Paroxysmal nocturnal hemoglobinuria: improving outcomes with novel strategies
Paroxysmal nocturnal hemoglobinuria (PNH) is a rare, potentially life-threatening clonal hematopoietic stem cell disorder characterized by hemolytic anemia, bone marrow failure, thrombosis, and peripheral blood cytopenia. The disease results from an acquired loss-of-function mutation of the PIGA gene involved in the synthesis of the glycosylphosphatidylinositol-anchored complement inhibitors CD55 and CD59 [1-4]. The absence of these proteins leads to uncontrolled activation of the terminal complement pathway and complement-mediated lysis of erythrocytes. PNH is associated with a high burden of disease and impaired health-related quality of life [4].
Treatment options for hemolytic PNH remained limited until the monoclonal antibody eculizumab became available. Eculizumab is administered intravenously; this agent targets the component 5 (C5) of the complement cascade, thereby preventing intravascular hemolysis [5, 6]. However, only one third of PNH patients was shown to achieve complete normalization of hemoglobin levels with eculizumab treatment [7]. Many continue to experience some degree of anemia, in some cases requiring regular red blood cell transfusions. Factors contributing to residual anemia include underlying bone marrow dysfunction, residual intravascular hemolysis, and the emergence of C3-mediated extravascular hemolysis, which is not improved by C5 inhibitors such as eculizumab [8]. Therefore, novel anti-complement treatment approaches focus on some of these mechanisms. Strategies that are being investigated in the setting of proximal complement inhibition include agents directed against C3 as well as factors D and B that are involved in the formation of the alternative pathway C3 convertase.
PEGASUS: pegcetacoplan induces hemoglobin increase
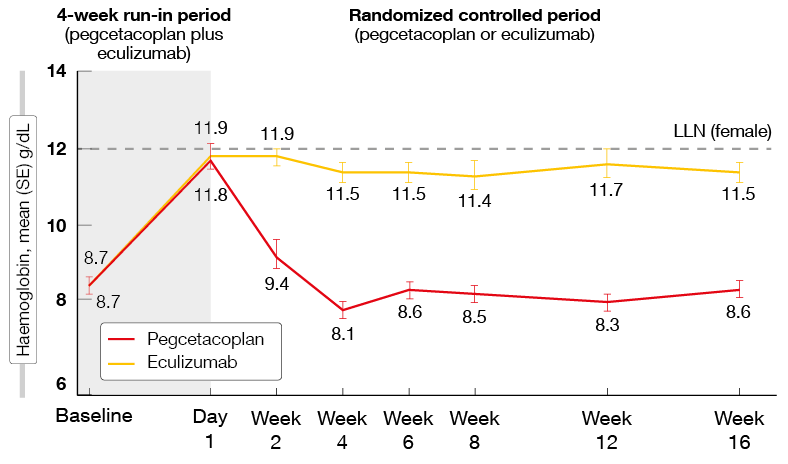
The investigational compound pegcetacoplan is a subcutaneously administered C3 inhibitor that has the potential to control both intra- and extravascular hemolysis in PNH [9]. Pegcetacoplan monotherapy was assessed in the randomized, open-label, controlled, phase III PEGASUS trial that included PNH patients who had been on eculizumab treatment for ≥ 6 months but still showed hemoglobin levels < 10.5 g/dL [10]. During the 4-week run-in period, all patients received pegcetacoplan 1,080 mg twice weekly in addition to the eculizumab standard dose. Subsequently, they were randomized to either pegcetacoplan 1,080 mg twice weekly (n = 41) or their current dose regimen of eculizumab (n = 39). The primary endpoint of the study was the change in hemoglobin from baseline level to week 16. At that time, the control patients crossed over and the entire population continued single-agent pegcetacoplan therapy for a 32-week open-label period.
The experimental treatment resulted in a highly significant improvement in hemoglobin levels compared to the standard of care, with an adjusted treatment difference of 3.84 g/dL (p < 0.0001). Baseline hemoglobin had been 8.7 g/dL in both groups; while this increased by 2.37 g/dL with pegcetacoplan, it further decreased by 1.47 g/dL in the eculizumab-treated group. This effect was observed irrespective of transfusion history (< 4 vs. ≥ 4 transfusion events). The Kaplan-Meier estimate shows that the increase was achieved during the run-in period and maintained in the pegcetacoplan arm, whereas it was lost again in the control patients whose hemoglobin levels returned to the baseline values within 4 weeks after randomization (Figure 1).
