Optimizing timing, efficacy and tolerability in chronic lymphocytic leukemia
Acalabrutinib vs. ibrutinib
In both treatment-naïve and relapsed/refractory patients with chronic lymphocytic leukemia (CLL) and small lymphocytic leukemia (SLL), inhibition of Bruton’s tyrosine kinase (BTK) represents a treatment standard as it has improved clinical outcomes [1]. Compared to the first-generation agent ibrutinib, the second-generation, highly selective BTK inhibitor acalabrutinib shows minimal off-target kinase inhibition [2], thus potentially offering an optimized safety profile. Acalabrutinib has been approved for the treatment of CLL in various countries including the USA, Australia, and India.
Given the lack of head-to-head trials, Davids et al. conducted matching-adjusted indirect comparisons of acalabrutinib and ibrutinib either as monotherapies or in combination with the anti-CD20 antibody obinutuzumab in patients with treatment-naïve CLL [3]. Indeed, the analysis suggested lower rates of clinically important AEs with the second-generation BTK inhibitor. At the same time, acalabrutinib with and without obinutuzumab showed a trend towards improved PFS and OS compared to ibrutinib with and without obinutuzumab. Acalabrutinib monotherapy significantly reduced the mortality risk versus ibrutinib plus obinutuzumab by 84 % (p < 0.001). As the authors noted, this warrants further investigations. The ongoing, randomized, head-to-head ELEVATE-CLL R/R study comparing ibrutinib with acalabrutinib will provide answers in a prospective manner.
ASCEND: final results in r/r CLL
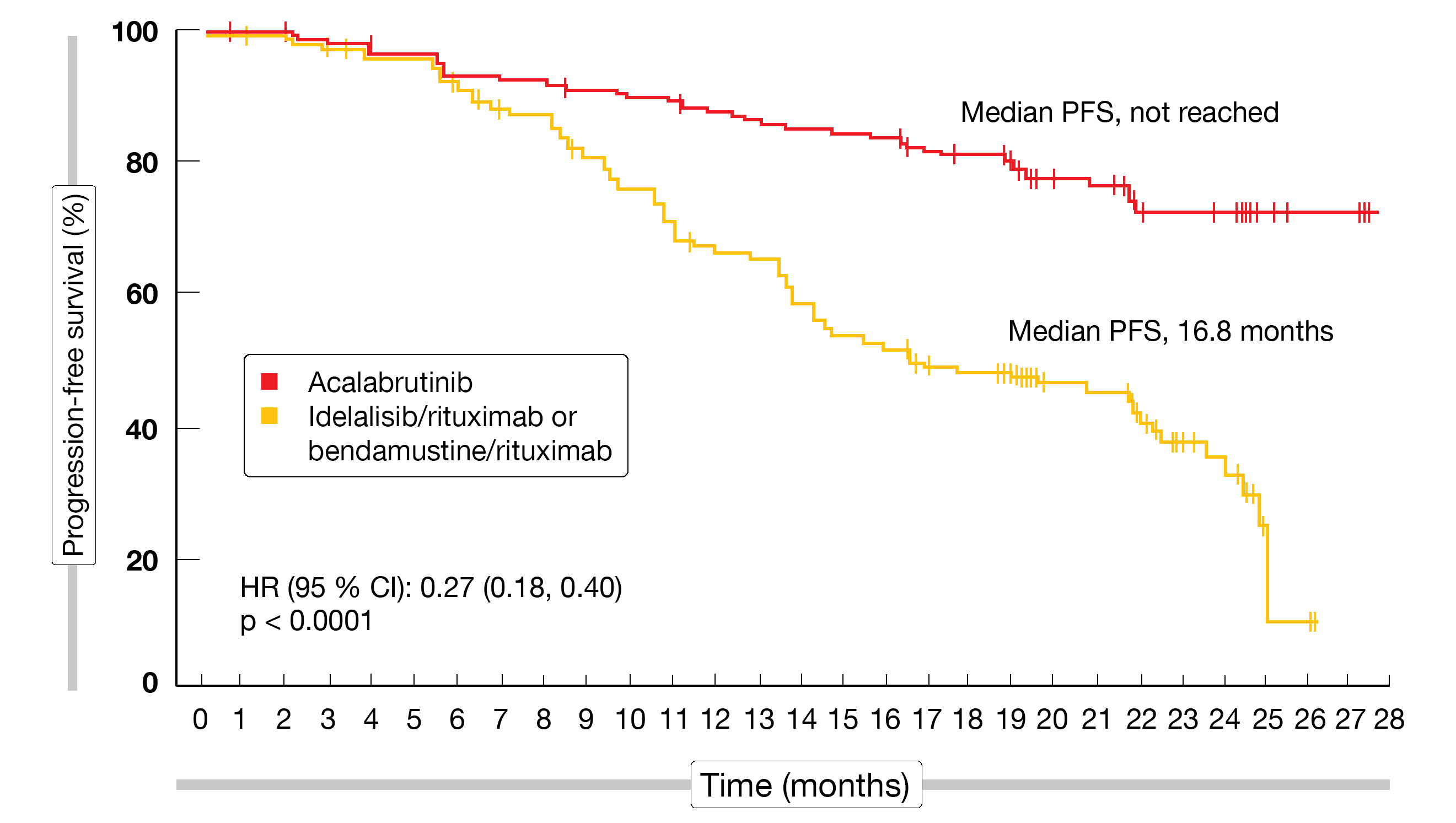
The randomized, phase III ASCEND study compared acalabrutinib (n = 155) with investigator’s choice of idelalisib/rituximab (n = 119) or bendamustine/rituximab (n = 36) in patients with relapsed/refractory CLL (r/r CLL). After a median follow-up of 16.1 months, the pre-planned interim analysis already demonstrated significant superiority of acalabrutinib versus the comparator regimens with regard to PFS [4]. At the EHA 2020 Congress, Ghia et al. reported the final results of ASCEND after 22 months [5].
