Anti-angiogenic and immunotherapeutic approaches in mesothelioma
The LUME-Meso trial
Malignant pleural mesothelioma (MPM) is a rare tumour that is often diagnosed at an advanced stage. Limited efficacy of the available therapies contributes to the generally poor prognosis for MPM patients. Since 2003, the only approved regimen for MPM treatment has been chemotherapy with pemetrexed and cisplatin, with median survival of approximately 12 months [1].
The oral multikinase inhibitor nintedanib strongly inhibits MPM tumour growth in human xenograft models and reduces the colony-forming capacity and migratory activity of MPM cell lines [2, 3]. Based on these observations, the randomised, double-blind, phase II LUME Meso study investigated pemetrexed/ cisplatin plus either nintedanib or placebo in 87 patients with unresectable MPM who had not received prior chemotherapy. Patients who completed up to six cycles of chemotherapy without progression were able to continue with nintedanib or placebo maintenance therapy until progression. The primary analysis, which was presented in 2016, showed that the addition of nintedanib to chemotherapy induced clinically meaningful PFS improvement (9.4 vs. 5.7 months; HR, 0.56; p = 0.017) [4]. At that time, there was a trend for OS prolongation (18.3 vs. 14.5 months; HR, 0.78).
First-line benefit particularly in the epithelioid subgroup
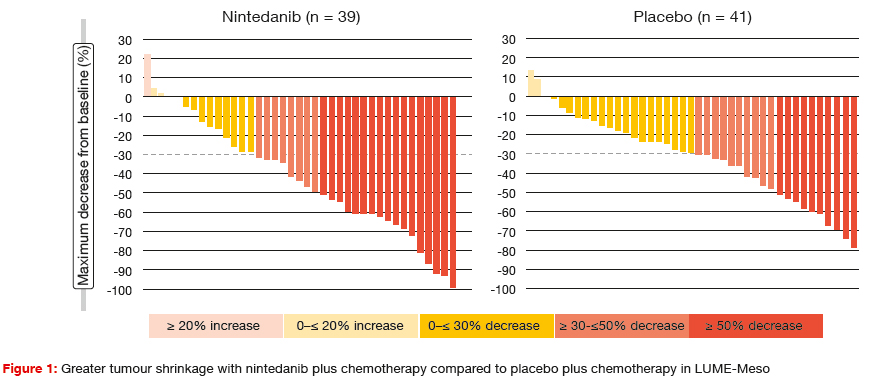
According to the primary OS analysis reported at the ASCO Congress, patients treated with nintedanib in the intention- to-treat (ITT) population derived a 4.1-month OS gain compared to the control arm (18.3 vs. 14.2 months; HR, 0.77; p = 0.319) [5]. A pre-planned subset analysis of patients whose tumours had epithelioid histology showed a survival advantage of 5.4 months (20.6 vs. 15.2 months; HR, 0.70; p = 0.197). Likewise, the PFS benefit according to the updated PFS analysis was greater for the cohort with epithelioid tumours (9.7 vs. 5.7 months; HR, 0.49; p = 0.006) than for the ITT population (9.4 vs. 5.7 months; HR, 0.54; p = 0.010). The addition of nintedanib led to deeper responses (Figure 1), with corresponding improvements in ORR (57 % vs. 44 %) and median duration of response (6.0 vs. 4.0 months). Importantly, nintedanib did not compromise delivery of the backbone chemotherapy. The safety profile of nintedanib was manageable and consistent with previous studies. Nintedanib-treated patients showed no excess of all-grade bleeding, thromboembolism or hypertension, although higher rates were noted in the experimental arm with respect to grade ≥ 3 hypertension. The ongoing LUME-Meso phase III trial is comparing nintedanib plus chemotherapy with chemotherapy alone in patients with epithelioid histology only.