Figure 1: PEGASUS: hemoglobin increase is maintained with pegcetacoplan therapy but lost with eculizumab after randomization
Benefits regarding secondary outcomes
With respect to the key secondary endpoint of transfusion avoidance, the analysis yielded an adjusted risk difference of 62.5 % favoring pegcetacoplan (85.4 % vs. 15.4 %). This benefit was particularly pronounced in patients who had a history of high transfusion requirements (85.7 % vs. 4.3 %), but also emerged in those with low numbers of transfusions (85.0 % vs. 31.3 %). Reticulocyte counts decreased with pegcetacoplan treatment by 136 x 109/L but increased with eculizumab by 28 x 109/L, resulting in a significant difference of 164 x 109/L. According to the sensitivity analysis without censoring for transfusion, normal ranges were restored for lactate dehydrogenase (LDH) levels, reticulocyte counts and bilirubin levels with pegcetacoplan, while eculizumab hardly affected these outcomes. Normalization regarding hemoglobin levels was achieved in 34 % vs. 0 %; for LDH levels, this was 70.7 % vs. 15.4 %, and for reticulocytes, 78 % vs. 2.6 %. Patients in the experimental arm also had less fatigue as assessed by the FACIT-Fatigue score throughout the study. Here, 73.2 % showed improvement of ≥ 3 points from baseline, whereas none did with eculizumab.
Adverse events were mainly mild or moderate. Among AEs of interest, injection site reactions occurred most frequently (36.6 % vs. 2. 6 % with pegcetacoplan and eculizumab, respectively), although the majority of events were mild and restricted to the initial treatment period. Discontinuations due to breakthrough hemolysis were seen in 3 patients in the experimental arm; 2 of these had lower than expected serum concentrations of pegcetacoplan prior to the events, and neither patient increased dosing to 1,080 mg every 3 days prior to treatment discontinuation. Overall, the PEGASUS results highlighted the ability of pegcetacoplan to control both intravascular and extravascular hemolysis in patients with PNH, leading to a potential new treatment option.
Age-related activity of eculizumab
Lee et al. presented an analysis evaluating the clinical outcomes obtained with eculizumab in patients aged ≥ 65 years included in the International PNH Registry [11]. This registry is an ongoing, prospective, international study on the natural history of PNH and the long-term efficacy and safety of eculizumab. Adult patients (aged 18-64 years, n = 1,537) were compared with advanced-age patients (n = 270) enrolled in the registry.
The results suggested age-independent efficacy of eculizumab in terms of reduction of intravascular hemolysis, obtaining transfusion independence, and prevention of thrombotic events and major adverse vascular events. Both patients in the adult and advanced-age cohorts achieved substantial reductions in the LDH ratio from more than 5 times the upper limit of normal (ULN) at baseline to normal or near normal range at last follow-up. Transfusion independence was achieved by approximately one third in both groups (35.9 % vs. 31.2 %). Also, changes in the proportions of patients with physician-reported PNH-related symptoms (e.g. abdominal pain, dysphagia, dyspnea) were comparable. However, younger patients showed significantly higher increases in hemoglobin levels from baseline to last follow-up (1.4 g/dL vs. 0.4 g/dL; p < 0.0001). Major vascular events occurred significantly more frequently in the advanced-age cohort, although both cohorts experienced similar changes from baseline to last follow-up regarding this outcome.
Infections occurred at low and comparable rates across cohorts. A larger proportion of patients in the advanced-age group died, although these deaths were generally unrelated to treatment with eculizumab. The authors concluded that eculizumab is effective and well tolerated as treatment of PNH in patients of advanced age in a real-world setting.
Danicopan: effect on transfusion requirements
The orally available factor D inhibitor danicopan blocks C3 convertase formation, thus potentially controlling both intra- and extravascular hemolysis. A phase II dose-finding, proof-of-concept trial tested the addition of danicopan to the current eculizumab regimen in PNH patients with an inadequate response to eculizumab who were transfusion-dependent. Eleven of 12 patients who completed treatment achieved clinically meaningful improvements in hemoglobin levels, transfusion needs, and other parameters [12].
