J-AXEL: nab-paclitaxel at least equal to docetaxel in pretreated NSCLC
Various advantages have been described for nab-paclitaxel, the albumin-bound, solvent-free, nanoparticle formulation of paclitaxel [1-3]. Phase II data showed favorable results in patients with pretreated advanced NSCLC who obtained an ORR of 32 % and median PFS of 5 months [4]. Therefore, the randomized, phase III study reported by Nakamura et al. compared nab-paclitaxel 100 mg/m2 on days 1, 8 and 15 three-weekly with docetaxel 60 mg/m2 every three weeks in patients with stage IIIB/IV or recurrent NSCLC previously treated with cytotoxic chemotherapy [5]. The analysis aimed to demonstrate non-inferiority of nab-paclitaxel with respect to OS. Both arms included approximately 250 patients.
Significant PFS and ORR benefits
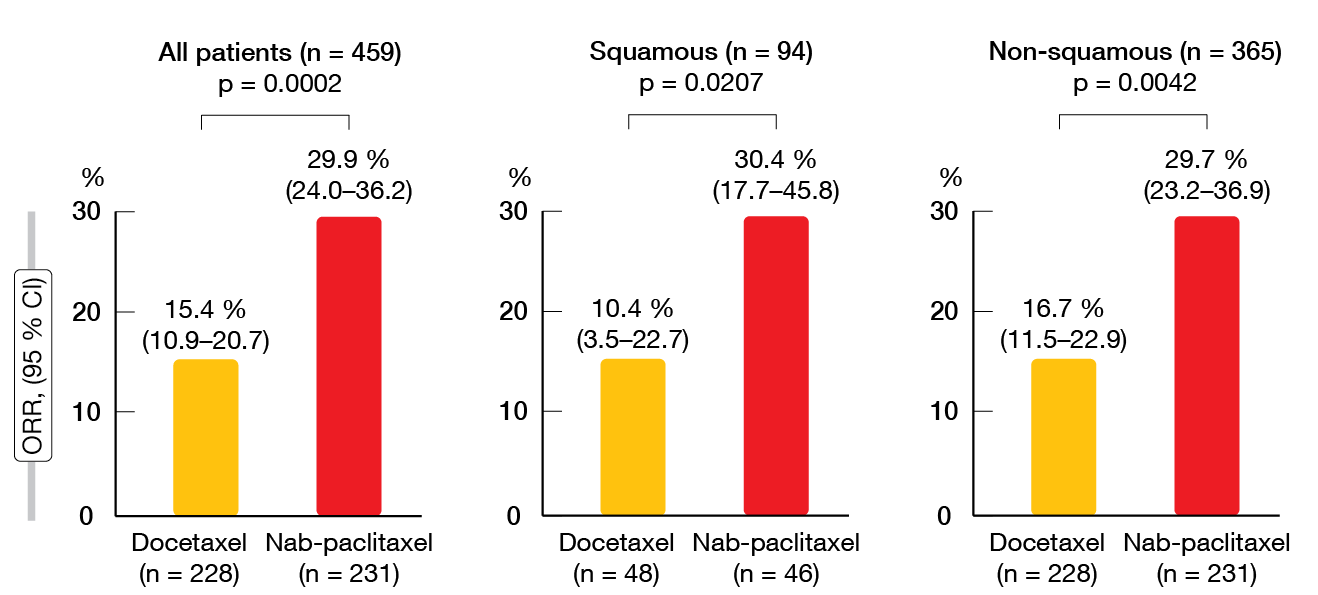
Non-inferiority of nab-paclitaxel in terms of OS was confirmed with the protocol-specified margin of 1.25 in the intent-to-treat population (HR, 0.85; 95.2 % CI, 0.68–1.070). Median OS amounted to 16.2 and 13.6 months with nab-paclitaxel and docetaxel, respectively. As specified by the protocol, superiority of nab-paclitaxel over docetaxel for OS was tested after non-inferiority had been shown. However, nab-paclitaxel did not significantly improve survival, although this was the case for both PFS and ORR. Median PFS was 4.2 vs. 3.4 months with nab-paclitaxel and docetaxel (HR, 0.76; p = 0.0042). In the total group (n = 459), 29.9 % vs. 15.4 % of patients responded to treatment (p = 0.0002; Figure). For the patients with squamous histology (n = 94), this was 30.4 % vs. 10.4 % (p = 0.0207), and for those with non-squamous NSCLC (n = 365), 29.7 % vs. 16.7 % (p = 0.0042). The results for both PFS and OS favored nab-paclitaxel across various subgroups pertaining to age, sex, ECOG performance status, histology, smoking status, disease stage, EGFR mutation status, and pretreatment.