Activity of second-line trabectedin: ATREUS
Validated treatment options beyond the failure of pemetrexed-based chemotherapy are lacking at present. As inflammation is a fundamental characteristic of MPM, there might be a rationale for the use of the alkylating agent trabectedin. Responses to trabectedin have been related to modulation of cytokines and chemokines, among others. Therefore, the single-arm, multi-centre, phase II ATREUS trial was designed to determine the activity of trabectedin in MPM patients. ATREUS included a pretreated cohort with epithelioid histology, and a treatment-naive and pretreated cohort with biphasic/ sarcomatoid histology. Patient enrollment in the biphasic/ sarcomatoid cohort is ongoing. Preliminary results obtained in the epithelioid group, for which recruitment is complete, suggest that trabectedin can be considered as a new option in patients with epithelioid MPM that has relapsed after platinum–pemetrexed therapy [6]. The proportion of patients free from progression or death at 12 weeks constituted the primary endpoint of the preliminary analysis. Twenty-five patients met this criteria which represented 42.4 % of patients in the per-protocol analysis set (n = 58), and 38.5 % of those in the ‘withdrawn considered failure’ group (n = 65). This latter group included all of the patients withdrawn before 12 weeks, with these considered as failures. Here, median PFS was 2.5 months, and OS was 9.4 months. At the end of the 18-month treatment period, one patient was free of progression, and 11 were still alive.
Transaminase elevations were the major concern observed with trabectedin treatment, but they were mild and recovered after treatment delay or dose reduction in the majority of cases. Other frequent AEs included fatigue, nausea, and respiratory toxicity. Most events were transient and manageable. Only a limited number of patients interrupted treatment due to toxicity. Based on these encouraging results, a phase III trial to evaluate trabectedin in pre-treated epithelioid MPM appears warranted.
Meaningful disease control through checkpoint inhibition
Another approach worth investigating is immunotherapeutic treatment, as MPM has been shown to be potentially immunogenic. PD-L1 expression is associated with poor prognosis in MPM patients [7, 8]. Conversely, those with high levels of intra-tumour cytotoxic CD8-positive T cells in resected MPM samples were shown to have better prognosis [9]. The randomised, non-comparative, phase II MAPS-2 study evaluated nivolumab 3 mg/kg every 2 weeks (n = 63) alone or in combination with ipilimumab 1 mg/kg every 6 weeks (n = 62) in patients with unresectable MPM that had progressed after a maximum of one or two previous lines of chemotherapy, including a pemetrexed/ platinum doublet [10]. The primary endpoint was DCR at 12 weeks, as centrally assessed by an independent and blinded expert panel of radiologists, according to the modified RECIST-meso criteria.
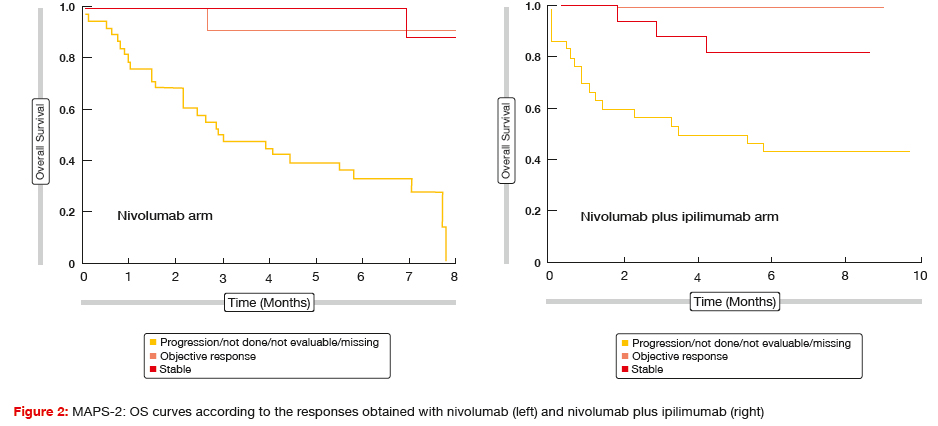
Both the nivolumab-alone regimen and the nivolumab plus ipilimumab regimen induced clinically meaningful disease control. DCR at 12 weeks for the first 108 eligible patients (i.e., the primary endpoint based on the statistical plan) was 44.4 % and 50.0 % for nivolumab and nivolumab plus ipilimumab, respectively. For the ITT population, which included 125 patients, the respective rates were 39.7 % and 51.6 %, and median PFS amounted to 4.0 and 5.6 months, respectively. Remarkably, these results resemble those generally achieved in the first-line setting. Preliminary OS in the ITT cohort was 10.4 months and not reached, respectively. Thus, patients from both arms of this study appeared to have prolonged median OS compared to all of the previous reports in the second-line/ third-line treatment setting of MPM. An OS analysis according to response showed that patients who achieved disease control had excellent survival in both arms (Figure 2).
