Innovations in the setting of rare mutations: ALK, ROS1, NTRK, NRG1
BFAST: blood-based NGS as a stand-alone test
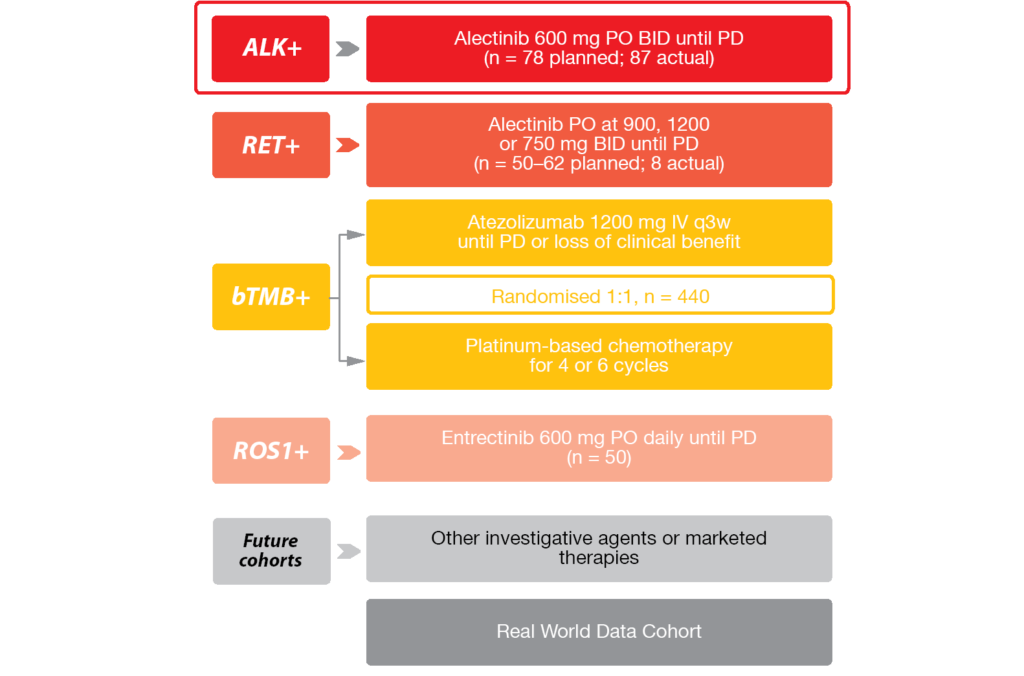
Oncogene-directed treatment requires molecular testing, but, as is known, limitations related to tissue collection and tissue-based testing can represent a serious obstacle in clinical practice. Blood-based next generation sequencing (NGS) has the potential to overcome some of these limitations. Therefore, the global, phase II/III, multi-cohort BFAST study was initiated with the aim of prospectively evaluating the relationship between blood-based biomarkers and the clinical activity of frontline targeted therapies or immunotherapy in advanced NSCLC. Patients were enrolled into specific treatment cohorts using only blood-based NGS testing, to establish its clinical utility as a stand-alone test. Biomarkers that were identified included ALK, RET, ROS1, and tumor mutational burden (Figure). BFAST is the first prospective trial to use blood-based NGS testing as the sole method of identifying actionable genetic alterations and assigning NSCLC patients to targeted agents or immunotherapy.
Figure: Cohorts treated in the BFAST trial. bTMB, blood tumor mutational burden
At ESMO 2019, Gadgeel et al. presented the results for the ALK-positive cohort [1]. For this group, it was intended to demonstrate consistency of activity of the ALK inhibitor alectinib with the results obtained in the global ALEX trial where tissue-based assessment had been used for patient selection [2]. Within the total screened population (n = 2,219), the 5.4 % prevalence of ALK translocation corresponded to the expected rate of 5 % [3]. Eighty-seven patients whose baseline characteristics resembled those of the population treated with alectinib in the ALEX trial entered the cohort and received alectinib 600 mg twice daily.
Confirmed ORR by investigator was defined as the primary endpoint. This was indeed achieved with ORRs of 87.4 % and 92.0 % according to investigator and independent review facility (IRF), respectively, that even exceeded the confirmed ORR of 72.4 % observed in ALEX [4]. Eleven patients (12.6 %) experienced complete remissions according to IRF. The presence of baseline CNS metastases did not affect response rates. In the group of confirmed responders, the event-free rate was 90.4 % at 6 months. Median PFS had not been reached yet at the time of the analysis, with the 12-month PFS rate being 78.38 %. The safety profile of alectinib was consistent with that established in previous phase III trials and post-marketing experience. Overall, these results demonstrated the clinical utility of blood-based NGS as a method to inform clinical decision making in patients with ALK-positive NSCLC.