The findings confirmed the results of the interim analysis, supporting the favorable efficacy and safety of acalabrutinib. PFS was significantly prolonged, with a 73 % reduction in the risk of progression or death (not reached vs. 16.8 months; HR, 0.27; p < 0.0001; Figure 1). At 18 months, PFS rates were 82 % vs. 48 % for acalabrutinib and the comparator regimens, respectively. Significant PFS benefits were also observed in patients with high-risk genetics including 17p deletion and TP53 mutations (HR, 0.11) and unmutated IGHV (HR, 0.28). The ORRs did not differ significantly across the arms, although the duration of response was significantly longer in the experimental arm (not reached vs. 18.0 months; HR, 0.19). Responses persisted in 85.4 % vs. 49.4 % at 18 months.
The incidences of grade ≥ 3 AEs, serious AEs, treatment-related AEs, drug discontinuations and dose modifications were lower with acalabrutinib than with idelalisib/rituximab, and similar to the respective rates observed for bendamustine/rituximab. Among events of clinical interest, any-grade hemorrhages were more common with acalabrutinib, but the incidence of major bleeding events was low and similar across arms. These data support the use of acalabrutinib in patients with r/r CLL, including those with high-risk features.
Figure 1: ASCEND: progression-free survival for acalabrutinib vs. idelalisib/rituximab or bendamustine/rituximab in the relapsed/refractory setting
Long-term acalabrutinib therapy
In the untreated symptomatic setting, the mature results of the single-arm, phase II ACE-CL-001 trial provide the longest safety and efficacy follow-up to date for single-agent acalabrutinib [6]. After 53 months, the analysis showed durable remissions and long-term tolerability of this treatment in 99 patients. The ORR amounted to 97 %, with CR and PR rates of 7 % and 90 %, respectively. Median duration of response had not been reached yet. In each high-risk group (i.e., unmutated IGHV, 17p deletion, TP53 mutation, complex karyotype), the ORR was 100 %. Reductions in lymph node disease occurred in all patients. Median event-free survival had not been reached yet at the time of the analysis; at 48 months, 90 % of patients were event-free.
AEs were mild, with only 6 % of patients discontinuing treatment due to toxicity. At data cut-off, 86 % were still on therapy. Diarrhea, headache and upper respiratory infections emerged as the most common AEs. No patient discontinued acalabrutinib due to bleeding events, hypertension, or atrial fibrillation. The incidence of side effects generally decreased over time. According to the authors’ conclusion, these long-term data support the positive phase III results obtained with acalabrutinib in treatment-naïve patients with CLL.
CLL14: fixed-duration venetoclax/obinutuzumab
With the advent of new targeted agents, there is increasing desire for time-limited treatment options. The open-label, randomized, phase III CLL14 trial assessed a fixed-duration approach in previously untreated patients with CLL and coexisting medical conditions. They were randomized to either the orally available BCL-2 inhibitor venetoclax plus obinutuzumab for 6 cycles followed by venetoclax for another 6 cycles or chlorambucil plus obinutuzumab followed by chlorambucil for the same number of cycles. Eligibility criteria included Cumulative Illness Rating scale (CIRS) scores > 6 (indicating clinically relevant burden of coexisting conditions) and/ or creatinine clearance < 70 mL/min. In each arm, 216 patients were treated. A considerable fraction showed an unfavorable molecular setup including unmutated IGHV status (approximately 60 % in both arms) and deleted and/or mutated TP53 (14 % each). For PFS, which was defined as the primary endpoint, the primary analysis yielded a significant 65 % risk reduction with the venetoclax-based regimen (HR, 0.35; p < 0.001) [7].
