Innovative and established approaches for patients with uncommon mutations
CROWN: first-line use of ALK inhibitor lorlatinib
The highly potent, brain-penetrant, third-generation ALK tyrosine kinase inhibitor lorlatinib has been widely approved for the treatment of patients with ALK-positive advanced NSCLC who have previously received ALK TKIs. In the first-line setting, lorlatinib was compared with crizotinib in the randomized, phase III CROWN study that included almost 300 patients with stage IIIB/IV, ALK-positive NSCLC. Solomon et al. reported results from a planned interim analysis of the trial at ESMO 2020 [1].
Overall, 104 centers in 23 countries participated in the CROWN study. In the experimental arm, 149 patients received lorlatinib 100 mg daily, and in the control arm, 147 patients were treated with crizotinib 250 mg twice daily. Asymptomatic treated or untreated CNS metastases were permitted. Approximately 25 % of patients in each arm had brain lesions at baseline. No crossover between the study arms was allowed. PFS according to blinded independent central review constituted the primary endpoint.
After a median follow-up for PFS of 18.3 and 14.8 in the lorlatinib and crizotinib arms, respectively, lorlatinib gave rise to a 72 % reduction in the risk of progression and death (median PFS, not reached vs. 9.3 months; HR, 0.28; p < 0.001). At 12 months, 78 % vs. 39 % of patients were progression-free. All of the pre-specified subgroups benefited from the third-generation ALK inhibitor. Specifically, the HR for patients with brain metastases was 0.20, corresponding to an 80 % risk reduction.
Intracranial remissions and CNS protection
Moreover, lorlatinib treatment resulted in significant benefits regarding ORR (76 % vs. 58 %, odds ratio, 2.25) and median duration of response (not reached vs. 11.0 months). Intracranial response rates in patients with measurable or non-measurable brain metastases at baseline were higher in the experimental arm (66 % vs. 20 %; OR, 8.41). In patients with measurable brain lesions, the intracranial response rates even amounted to 82 % vs. 23 % (OR, 16.83), and a remarkable difference arose for intracranial complete remissions (71 % vs. 8 %). Time to CNS progression was significantly longer in the experimental arm (not reached vs. 16.6 months; HR, 0.07; p < 0.001; Figure 1), which translated into a risk reduction of 93 %. Notably, the curves showed a wide separation due to the paucity of intracranial progression events in the lorlatinib arm. These findings indicated the ability of lorlatinib not only to delay progression of existing brain lesions, but also to prevent the development of new ones.
Although OS data remained immature at the time of the interim analysis, the HR of 0.72 favored lorlatinib. The safety profile of lorlatinib resembled that reported in previous studies. Grade 3/4 AEs occurred more frequently with lorlatinib than with crizotinib; however, the majority of these were laboratory abnormalities that were asymptomatic and readily managed. No differences emerged across the arms in terms of AEs leading to permanent treatment discontinuation or temporary dose interruption.
According to patient-reported outcomes as assessed using the EORTC QLQ-C30 questionnaire, lorlatinib treatment led to significantly greater improvement in global quality of life from baseline (p < 0.01). This was seen early on and was maintained over the course of 18 cycles. As the authors noted in their summary, the results of the CROWN trial support the use of lorlatinib as a first-line therapy for patients with advanced ALK-positive NSCLC. No data on the activity of subsequent therapies after lorlatinib failure, particularly with respect to other ALK TKIs, were presented yet.
Figure 1: Time to CNS progression with lorlatinib vs. crizotinib as first-line treatment in patients with ALK-positive lung cancer
First-in-class KRAS inhibitor sotorasib
Despite the discovery of the KRAS oncogene almost 4 decades ago, no approved targeted therapy has been established thus far. The KRASG12C mutation is found in approximately 13 % of patients with NSCLC [2-4].
Sotorasib (AMG 510) is a novel, highly selective, first-in-class KRASG12C inhibitor that has demonstrated anticancer activity and a manageable safety profile in patients with KRASG12C-mutant solid tumors [4, 5]. The phase I, multicenter, open-label, dose-escalation, dose-expansion CodeBreaK100 trial assessed sotorasib in patients with KRASG12C-mutant, locally advanced or metastatic solid tumors after prior standard therapies. Sotorasib 960 mg orally daily was identified as the recommended phase II dose. Overall, 129 patients with 13 different tumor types participated, including 59 with NSCLC.
Hong et al. presented the data obtained for the NSCLC cohort at ESMO 2020 [6]. Within this group, 34 patients received the 960 mg dose. Prior anti-PD-(L)1 therapy had been administered in 82.4 % and 89.8 % of the 960 mg dose cohort and the total NSCLC group, respectively. All patients had received platinum-based chemotherapy, and 75 % had been treated with ≥ 2 prior systemic anticancer therapy lines.