Final PFS data from the ALEX trial
The global, randomized, phase III ALEX study was conducted to compare the efficacy and safety of alectinib versus crizotinib in 303 patients with treatment-naïve, ALK-positive advanced NSCLC. Mok et al. reported mature PFS and updated OS data after a median follow-up of 37.8 months with alectinib and 23.0 months with crizotinib [4].
According to this analysis, median investigator-assessed PFS was 34.8 and 10.9 months for alectinib and crizotinib, respectively (HR, 0.43; p < 0.0001). The PFS benefit was consistent in patients with and without baseline CNS metastases (HRs, 0.37 and 0.46, respectively). In the alectinib group, patients showed higher event-free rates with respect to PFS regardless of the presence or absence of baseline CNS metastases. At 4 years, 43.7 % of the entire alectinib-treated cohort were event-free, while this applied to none of the patients in the crizotinib arm.
Overall survival data remained immature; here, 4-year rates were 64.5 % and 52.2 % for alectinib and crizotinib, respectively. The confirmed ORRs of 72.4 % vs. 60.9 % remained consistent with the results from the primary data cut-off [2]. Considering the difference in median treatment duration (27.7 vs. 10.8 months), alectinib continued to show a favorable safety profile compared with crizotinib. The authors concluded that this analysis confirms the superior efficacy of alectinib in untreated ALK-positive NSCLC patients.
Entrectinib in NTRK-positive disease
Neurotrophic tropomyosin receptor kinase (NTRK) gene fusions act as oncogenic drivers and occur in approximately 0.3 % of solid tumors, among them lung cancer [5]. The TRKA/B/C, ROS1 and ALK inhibitor entrectinib has been designed to work in tumors with NTRK gene fusions, both at a systemic level and inside the CNS. The accelerated FDA approval of entrectinib for the treatment of adult patients with solid tumors that harbor NTRK gene fusions was based on the results from an integrated analysis of three phase I/II trials conducted at more than 150 sites in 15 countries: ALKA-372-001, STARTRK-1, and STARTRK-2 [6]. All of these studies included patients with NTRK-, ROS1– or ALK-positive tumors.
At ESMO 2019, Rolfo et al. presented updated data from the integrated analysis after an additional follow-up of 5 months [7]. The efficacy-evaluable population included 54 patients with advanced NTRK-positive solid tumors, among them 10 individuals with lung cancer. At the time of the analysis, ORR remained high at 59.3 %, with four complete remissions (7.4 %). Responses lasted for a median of 12.9 months. Median PFS and OS were 11.8 and 23.9 months, respectively. Systemic response rates did not differ according to baseline CNS disease status. The authors noted that both OS and duration of response were longer than reported in the previous analysis [6].
In patients who had baseline brain metastases, entrectinib gave rise to clinically meaningful and durable benefits. Intracranial ORR and PFS were 54.5 % and 14.3 months, respectively, and median intracranial response had not been reached yet.
The ROS1-rearranged treatment group
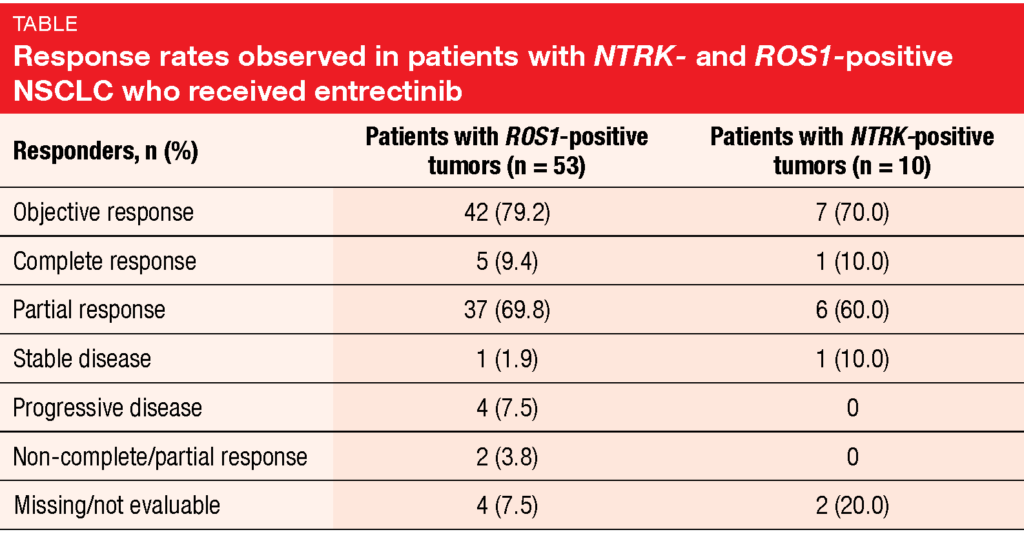
De Braud et al. reported updated results from the integrated analysis of the ALKA-372-001, STARTRK-1 and STARTRK-2 trials that focused on lung cancer patients [8]. Out of 63 individuals, 53 and 10 had ROS1-positive and NTRK-positive lung cancer, respectively. ORRs were 79.2 % and 70.0 %, respectively, for these two cohorts (Table). Complete responses occurred in approximately 10 % in each group. In the ROS1-positive population, median duration of response was 24.6 months; median PFS amounted to 19.0 months, and median OS had not been reached yet.