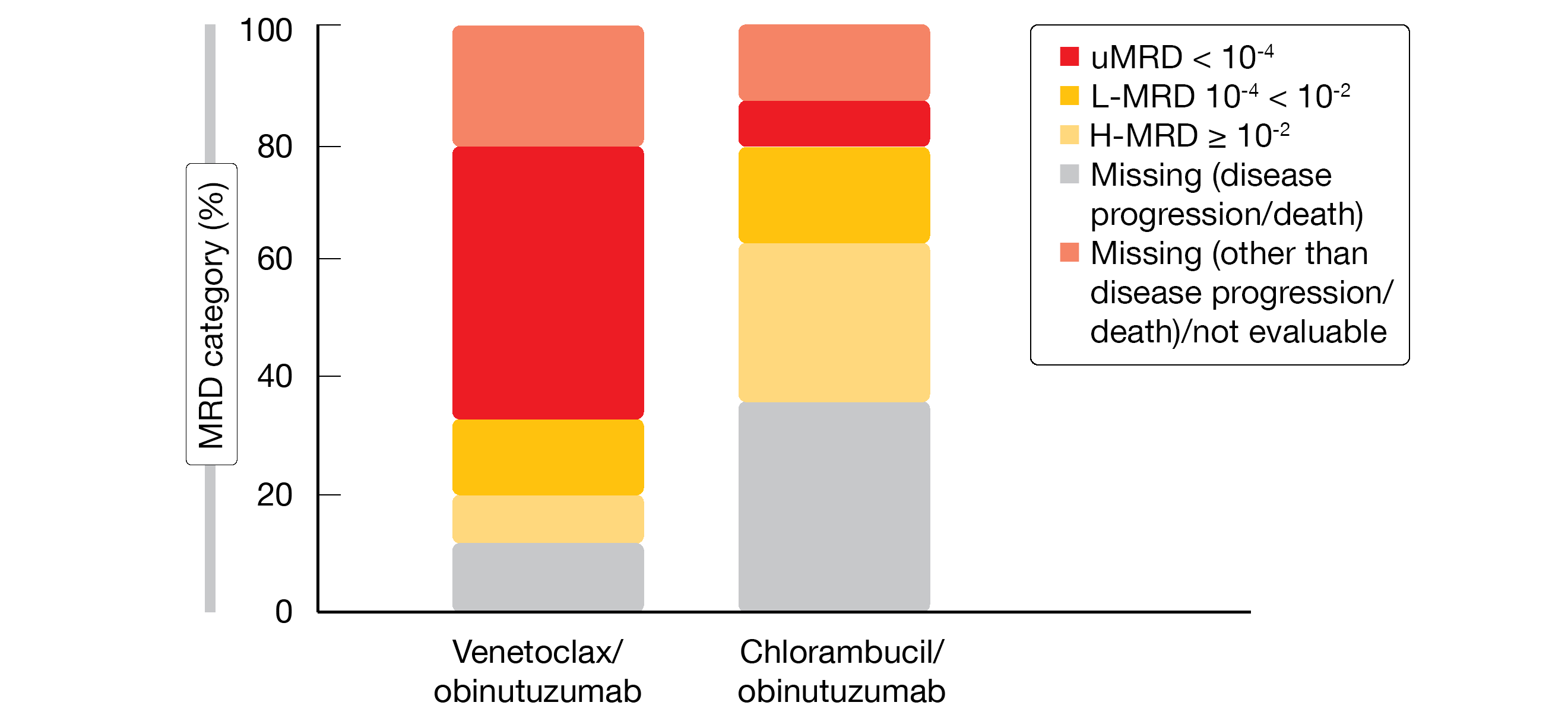
Al-Sawaf et al. reported updated results of the CLL14 study at the EHA Congress [8]. More than 2 years after treatment cessation, after a follow-up of 39.6 months, the reduction in the risk of progression and death in the experimental arm had risen to 69 % (not reached vs. 35.6 months; HR, 0.31; p < 0.0001). At 3 years, 81.9 % vs. 49.5 % of patients were progression-free. Venetoclax/obinutuzumab was superior to chlorambucil/obinutuzumab in the subgroup of patients with TP53 aberrations, although TP53 still remained a prognostic factor. Patients with both mutated and unmutated IGHV status definitely derived greater benefit from the venetoclax-based regimen than from the chlorambucil combination. Time to next treatment was considerably longer in the experimental arm, with 3-year rates of 84.5 % vs. 72.1 % (HR, 0.51), which implies long-term disease control. Correspondingly, approximately half of venetoclax-treated patients maintained undetectable minimal residual disease (uMRD) 18 months after treatment cessation (47.2 %), while this was only the case in 7.4 % in the chlorambucil arm (Figure 2).
With regard to safety, the analysis showed that AEs subsided after cessation of therapy. However, the post-treatment rate of second primary malignancies was higher in the experimental arm (6.4 % vs. 1.9 %). This difference was mainly driven by solid organ tumors, with no clear pattern of neoplasms occurring more often in venetoclax-treated patients. Further follow-up is warranted here as the clinical significance of this observation is currently unknown. The investigators noted that the updated results of the CLL14 study confirm the indication of venetoclax/obinutuzumab in patients with previously untreated CLL.
Figure 2: MRD results with venetoclax/obinutuzumab vs. chlorambucil/obinutzumab at 18 months in a population of untreated patients with comorbidities included in the CLL14 study
CAPTIVATE: MRD outcomes with ibrutinib/venetoclax
The multicenter, single-arm, phase II CAPTIVATE study evaluated the combination of ibrutinib and venetoclax to assess the depth of the MRD response in the first-line treatment of CLL/SLL. Two cohorts (MRD and Fixed Duration) have been implemented in this trial. The MRD Cohort received an ibrutinib lead-in followed by 12 cycles of ibrutinib plus venetoclax prior to restaging and MRD-guided randomization. Here, patients with confirmed uMRD were randomized to either ibrutinib or placebo, while those without confirmed uMRD received either ibrutinib alone or ibrutinib plus venetoclax. At the EHA Congress, Siddiqi et al. presented the pre-randomization results for the MRD Cohort (n = 164) [9]. Considerable proportions of patients had poor-risk features such as 17p deletion, complex karyotype and unmutated IGHV. Ninety percent completed all 12 cycles of ibrutinib/venetoclax.