Toxicity was globally manageable, although three treatment-related deaths occurred in the combination arm due to fulminant hepatitis, encephalitis, and acute renal failure. The authors concluded that immunotherapy with nivolumab and ipilimumab might provide a new therapeutic option as second-line or third-line treatment for relapsing MPM patients. The randomised, open-label, phase III CheckMate 743 trial is currently investigating first-line nivolumab plus ipilimumab compared to pemetrexed plus cisplatin or carboplatin in unresectable MPM [11]. The primary results of this study should become available in October 2020.
PD-L1 expression and beyond
Rivalland et al. evaluated the effects of PD-L1 expression on clinical outcomes in 46 patients with unresectable pleural or peritoneal malignant mesothelioma who received treatment with anti-PD-1 antibodies [12]. PD-1 inhibition demonstrated clinically meaningful activity. Disease control was achieved in 48 %, and median PFS and OS were 3.1 and 8.0 months, respectively. The initial analysis suggested that PD-L1 expression correlates with improved response and survival, especially in cases with TPS > 50 %. Thirty-six percent of patients expressed PD-L1 at > 5 % (PDL1+), and 23 % at ≥ 50 % (PD-L1hi). In PD-L1+ cases, ORR and OS were 38 % and 8.9 months, respectively. For those with PD-L1hi, these were 60 % and not yet reached, respectively. On the other hand, patients with PD-L1low (< 50 % staining) demonstrated lower ORR and OS (12 % and 4.8 months, respectively).
As well as PD-L1, the expression of other checkpoint receptors and their interplay in the MPM tumour microenvironment might affect the design of trials to evaluate single or combination checkpoint inhibition. In an assessment of 329 MPM cases, Thapa et al. showed significant expression of PD-L1, PD-L2, and TIM3 [13]. The expression of these markers was mutually exclusive in a significant proportion of samples. As the authors noted, a comprehensive assessment of multiple immunosuppressive pathways might be necessary to truly gauge the immunosuppressive environment to allow tailoring of immunotherapy for individual cases.