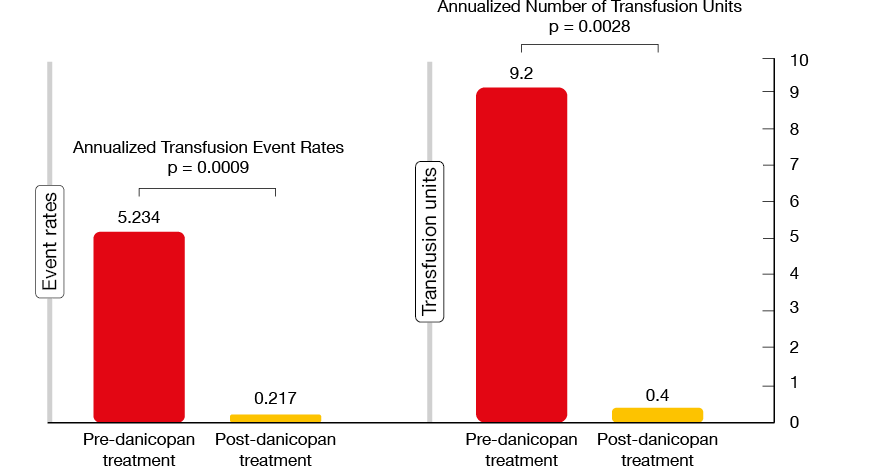
A post-hoc analysis of this study presented at EHA 2020 evaluated the impact of the addition of danicopan on transfusion requirements [13]. In the course of 24 weeks, the hemoglobin level increased from 7.9 g/dL to 10.3 g/dL, and the transfusion frequency showed a highly statistically significant 95.8 % reduction (p = 0.0009; Figure 2). This also applied to the number of transfused red cell units (p = 0.0028). As the authors noted, the added benefit is likely due to the prevention of C3-mediated extravascular hemolysis. Danicopan was generally well tolerated. Almost all treatment-emergent AEs were mild or moderate except for one case of breakthrough hemolysis and a case of severe pneumonia in a patient who had a history of neutropenia.
Figure 2: Impact of danicopan treatment on annualized transfusion requirements
Long-acting formulation of ravulizumab
Ravulizumab, a C5 inhibitor designed for intravenous application, is an emerging standard of care for patients with PNH in countries where it has been approved. The long duration of action of this agent enables a decreased infusion frequency with dosing intervals of 8 weeks. Ravulizumab 10 mg/mL has demonstrated efficacy and safety in two large phase III trials [14, 15]. An open-label, phase II study assessed multiple ascending doses of ravulizumab in complement-inhibitor–naïve patients with PNH based on the observation that the 100 mg/mL formulation decreases the dose infusion time by 78-102 minutes compared with the 10 mg/mL formulation. Twenty-five patients were divided into 4 cohorts that received ravulizumab maintenance doses of 1,000 mg/4 weeks, 1,600 mg/6 weeks, 2,400 mg/8 weeks, or 5,400 mg/12 weeks. After the initial treatment period, cohorts 1-3 began weight-based dosing regimens during the extension period, which is ongoing. All cohorts started on ravulizumab 10 mg/mL and switched to 100 mg/mL during the extension period.
The interim analysis showed similar efficacy, safety, pharmacokinetics, and immunogenicity of ravulizumab 100 mg/mL compared to the 10 mg/mL formulation [16]. LDH levels did not change significantly after the switch in all cohorts. Treatment-emergent AEs were consistent with the established safety profile of ravulizumab 10 mg/mL. No toxicities necessitated treatment discontinuation or interruption. Also, serum trough concentrations did not differ in a meaningful manner after the switch, and neither formulation gave rise to anti-drug-antibody responses. Compared with the 10 mg/mL formulation, the infusion times were reduced by 78-102 minutes. The investigators concluded that ravulizumab 100 mg/mL provides a reduction in infusion time of 60 % to 77 % while maintaining efficacy, thus reducing the treatment burden for patients, their caregivers, and healthcare providers.