Three cycles of ibrutinib lead-in already induced tumor debulking and reduced the risk of tumor lysis syndrome (TLS). Among patients with high baseline TLS risk, 90 % shifted to medium or low risk. Ibrutinib plus venetoclax gave rise to high rates of uMRD in both peripheral blood and bone marrow (75 % and 72 %, respectively). This was achieved irrespective of baseline disease risk characteristics such as the presence of 17p deletion or TP53 mutation. The proportion of patients with uMRD in the blood increased over time. ORR resulted in 97 % of patients, with CR or CR with incomplete bone marrow recovery (CRi) in 51 %. The group obtaining CR achieved uMRD in the blood and marrow in 85 % and 80 %, respectively.
AEs with ibrutinib/venetoclax were mostly grade 1/2 events. Among grade 3/4 events, neutropenia (35 %), hypertension (7 %), thrombocytopenia (5 %) and diarrhea (5 %) prevailed. No patient developed clinical TLS. The rates of grade 3 atrial fibrillation, major hemorrhage, infections, febrile neutropenia, and laboratory TLS were low. Only 5 % discontinued treatment due to AEs. At present, the post-randomization follow-up is ongoing, and PFS data will be reported at future meetings.
GIVe: response-adapted treatment for up to 15 cycles
In patients with CLL and high-risk genetic aberrations such as 17p deletion or TP53 mutation, clinical outcomes are still inferior, even with novel agents [10, 11]. Therefore, the multicenter, open-label phase II CLL2-GIVe trial is investigating the triple combination regimen of obinutuzumab, ibrutinib and venetoclax (GIVe) as first-line therapy in 41 patients with 17p deletion and/or TP53 mutation [12]. Patients received obinutuzumab for six 28-day cycles during the induction phase. Venetoclax was started on day 22 of cycle 1 and continued for up to 12 cycles throughout the induction and consolidation periods. The ibrutinib treatment covered the longest period, with 12 cycles of induction and consolidation followed by maintenance until cycle 36. However, if uMRD (< 10-4) was observed after both cycles 9 and 12 and CR/CRi was confirmed, the treatment was discontinued at cycle 15. Almost 90 % of patients belonged to the very high CLL-IPI risk group. The TLS risk was increased in 95 %.
In cycles 9 and 12, 87.8 % of patients had uMRD in the peripheral blood. At cycle 15, uMRD was present in the blood and bone marrow in 80.4 % and 68.3 %, respectively. The CR rate at cycle 15 was defined as the primary endpoint; here, 58.5 % of patients showed CR or CRi. PR was achieved in 34.2 %. Among patients with CR/CRi, 95.8 % had uMRD in the blood, with no MRD-positive case. The bone marrow analysis demonstrated uMRD in 87.5 % and MRD-positivity in 4.2 %. In comparison, the percentages of patients with uMRD were considerably lower in the group that only achieved PR.
Overall, the safety profile was acceptable. Grade ≥ 3 neutropenia occurred in 43.9 %, and grade ≥ 3 infections and infestations were observed in 19.5 %. One patient developed grade 4 cerebral aspergillosis. In their summary, the investigators emphasized that the obinutuzumab/ibrutinib/venetoclax triple combination is a promising first-line regimen for patients with high-risk CLL. The response rates are encouraging, although some high-grade infections have raised concerns.
Zanubrutinib, obinutuzumab & venetoclax
Another trial using a time-limited, MRD-driven chemotherapy-free triple regimen investigated the use of obinutuzumab, venetoclax and the second-generation BTK inhibitor zanubrutinib in the first-line setting [13]. Given its improved off-target tyrosine kinase inhibition profile [14, 15] and 100 % BTK occupancy in lymphoid tissue [16], zanubrutinib appears to be a favorable combination partner of anti-CD20 therapy and venetoclax. In the study, this regimen was discontinued if the prespecified uMRD endpoint was achieved after a minimum of 8 cycles and a maximum of 24 cycles. The treatment was discontinued if uMRD was obtained twice in the peripheral blood and once in the bone marrow. Among 39 patients included in the analysis, 72 % had high or very high risk according to CLL-IPI scoring. Unmutated IGHV status and TP53 aberrations were present in 72 % and 15 %, respectively.