References:
- Vogelzang NJ et al., Phase III study of pemetrexed in combination with cisplatin versus cisplatin alone in patients with malignant pleural mesothelioma. J Clin Oncol 2003; 21(14): 2636-2644
- Lakatos et al., University of Budapest Symposium, 5-6 Nov 2015
- Laszlo et al., 13th International Mesothelioma Interest Group 2016
- Grosso F et al., Nintedanib plus pemetrexed/ cisplatin in patients with MPM: phase II findings from the placebo-controlled LUME-Meso trial, WCLC 2016, OA22.02
- Nowak AK et al., Mature overall survival results from the LUME-Meso study of nintedanib + pemetrexed/ cisplatin vs placebo + pemetrexed/cisplatin in chemo-naïve patients with malignant pleural mesothelioma. ASCO 2017, abstract 8506
- Cortinovis D et al., Trabectedin (t) as second line treatment option for patients with epithelioid malignant pleural mesothelioma (MPM) in progression following pemetrexed/platin-derivates chemotherapy: ATREUS trial. ASCO 2017, abstract 8513
- Cedrés S et al., Analysis of expression of programmed cell death 1 ligand 1 (PD-L1) in malignant pleural mesothelioma (MPM). PLoS One 2015; 10(3): e0121071
- Combaz-Lair C et al., Immune biomarkers PD-1/PD-L1 and TLR3 in malignant pleural mesotheliomas. Hum Pathol 2016; 52: 9-18
- Lievense LA et al., Checkpoint blockade in lung cancer and mesothelioma. Am J Respir Crit Care Med 2017. doi: 10.1164/rccm.201608- 1755CI. [Epub ahead of print]
- Scherpereel A et al., Second or third-line nivolumab versus nivolumab plus ipilimumab in malignant pleural mesothelioma patients: results of the IFCT-1501 MAPS-2 randomized phase 3 trial. ASCO 2017, abstract LBA8507
- Zalcman G et al., CheckMate 743: a phase III, randomized, open-label trial of nivolumab plus ipilimumab vs. pemetrexed plus cisplatin or carboplatin as first-line therapy in unresectable pleural mesothelioma. ASCO 2017, abstract TPS8581
- Rivalland G et al., Outcomes of anti-PD-1 therapy in mesothelioma and correlation with PDL1 expression. ASCO 2017, abstract 8514
- Thapa B et al., Immune microenvironment in mesothelioma: Looking beyond PD-L1. ASCO 2017, abstract 8515
More posts
Anti-angiogenic and immunotherapeutic approaches in mesothelioma
Malignant pleural mesothelioma (MPM) is a rare tumour that is often diagnosed at an advanced stage. Limited efficacy of the available therapies contributes to the generally poor prognosis for MPM patients. Since 2003, the only approved regimen for MPM treatment has been chemotherapy with pemetrexed and cisplatin, with median survival of approximately 12 months.
Interview: Lung cancer in China: hurdles and progress
Lung cancer is a considerable issue in China. Every year, we have 700,000 new cases. There is a need to perform clinical trials and to launch innovative drugs. With regard to the introduction of targeted therapies, China lags 3 to 4 years behind when compared to the western countries. Two months ago, the EGFR TKI afatinib was launched, offering Chinese patients with EGFR-mutant lung cancer an effective treatment option.
Real-world utility of ctDNA NGS to identify matched targeted therapy
Liquid biopsy for plasma circulating tumour DNA (ctDNA) next generation sequencing (NGS) is a rapidly evolving science. Plasma ctDNA assays are now commercially available, and are increasingly adopted in the community with a paucity of evidence-based guidance on timing and value of this test. Sabari et al. sought to determine the feasibility and utility of plasma ctDNA NGS to identify matched targeted therapy in a real-world clinical setting.
Further defining the optimal use of immune checkpoint inhibitors
As the anti-PD-1 antibody nivolumab is known to induce deep and durable responses in a subset of lung cancer patients, this agent was investigated in the neoadjuvant setting, which is an area of unmet need. There have been no advances in systemic treatment of resectable lung cancer since 2004. Chaft et al. hypothesised that neoadjuvant nivolumab treatment might induce immunity against micrometastases.
Established targeted agents taking root in the HER2-positive setting
HER2 aberrations in lung cancer are being increasingly identified due to the use of sensitive testing procedures, such as multiplexed testing and next-generation sequencing. Mutations of the HER2 gene need to be distinguished from HER2 amplifications and HER2 protein overexpression. In contrast to breast and gastric cancer, HER2 overexpression in NSCLC does not always occur with HER2 amplification, while amplifications and HER2 mutations are generally mutually exclusive.
Diagnostics of EGFR-mutant disease: biomarkers with significant clinical implications
The clinical relevance of additional genetic alterations in advanced EGFR-mutant NSCLC is not clear. Blakely et al. hypothesised that co-occurring genomic alterations in cancer-related genes can cooperate with the mutant EGFR to drive de-novo resistance to EGFR TKI treatments. The investigators performed targeted exome sequencing of plasma cell-free DNA (cfDNA) in 86 samples collected from 81 patients with known clinical history.