Durable disease control
According to the investigators’ assessment, ORR was 35.3 % and 32.2 % for the 960 mg dose cohort and the total NSCLC group, respectively. Responses lasted for a median of 10.9 months, with 10 of the 19 responders still responding at the time of data cutoff. Disease control was obtained by 91.2 % and 88.1 % of patients, respectively. Tumor reductions were seen across all dose levels. Median PFS amounted to 6.3 months in the total group. Sotorasib demonstrated clinical activity across a range of KRASG12C mutational allele frequencies, PD-L1 tissue expression levels, and plasma tumor mutational burden levels. Also, clinical activity was found irrespective of tissue co-mutational profiles (e.g., TP53, SMAD4, PTEN/PIK3CA, KEAP1, EGFR).
Most AEs reported in the study were mild or moderate, with grade 3/4 treatment-related AEs showing an incidence of 18.6 %. Only one patient discontinued treatment due to a grade 3 AE, which was transaminase elevation. No dose-limiting toxicities and no treatment-related fatal AEs occurred. The most commonly observed any-grade AEs included diarrhea, transaminase elevations, fatigue, and nausea. In their conclusions, the authors emphasized that sotorasib showed a favorable safety profile and demonstrated durable disease control in this heavily pretreated patient population. Additional trials of the CodeBreaK program evaluating sotorasib as monotherapy or in combination with other anticancer agents are underway.
Afatinib in uncommon EGFR mutations
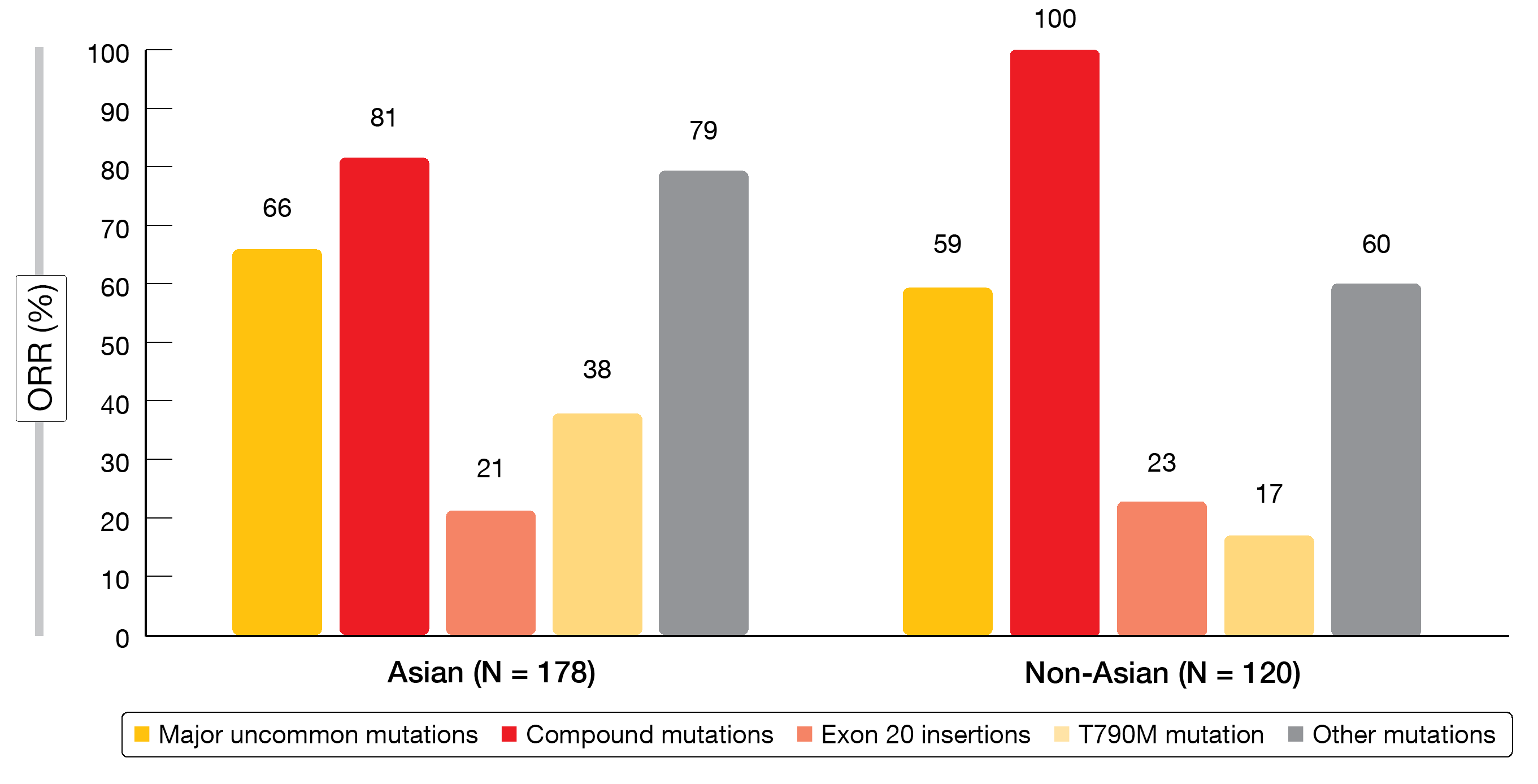
Uncommon EGFR mutations account for 7 % to 23 % of EGFR-mutated NSCLC cases and affect sensitivity to EGFR TKI treatment [7]. The irreversible ErbB receptor family blocker afatinib has shown clinical activity against major uncommon mutations including G719X, L861Q, and S768I [8]. However, there are few clinical data regarding the efficacy of EGFR TKIs against other uncommon EGFR mutations, and knowledge of ethnic differences in prevalence and outcomes is lacking. A pooled sub-analysis of the afatinib uncommon mutations database therefore assessed the efficacy of afatinib in the treatment of Asian (n = 178) and non-Asian (n = 120) patients with EGFR-mutated NSCLC harboring uncommon mutations [9]. Five mutation categories were defined: major uncommon (G719X, L861Q, S768I), compound, exon 20 insertions, T790M, and other mutations.
Asian patients were shown to have a high proportion of major uncommon mutations (61.8 %), for which afatinib has been approved. In the non-Asian group, exon 20 insertions ranked first (39.2 %), followed by major uncommon mutations (35.0 %). Afatinib showed activity in both Asian and non-Asian patients, with ORRs ranging from 17 % to 100 % (Figure 2). A certain percentage of patients with exon 20 insertions responded, which reflects the heterogeneity of this subgroup.