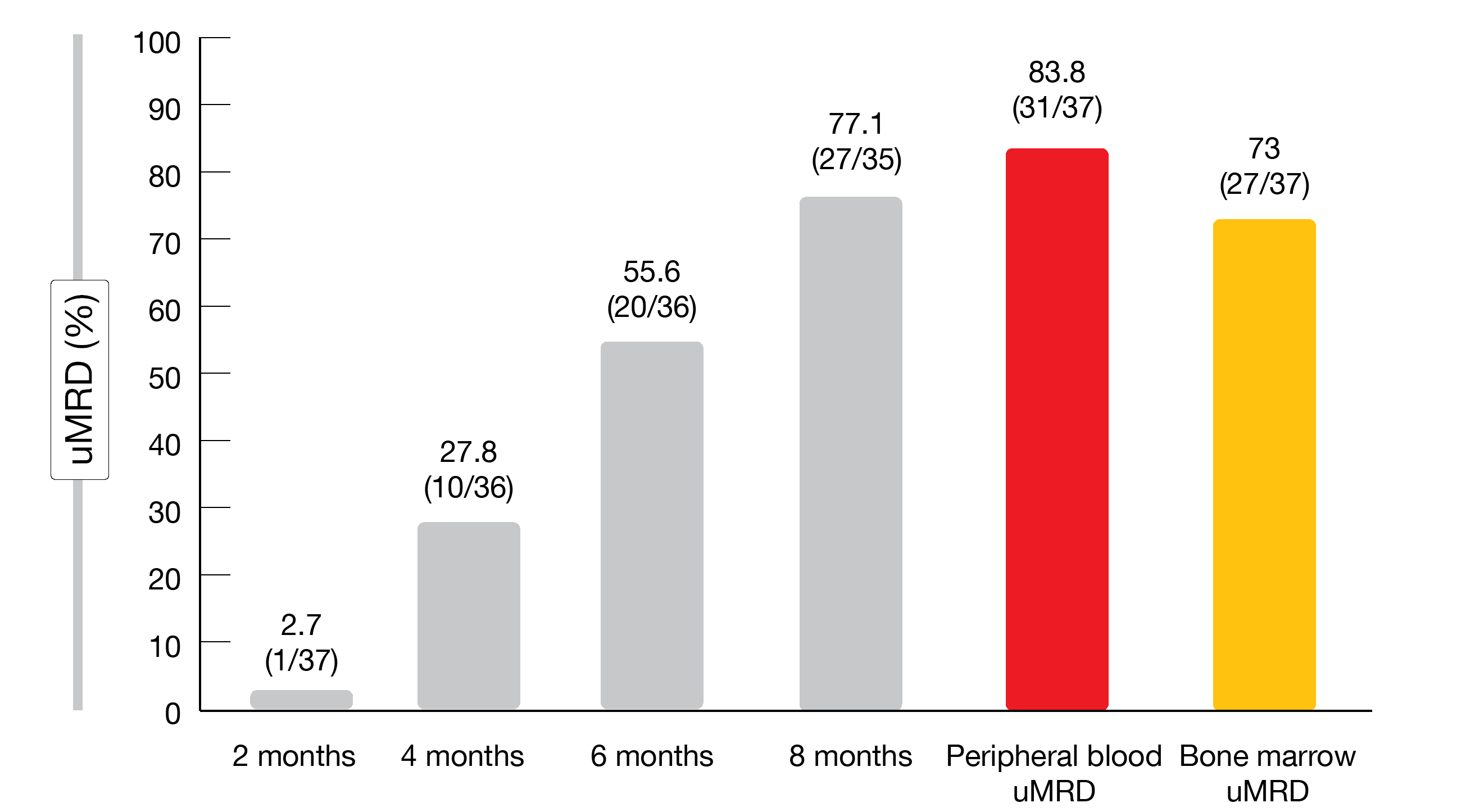
A 2-month zanubrutinib/obinutuzumab lead-in preceded the initiation of venetoclax and indeed prevented the occurrence of laboratory or clinical TLS. The uMRD rates increased rapidly over time (Figure 3). After a median follow-up of 11 months, a total of 83.8 % and 73 % of patients had achieved uMRD in blood and bone marrow, respectively. Sixty-two percent met the uMRD endpoint and stopped therapy at a median of 8 months. At treatment discontinuation, CR/CRi had been obtained in 57 %, and PR was present in 43 %.
The regimen proved well tolerable, with a low rate of grade 3/4 neutropenia of 15.4 %. Atrial fibrillation occurred in one patient who had a history of paroxysmal atrial fibrillation. The value of MRD-directed treatment duration will be evaluated with continued post-discontinuation follow-up.
Figure 3: Rapid emergence of undetectable MRD with the triple combination of zanubrutinib, obinutuzumab and venetoclax
Kinetics of response in r/r CLL
Based on the previously published phase II CLARITY trial that investigated ibrutinib plus venetoclax in patients with r/r CLL [17], Rawstron et al. investigated the impact of early MRD clearance on long-term outcomes [18]. Fifty patients after at least one previous therapy were included in the combination part of the study that started after a 2-month ibrutinib lead-in. The duration of the combined administration of ibrutinib and venetoclax was defined by sequential MRD assessments, with treatment being discontinued at certain timepoints once MRD eradication had been achieved. Twelve months was the minimum amount of time on combination treatment. Eventually, the trial was amended to allow for a third year of treatment after the administration of venetoclax had initially been limited to 24 months.
The CR rate improved steadily over time, from 40 % at month 8 to 62 % at month 26. ORRs were 100 % and 90 % at months 8 and 26, respectively. MRD eradication (< 0.01 % CLL cells) in the bone marrow after 12 months of combination therapy was defined as the primary endpoint. This was 40 % in all patients. Around month 24, the depth of response appeared to reach its peak and did not change substantially thereafter. The MRD levels in the peripheral blood at 8 months correlated with the marrow MRD response at 14 months.
The initial rate of disease depletion was shown to be highly predictive of longer-term response. Half of patients (n = 25) achieved > 2 log depletion in the first 2 months. In this group, the CR rates with MRD < 0.01 % at 14 and 26 months were markedly higher than in patients with < 2 log depletion. Overall, 23 patients stopped treatment because of achieving sustained MRD < 0.01 % in both blood and bone marrow. One year after treatment discontinuation, MRD levels remained undetectable or low in the majority of patients. Fifteen individuals continued to have MRD < 0.01 % in the blood, while 6 had 0.01 % to 1 % and only 2 showed levels > 1 %. These data demonstrated that patients with rapid disease clearance can experience prolonged remission and treatment-free periods. Those who did not achieve rapid depletion and had persistent MRD after 12 months of ibrutinib and venetoclax still showed stable or slowly decreasing disease levels.