Novel C5 inhibitor pozelimab
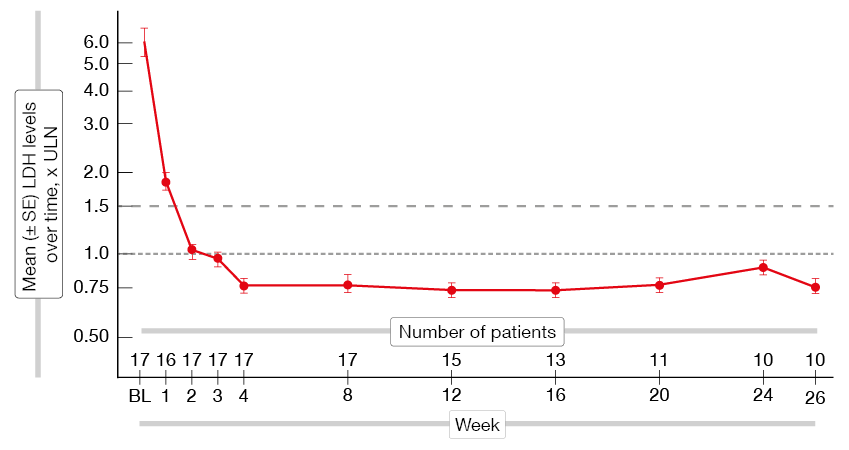
An ongoing, open-label, single-arm, phase II study is assessing the C5 inhibitor pozelimab in patients with active PNH who are treatment-naïve to complement inhibitor therapy or have not recently received complement inhibition [17]. Pozelimab is administered subcutaneously once weekly for 26 weeks. An interim analysis including 17 patients demonstrated that pozelimab led to rapid and durable LDH responses (Figure 3). All patients achieved reductions to below the clinically significant threshold of ≤ 1.5 x ULN that were sustained until study day 183. At week 2, 16 individuals achieved control of intravascular hemolysis (LDH, ≤ 1.5 x ULN), and at week 4, normalization of LDH levels (≤ 1.0 x ULN) was observed in all but one patient. Importantly, one patient who is a carrier of a C5 variant known to be resistant to blockade by eculizumab/ravulizumab showed rapid and sustained normalization of LDH. Hemoglobin levels increased over 26 weeks, which was mirrored by improvement of the FACIT-Fatigue score. Pozelimab was well tolerated. No serious AEs, AEs leading to treatment discontinuation, or breakthrough hemolysis events occurred. Headache and nausea were reported as the most common AEs.
According to the investigators, these interim data support the continued development of pozelimab for the treatment of PNH and potentially other complement-mediated diseases. The findings indicated that a subcutaneous regimen might provide an alternative to currently available intravenous regimens.
Figure 3: Rapid and sustained reduction of the LDH levels with pozelimab
REFERENCES
- Takeda J et al., Deficiency of the GPI anchor caused by a somatic mutation of the PIGA gene in paroxysmal nocturnal hemoglobinuria. Cell 1993: 73:703-711
- Bessler M et al., Paroxysmal nocturnal haemoglobinuria (PNH) is caused by somatic mutations in the PIG-A gene. EMBO J 1994; 13: 110-117
- Miyata T et al., Abnormalities of PIG-A transcripts in granulocytes from patients with paroxysmal nocturnal hemoglobinuria. N Engl J Med 1994: 330: 249-255
- Brodsky RA, Paroxysmal nocturnal hemoglobinuria. Blood 2014; 124(18): 2804-2811
- Hillmen P et al., The complement inhibitor eculizumab in paroxysmal nocturnal hemoglobinuria. N Engl J Med 2006; 355: 1233-1243
- Brodsky RA et al., Multicenter phase 3 study of the complement inhibitor eculizumab for the treatment of patients with paroxysmal nocturnal hemoglobinuria. Blood 2008; 111: 1840-1847
- Risitano AM et al., Complement fraction 3 binding on erythrocytes as additional mechanism of disease in paroxysmal nocturnal hemoglobinuria patients treated by eculizumab. Blood 2009; 113: 4094-4100
- Risitano AM et al, Anti-complement treatment for paroxysmal nocturnal hemoglobinuria: time for proximal complement inhibition? A position paper from the SAAWP of the EBMT. Front Immunol 2019; 10: 1157
- Risitano AM et al., Peptide inhibitors of C3 activation as a novel strategy of complement inhibition for the treatment of paroxysmal nocturnal hemoglobinuria. Blood 2014; 123(13): 2094-2101
- Hillmen P et al., Results of the PEGASUS phase 3 randomized trial demonstrating superiority of the C3 inhibitor, pegcetacoplan, compared to eculizumab in patients with paroxysmal nocturnal hemoglobinuria. EHA 2020, abstract S192
- Lee JW et al., Effectiveness of eculizumab treatment in patients aged ≥ 65 years with paroxysmal nocturnal hemoglobinuria (PNH): results from the International PNH Registry. EHA 2020, abstract EP853
- Kulasekararaj A et al., A phase 2 open-label study of danicopan (ACH-0144471) in patients with paroxysmal nocturnal hemoglobinuria (PNH) who have an inadequate response to eculizumab monotherapy. Blood (2019) 134 (Supplement_1): 3514