According to the assessment of duration of response and time to treatment failure, clinical activity was durable, particularly in patients with major uncommon, compound or other uncommon mutations. The authors concluded that afatinib should be considered as a first-line option in Asian and non-Asian patients with major uncommon and compound EGFR mutations.
Figure 2: Objective response rates observed with afatinib in Asian and non-Asian patients harboring uncommon EGFR mutations
Treatment patterns of patients with NRG1 fusions
NRG1 gene fusions are present in 0.2 % of all solid tumors, although their prevalence is higher in certain types such as invasive mucinous lung adenocarcinomas, where it has been estimated at 31 % [10, 11]. No approved therapies for patients with NRG1 gene fusion-positive tumors are available yet. However, several reports suggest durable responses to treatment with afatinib [12-14], which might therefore represent a novel therapeutic option in patients with NRG1-positive cancer.
A retrospective, real-world feasibility study identified US-based patients with solid tumors harboring NRG1 gene fusions who had received afatinib in any line of treatment or other systemic therapies without prior afatinib [15]. The objectives of the study comprised the assessment of the number of patients with NRG1 gene fusion-positive solid tumors available for analyses and gaining insights into treatment patterns and testing. A total of 108 patients were identified 67 of whom had received afatinib.
The data support previous findings that NRG1 gene fusions are detected across multiple tumor types [10]. In both the afatinib-treated group and the group receiving other systemic therapies, NSCLC constituted the most common tumor type (40 % and 56 %, respectively), followed by gastrointestinal cancers and breast cancer. The most common NRG1 gene fusion partners were SDC4, CD74, and ATP1B1. mRNA sequencing constituted the most commonly used testing methodology. Afatinib was mainly prescribed in the second line, while other treatments dominated the first-line setting.
According to the authors, these findings provide a rationale for a larger, retrospective, chart-based cohort study evaluating treatment outcomes. The use of afatinib in NRG1-positive tumors is under investigation in ongoing prospective studies including the Drug Rediscovery Protocol trial (DRUP; NCT02925234) and the Targeted Agent and Profiling Utilization Registry study (TAPUR; NCT02693535).