Long-term results with venetoclax monotherapy
Single-agent venetoclax has demonstrated deep and durable responses in patients with r/r CLL, including those with 17p deletion [19, 20]. The phase IIIb VENICE-I trial is the largest multicenter study to evaluate the efficacy of venetoclax monotherapy in relapsed and refractory disease to date. Kater et al. reported efficacy and safety results at 48 weeks at the EHA Congress [21]. A total of 258 patients with and without 17p deletion or TP53 mutation were included in this trial. Previous B-cell receptor pathway inhibition (BCRi) was permitted.
VENICE-I met its primary endpoint. At week 48, the CR/CRi rate in the BCRi-naïve population (n = 191) was 35 %, with an ORR of 85 %. For the total group, these were 33 % and 80 %, respectively. Two-year PFS rates amounted to 79.4 % in the BCRi-naïve population (total group, 77.0 %). Patients who achieved a CR showed a higher 2-year PFS rate than those who obtained PR (95.0 % and 80.9 %, respectively). Moreover, PFS results were more favorable after only one prior treatment line (82.6 % at 2 years) than after ≥ 3 lines (68.9 %). Exploratory PFS was assessed for patients with prior ibrutinib failure; here, patients who had discontinued this treatment due to toxicity showed a considerably higher 2-year PFS rate than those who had experienced progression on ibrutinib therapy (76.2 % and 48.7 %, respectively).
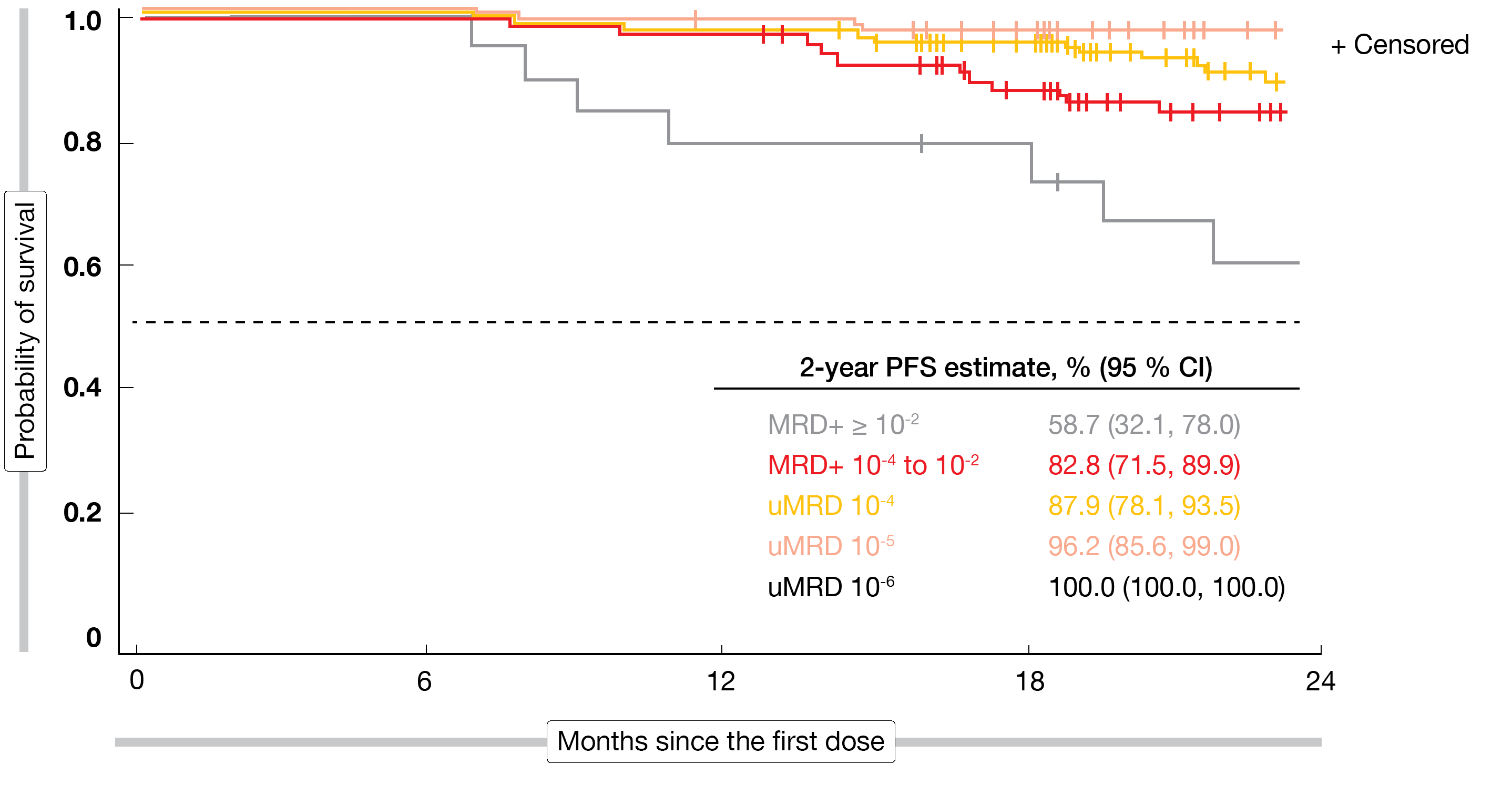
The rates and depth of MRD responses increased over time. uMRD < 10-4 plus < 10-5 was present in 25 % in the total population at week 24. Another 24 weeks later, 33 % had at least 10-4, with 5 % even showing < 10-6. Although the study was not powered to assess changes in MRD, the analyses indicated that deeper responses correlate with, and might be predictive of, longer PFS. In patients who achieved uMRD 10-6, the 2-year PFS rate was 100 %, while it was only 58.7 % in the group with MRD ≥ 10-2 (Figure 4). The study revealed no new safety signals. In 14.3 %, discontinuation was due to AEs. No clinical cases of TLS occurred. As the authors noted, VENICE-I confirms that venetoclax monotherapy can achieve deep and durable responses and has a tolerable and manageable safety profile in patients with r/r CLL.
