development of metastatic disease are characteristic of small-cell lung cancer (SCLC), which constitutes approximately 15 % of all lung cancer cases. In limited-stage disease, a cure is possible with chemoradiotherapy. However, 68 % of patients present with extensive-stage SCLC (ES-SCLC). Although high initial responses to platinum-based chemotherapy and radiotherapy are observed, recurrence of chemo-refractory disease takes place as a rule.
Rare driver mutations: encouraging results in small patient populations
As well as ALK fusion mutations and EGFR mutations, studies of the genetic profiles of patients with NSCLC have identified other mutations that might be used for additional targeted therapies. Among these, ROS1 and RET rearrangements both occur in 1 % to 2 % of patients with NSCLC. Crizotinib is known to target not only ALK, but also ROS1, among others. Patients with ROS1-positive advanced NSCLC are being treated with crizotinib 250 mg twice daily in the ongoing phase I, open-label, PROFILE 1001 study.
EGFR-targeted therapy: at the right time in the right patient
Approximately 11 % of Caucasian patients with NSCLC have tumours that harbour EGFR mutations, which occur in exons 18, 19, 20 and 21 of the EGFR gene. Common mutations include exon 19 in-frame deletions and the exon 21 Leu858Arg point mutation (L858R). Exon 20 insertions are known to mediate resistance [3]. Little data are available for the other more uncommon mutations.
Targeting angiogenesis can prolong life
How important is anti-angiogenesis in the treatment concept of lung cancer?
Next-generation ALK inhibitors excel after crizotinib failure
ALK fusion-gene–positive lung cancer occurs in approximately 5 % of patients with advanced NSCLC [1]. The ALK inhibitor crizotinib demonstrates significant initial efficacy in patients with ALK-positive advanced NSCLC. However, most patients eventually develop resistance, with the central nervous system (CNS) being one of the most common sites of first progression.
Immune checkpoint inhibition: the picture is slowly completing itself
The anti–PD-1 antibody pembrolizumab has been approved for treatment of patients with PD-L1–expressing, advanced NSCLC. The KEYNOTE-024 study focused on the first-line comparison of pembrolizumab with platinum-doublet chemotherapy. Chemotherapy regimens comprised five options, two of which (pemetrexed plus carboplatin; pemetrexed plus cisplatin) were used with non-squamous non-small–cell lung cancer (NSCLC) only.