- Kulasekararaj A et al., Effects of oral, factor D inhibitor danicopan on transfusion rates in transfusion-dependent paroxysmal nocturnal hemoglobinuria (PNH) patients with an inadequate response to eculizumab: phase 2 study. EHA 2020, EP855
- Lee JW et al., Ravulizumab (ALXN1210) vs eculizumab in adult patients with PNH naive to complement inhibitors: the 301 Study. Blood 2019; 133(6): 530-539
- Kulasekararaj AG et al., Ravulizumab (ALXN1210) vs eculizumab in C5-inhibitor–experienced adult patients with PNH: the 302 study. Blood 2019; 133(6): 540-549
- Röth A et al., An interim analysis of a phase 2 study evaluating the efficacy, safety, and pharmacokinetics of intravenous ravulizumab 100 mg/mL formulation in patients with paroxysmal nocturnal hemoglobinuria. EHA 2020, abstract EP862
- Jang JH et al., Pozelimab inhibits hemolysis in patients with paroxysmal nocturnal hemoglobinuria. EHA 2020, abstract EP860
© 2019 Springer-Verlag GmbH, Impressum
More posts
Patient and disease characteristics in a small CAD cohort
A retrospective analysis hints at the wide range of cold agglutinin disease (CAD) clinical behavior. Koudouna et al. investigated the characteristics of 8 patients with CAD at the time of diagnosis [1]. Median age was 62 years, and 5 patients were women. Hematologic malignancies constituted 50 % of underlying medical conditions; in 37 %, hepatitis B/C was the associated disease, and in 13 %, autoimmune disorders.
Cold agglutinin disease: on the road to new insights and potential treatment options
Cold agglutinin disease (CAD) is a rare type of autoimmune hemolytic anemia (AIHA) elicited by cold-sensitive antibodies including cold agglutinins. Ninety percent of cold agglutinins belong to the IgM kappa category and bind to red blood cell surface antigens at temperatures of ≤ 37 °C, thus inducing hemolysis.
Paroxysmal nocturnal hemoglobinuria: improving outcomes with novel strategies
Paroxysmal nocturnal hemoglobinuria (PNH) is a rare, potentially life-threatening clonal hematopoietic stem cell disorder characterized by hemolytic anemia, bone marrow failure, thrombosis, and peripheral blood cytopenia. The disease results from an acquired loss-of-function mutation of the PIGA gene involved in the synthesis of the glycosylphosphatidylinositol-anchored complement inhibitors CD55 and CD59.
Targeted approaches in various B-cell malignancies
BTK inhibitors are active in many B-cell malignancies such as mantle cell lymphoma, CLL and Waldenström’s macroglobulinemia, but also in diffuse large B-cell lymphoma (DLBCL), follicular lymphoma (FL), and marginal zone lymphoma (MZL). Zanubrutinib is currently being assessed in pivotal phase II and III studies in all of these indications.
Changing paradigms in the management of mantle cell lymphoma
Mantle cell lymphoma (MCL) is a rare, heterogenous and generally aggressive subtype of B-cell non-Hodgkin lymphoma that remains incurable in the majority of cases. Median survival in non-trial patients has been estimated at 3 to 5 years. First-line therapy usually consists of chemoimmunotherapy, while both immunochemotherapy and targeted agents are recommended in relapsed disease.
Optimizing timing, efficacy and tolerability in chronic lymphocytic leukemia
In both treatment-naïve and relapsed/refractory patients with chronic lymphocytic leukemia (CLL) and small lymphocytic leukemia (SLL), inhibition of Bruton’s tyrosine kinase (BTK) represents a treatment standard as it has improved clinical outcomes. Compared to the first-generation agent ibrutinib, the second-generation, highly selective BTK inhibitor acalabrutinib shows minimal off-target kinase inhibition, thus potentially offering an optimized safety profile.