Figure 4: Progression-free survival in patients with relapsed/refractory CLL who were treated with single-agent venetoclax according to their MRD status
REFERENCES
- Byrd JC et al., Targeting BTK with ibrutinib in relapsed chronic lymphocytic leukemia. J Engl J Med 2013; 369(1): 32-42
- Barf T et al., Acalabrutinib (ACP-196): a covalent bruton tyrosine kinase inhibitor with a differentiated selectivity and in vivo potency profile. J Pharmacol Exp Ther 2017; 363(2): 240-252
- Davids MS et al., Matching-adjusted indirect comparisons of efficacy and safety of acalabrutinib versus ibrutinib in treatment-naïve chronic lymphocyctic leukemia. EHA 2020, abstract EP724
- Ghia P et al., ASCEND: phase III, randomized trial of acalabrutinib versus idelalisib plus rituximab or bendamustine plus rituximab in relapsed or refractory chronic lymphocytic leukemia. J Clin Oncol 2020 May 27; JCO1903355
- Ghia P et al., Acalabrutinib vs. idelalisib plus rituximab or bendamustine plus rituximab in relapsed/refractory chronic lymphocyctic leukemia: ASCEND final results. EHA 2020, abstract S159
- Byrd JC et al., Acalabrutinib in treatment-naïve chronic lymphocyctic leukemia: mature results from phase 2 study demonstrating durable remissions and long-term tolerability. EHA 2020, abstract S163
- Fischer K et al., Venetoclax and obinutuzumab in patients with CLL and coexisting conditions. N Engl J Med 2019; 380: 2225-2236
- Al-Sawaf O et al., Fixed-duration venetoclax-obinutuzumab for previously untreated chronic lymphocyctic leukaemia: follow-up of efficacy and safety results from the multicenter, open-label, randomized phase 3 CLL14 trial. EHA 2020, abstract S155
- Siddiqi T et al., First-line ibrutinib + venetoclax for patients with chronic lymphocytic leukemia/small lymphocytic lymphoma: efficacy and safety results from CAPTIVATE MRD Cohort. EHA 2020, abstract S158
- Tausch E et al., Prognostic and predictive impact of genetic markers in patients with CLL treated with obinutuzumab and venetoclax. Blood 2020; blood.2019004492
- Woyach JA et al., Ibrutinib regimens versus chemoimmunotherapy in older patients with untreated CLL. N Engl J Med 2018; 379(26): 2517-2528
- Huber H et al., Preliminary safety and efficacy results from a phase-II study of obinutuzumab, ibrutinib and venetoclax (CLL2-GIVe) in untreated CLL with TP53 mutation and/or 17p deletion. EHA 2020, abstract S157
- Soumerai JD et al., Initial results of a multicenter, investigator-initiated study of MRD-driven, time-limited therapy with zanubrutinib, obinutuzumab, and venetoclax in patients with previously untreated CLL. EHA 2020, abstract S162
- Flinsenberg TWH et al., Differential effects of BTK inhibitors ibrutinib and zanubrutinib on NK-cell effector function in patients with mantle cell lymphoma. Haematologica 2020; 105(2): e76-e79
- Sharman JP et al., Acalabrutinib with or without obinutuzumab versus chlorambucil and obinutuzumab for treatment-naive chronic lymphocytic leukaemia (ELEVATE TN): a randomised, controlled, phase 3 trial. Lancet 2020; 395(10232): 1278-1291
- Tam CS et al., Phase 1 study of the selective BTK inhibitor zanubrutinib in B-cell malignancies and safety and efficacy evaluation in CLL. Blood 2019; 134(11): 851-859
- Hillmen P et al., Ibrutinib plus venetoclax in relapsed/refractory chronic lymphocytic leukemia: the CLARITY study. J Clin Oncol 2019; 37(30): 2722-2729
- Rawstron A et al., Kinetics of response in the peripheral blood predicts long term responses to ibrutinib + venetoclax treatment for relapsed/refractory CLL in the Bloodwise TAP CLARITY trial. EHA 2020, abstract S164
- Mato AR et al., A retrospective comparison of venetoclax alone or in combination with an anti-CD20 monoclonal antibody in R/R CLL. Blood Adv 2019; 3(10): 1568-1573
- Eyre TA et al., Efficacy of venetoclax monotherapy in patients with relapsed chronic lymphocytic leukaemia in the post-BCR inhibitor setting: a UK wide analysis. Br J Haematol 2019; 185(4): 656-669
- Kater AP et al., Efficacy of venetoclax in patients with relapsed/refractory chronic lymphocyctic leukemia: primary endpoint analysis of the international phase 3b trial (VENICE-I). EHA 2020, abstract S156
© 2019 Springer-Verlag GmbH, Impressum
More posts
New therapeutic options being currently investigated in advanced or metastatic colorectal cancer
Colorectal cancer (CRC) is the second leading cause of cancer death in the United States, and it is the fourth most frequent cancer diagnosis.A current treatment option for RAS and BRAF wild-type (WT) metastatic colorectal cancer (mCRC) is the chemotherapy doublet (FOLFOX/FOLFIRI) with an anti-EGFR monoclonal antibody (cetuximab or panitumumab).