REFERENCES
- Solomon B et al., Lorlatinib vs crizotinib in the first-line treatment of patients with advanced ALK-positive non-small cell lung cancer: results of the phase 3 CROWN study. ESMO 2020, LBA2
- Biernacka A et al., The potential utility of re-mining results of somatic mutation testing: KRAS status in lung adenocarcinoma. Cancer Genet 2016; 209(5): 195-198
- Wiesweg M et al., Impact of RAS mutation subtype on clinical outcome-a cross-entity comparison of patients with advanced non-small cell lung cancer and colorectal cancer. Oncogene 2019; 38(16): 2953-2966
- Canon J et al., The clinical KRAS(G12C) inhibitor AMG 510 drives anti-tumour immunity. Nature 2019; 575(7781): 217-223
- Govindan R et al., Phase I study of AMG 510, a novel molecule targeting KRAS G12C mutant solid tumors. Ann Oncol 2019; 30(suppl_5): v159-v193
- Hong DS et al., Durability of clinical benefit and biomarkers in patients with advanced non-small cell lung cancer treated with AMG 510 (sotorasib): CodeBreaK 100. ESMO 2020, 12570
- Kohsaka S et al., A method of high-throughput functional evaluation of EGFR gene variants of unknown significance in cancer. Sci Transl Med 2017; 9(416): eaan6566
- Yang JCH et al., Clinical activity of afatinib in patients with advanced non-small-cell lung cancer harbouring uncommon EGFR mutations: a combined post-hoc analysis of LUX-Lung 2, LUX-Lung 3, and LUX-Lung 6. Lancet Oncol 2015; 16(7): 830-838
- Yang JCH et al., Afatinib in Asian and non-Asian patients with EGFR mutation-positive NSCLC harboring uncommon mutations. ESMO 2020, 1341P
- Jonna S et al., Detection of NRG1 gene fusions in solid tumors. Clin Cancer Res 2019; 25(16): 4966-4972
- Trombetta D et al., Frequent NRG1 fusions in Caucasian pulmonary mucinous adenocarcinoma predicted by Phospho-ErbB3 expression. Oncotarget 2018; 9(11): 9661-9671
- Gay ND et al., Durable response to afatinib in lung adenocarcinoma harboring NRG1 gene fusions. J Thorac Oncol 2017; 12(8): e107-e110
- Jones MR et al., Successful targeting of the NRG1 pathway indicates novel treatment strategy for metastatic cancer. Ann Oncol 2017; 28(12): 3092-3097
- Cadranel J al., Therapeutic potential of afatinib in NRG1 fusion-driven solid tumors: a case series. Oncologist 2020 Aug 27. DOI: 10.1634/theoncologist.2020-0379
- Gajra A et al., A real-world feasibility study of patients with solid tumours harbouring NRG1 fusions. ESMO 2020, 1956P
© 2020 Springer-Verlag GmbH, Impressum
More posts
Interview – Malignant mesothelioma: implementation of immunotherapy-based standards
Which outcomes can be expected in European patients with malignant mesothelioma who receive the current standard treatment?
EGFR-targeted options in a changing treatment landscape
The combination of amivantamab, a bispecific antibody that targets both EGFR and MET, and the potent third-generation EGFR TKI lazertinib is being explored in patients with advanced NSCLC. Amivantamab has demonstrated clinical activity across various types of EGFR-mutant NSCLC harboring both activating and resistance mutations and was granted FDA Breakthrough Therapy Designation for EGFR-mutant NSCLC with exon 20 insertion after progression on platinum-based chemotherapy.
Innovative and established approaches for patients with uncommon mutations
The highly potent, brain-penetrant, third-generation ALK tyrosine kinase inhibitor lorlatinib has been widely approved for the treatment of patients with ALK-positive advanced NSCLC who have previously received ALK TKIs. In the first-line setting, lorlatinib was compared with crizotinib in the randomized, phase III CROWN study that included almost 300 patients with stage IIIB/IV, ALK-positive NSCLC. Solomon et al.
Interview – Exploring interactions between radiotherapy and the immune system
The modulation of molecular pathways that determine the patient response to radiotherapy might contribute to improving patient outcomes. What insights have been gained to date in this field of research that may be relevant in the years to come?
Determinants of treatment success in small-cell lung cancer
The randomized, controlled, open-label, phase III CASPIAN trial has assessed first-line treatment with durvalumab ± tremelimumab plus platinum/etoposide (EP) compared to EP alone in patients with extensive-stage small-cell lung cancer (ES-SCLC). Durvalumab plus EP significantly improved OS compared to EP alone (HR, 0.73; p = 0.0047).
Early-stage lung cancer: noteworthy findings for different types of therapy
Postoperative radiotherapy (PORT) in patients with completely resected NSCLC has been a subject of debate for years. In the absence of robust data confirming the benefit of this intervention, its feasibility was additionally challenged by a multitude of changes that have taken place over the last two decades with respect to patient selection, (neo)adjuvant chemotherapy, surgery, and radiotherapy.