Hematologic toxicity with docetaxel and neuropathy with nab-paclitaxel
Among AEs, leukopenia and neutropenia occurred significantly more often with docetaxel than with nab-paclitaxel (p < 0.0001 for both comparisons); correspondingly, docetaxel conferred a significantly higher incidence of febrile neutropenia (22.1 % vs 2.0 %). On the other hand, peripheral sensory neuropathy was more frequent with nab-paclitaxel (55.5 % vs. 20.1 %, p < 0.0001). The authors stressed in their summary that nab-paclitaxel should be considered a standard option for previously treated patients with advanced NSCLC.
Figure: Objective responses rates achieved with nab-paclitaxel vs. docetaxel in the ITT population and according to histology
REFERENCES
- Desai N et al., Increased antitumor activity, intratumor paclitaxel concentrations, and endothelial cell transport of cremophor-free, albumin-bound paclitaxel, ABI-007, compared with cremophor-based paclitaxel. Clin Cancer Res 2006; 12(4): 1317-1324
- Sasaki Y et al., Phase II trial of nanoparticle albumin-bound paclitaxel as second-line chemotherapy for unresectable or recurrent gastric cancer. Cancer Sci 2014; 105(7): 812-817
- Gradishar WJ et al., Significantly longer progression-free survival with nab-paclitaxel compared with docetaxel as first-line therapy for metastatic breast cancer. J Clin Oncol 2009; 27(22): 3611-3619
- Sakata S et al., Phase II trial of weekly nab-paclitaxel for previously treated advanced non-small cell lung cancer: Kumamoto thoracic oncology study group (KTOSG) trial 1301. Lung Cancer 2016; 99: 41-45
- Nakamura A et al., Phase III study comparing nab-paclitaxel with docetaxel in patients with previously treated advanced non-small cell lung cancer_J-AXEL. WCLC 2020, OA03.05
© 2020 Springer-Verlag GmbH, Impressum
More posts
KRAS, HER2 & ALK: targeted options and sequencing issues
The KRAS p.G12C mutation is a key oncogenic driver occurring in approximately 13 % of lung adenocarcinomas and is associated with poor patient outcomes. The first-in-class, highly selective and irreversible KRASG12C inhibitor sotorasib has shown durable clinical benefit in a cohort of 59 heavily pretreated patients with NSCLC included in phase I of the CodeBreaK 100 study.
Pushing the bounds in early-stage lung cancer
Approximately 30 % of patients with non-small-cell lung cancer (NSCLC) present with resectable disease at diagnosis. Surgery with curative intent is the recommended treatment here, followed by adjuvant cisplatin-based chemotherapy in stage II/IIIA and select cases of stage IB disease. However, recurrence rates remain high across disease stages, regardless of postoperative chemotherapy use.
Preface
The 2020 World Conference of Lung Cancer (WCLC) originally scheduled for August 2020 in Singapore had to be postponed to January 2021 due to the COVID-19 pandemic and was finally held as a worldwide virtual conference from 28th to 31st January. WCLC, which is the leading gathering of international scientists, researchers and patient advocates in the field of lung cancer and thoracic malignancies, continues to provide a forum to connect, share knowledge and learn about the latest developments in the research and treatment of these diseases.