An update and future directions in advanced gastric or gastrointestinal junction cancer (G/GEJC)
With more than 1 million newly diagnosed cases in 2020, gastric cancer (GC) is the fifth most frequent cancer; it was also the third leading cause of cancer-related death worldwide. Gastroesophageal junction (GEJ) cancer concerns a form of gastric cancer developing around the digestive tract where esophagus and stomach connect; in the last years, the prevalence of GEJ constantly increased.
Innovative combinations in esophageal squamous cell carcinoma
Each year, esophageal cancer (EC) is responsible for more than half a million deaths worldwide. Among them, esophageal squamous cell carcinoma (ESCC) accounts for the vast majority (~ 85 %) of EC incidences . At diagnosis, 70 % of ESCC is unresectable [3] and the 5-year survival rate is limited (30 % - 40 %). Patients with advanced or metastatic ESCC have a poor prognosis; their overall survival (OS) after standard first-line chemotherapy is limited to less than a year and other treatment options are scarce.
Novel agents or combinations in recurrent or metastatic nasopharyngeal cancer
Nasopharyngeal cancer (NPC) is a rare malignancy with an incidence of approximately 133,000 annually worldwide, resulting in about 80,000 deaths per year. Whereas early-stage and locally advanced NPC have a good prognosis, treatment of recurrent or metastatic nasopharyngeal cancer is a challenging; it is thus associated with a poor prognosis, especially in patients who have failed two or more lines of systemic therapy, with a median progression-free survival (mPFS) of seven months and median overall survival (mOS) of 22 months.
Preface ASCO Solid Tumor 2022
After 2 years of the COVID-19 pandemic, the Annual Meeting of the American Society of Clinical Oncology (ASCO), was held in Chicago, USA, and virtually from 3rd–7th June 2022.As always, the very much-anticipated event brought leading experts from across the globe together to learn and discuss the groundbreaking updates and scientific advancements which were covered in more than 2,000 abstracts, along with 85 livestream sessions, and more than 2,500 poster presentations.
More posts
Patient and disease characteristics in a small CAD cohort
A retrospective analysis hints at the wide range of cold agglutinin disease (CAD) clinical behavior. Koudouna et al. investigated the characteristics of 8 patients with CAD at the time of diagnosis [1]. Median age was 62 years, and 5 patients were women. Hematologic malignancies constituted 50 % of underlying medical conditions; in 37 %, hepatitis B/C was the associated disease, and in 13 %, autoimmune disorders.
Cold agglutinin disease: on the road to new insights and potential treatment options
Cold agglutinin disease (CAD) is a rare type of autoimmune hemolytic anemia (AIHA) elicited by cold-sensitive antibodies including cold agglutinins. Ninety percent of cold agglutinins belong to the IgM kappa category and bind to red blood cell surface antigens at temperatures of ≤ 37 °C, thus inducing hemolysis.
Paroxysmal nocturnal hemoglobinuria: improving outcomes with novel strategies
Paroxysmal nocturnal hemoglobinuria (PNH) is a rare, potentially life-threatening clonal hematopoietic stem cell disorder characterized by hemolytic anemia, bone marrow failure, thrombosis, and peripheral blood cytopenia. The disease results from an acquired loss-of-function mutation of the PIGA gene involved in the synthesis of the glycosylphosphatidylinositol-anchored complement inhibitors CD55 and CD59.
Targeted approaches in various B-cell malignancies
BTK inhibitors are active in many B-cell malignancies such as mantle cell lymphoma, CLL and Waldenström’s macroglobulinemia, but also in diffuse large B-cell lymphoma (DLBCL), follicular lymphoma (FL), and marginal zone lymphoma (MZL). Zanubrutinib is currently being assessed in pivotal phase II and III studies in all of these indications.
Changing paradigms in the management of mantle cell lymphoma
Mantle cell lymphoma (MCL) is a rare, heterogenous and generally aggressive subtype of B-cell non-Hodgkin lymphoma that remains incurable in the majority of cases. Median survival in non-trial patients has been estimated at 3 to 5 years. First-line therapy usually consists of chemoimmunotherapy, while both immunochemotherapy and targeted agents are recommended in relapsed disease.
Optimizing timing, efficacy and tolerability in chronic lymphocytic leukemia
In both treatment-naïve and relapsed/refractory patients with chronic lymphocytic leukemia (CLL) and small lymphocytic leukemia (SLL), inhibition of Bruton’s tyrosine kinase (BTK) represents a treatment standard as it has improved clinical outcomes. Compared to the first-generation agent ibrutinib, the second-generation, highly selective BTK inhibitor acalabrutinib shows minimal off-target kinase inhibition, thus potentially offering an optimized safety profile.