Clinically meaningful and durable responses were observed in ROS1-positive patients with brain metastases (ORR, 73.9 %; median duration of response, not estimable) as well as those without (ORR, 83.3 %; median duration of response, 24.6 months). The cohort with baseline CNS lesions responded intracranially in 55 %. Here, responses lasted for a median of 12.9 months, and intracranial PFS was 7.7 months. Both analyses of STARTRK-2, STARTRK- and ALK-372-001 revealed good tolerability of entrectinib, with a safety profile consistent with that previously reported.
NRG1 fusion: clinical experience with afatinib
Various types of tumors harbor actionable NRG1 gene fusions that were shown to increase cell proliferation through ErbB signaling and may function as oncogenic drivers [9–11].
The estimated overall prevalence of these fusions across solid tumors is approximately 0.2 % but was reported to be up to 31 % in invasive mucinous adenocarcinoma of the lung [11, 12]. Based on the involvement of ErbB signaling pathways in NRG1-positive tumors, the pan-ErbB family blocker afatinib is a potential treatment option in these patients. Liu et al. presented seven case reports including four lung cancer cases that support this assumption [13]. Three patients were treated for non-mucinous adenocarcinoma of the lung, and one for invasive mucinous adenocarcinoma. All of them had had several treatment lines before being prescribed afatinib, with one patient even receiving 14 lines of previous therapy.
Partial responses were achieved as best overall responses in three individuals and proved durable (duration of best response, 18 to 24 months), with treatment ongoing in two cases. One patient obtained stable disease that lasted 4 months. According to the conclusion of the authors, these findings add to the growing body of evidence showing that afatinib is a potential treatment option for patients with NRG1-fusion–positive tumors, particularly in the absence of other driver aberrations. Molecular testing would be of particular importance in invasive mucinous adenocarcinoma of the lung where NRG1 fusion prevalence is relatively high.
REFERENCES
- Gadgeel SM et al., Phase II/III blood-first assay screening trial (BFAST) in treatment-naïve NSCLC: initial results from the ALK+ cohort. ESMO 2019, abstract LBA81_PR
- Peters S et al., Alectinib versus crizotinib in untreated ALK-positive non-small-cell lung cancer. N Engl J Med 2017; 377(9): 829-838
- Dearden S et al., Mutation incidence and coincidence in non small-cell lung cancer: meta-analyses by ethnicity and histology (mutMap). Ann Oncol 2013; 24(9): 2371-2376
- Mok T et al., Final PFS, updated OS and safety data from the randomized, phase III ALEX study of alectinib versus crizotinib in untreated advanced ALK+ NSCLC. ESMO 2019, abstract 1484PD
- Okamura R et al., Analysis of NTRK alterations in pan-cancer adult and pediatric malignancies: implications for NTRK-targeted therapeutics. JCO Precis Oncol 2018; 2018. doi: 10.1200/PO.18.00183. Epub 2018 Nov 15.
- https://www.roche.com/media/releases/med-cor-2019-08-16.htm
- Rolfo C et al., Updated efficacy and safety of entrectinib in patients with NTRK fusion-positive tumours: integrated analysis of STARTRK-2, STARTRK-1 and ALKA-372-001. ESMO 2019, abstract 476P
- De Braud F et al., Entrectinib in locally advanced/metastatic ROS1 and NTRK fusion-positive non-small cell lung cancer: updated integrated analysis of STARTRK-2, STARTRK- and ALK-372-00. ESMO 2019, abstract 1488PD
- Fernandez-Cuesta L & Thomas RK, Molecular pathways: targeting NRG1 fusions in lung cancer. Clin Cancer Res 2014; 21(9): 1989-1994
- Duruisseaux M et al., NRG1 fusion-positive lung cancers: clinicopathologic profile and treatment outcomes from a global multicenter registry. J Clin Oncol 37, 2019 (suppl; abstr 9081)
- Jonna S et al., Detection of NRG1 gene fusions in solid tumors. Clin Cancer Res 2019; 25(16): 4966-4972
- Trombetta D et al., Frequent NRG1 fusions in Caucasian pulmonary mucinous adenocarcinoma predicted by Phospho-ErbB3 expression. Oncotarget 2018; 9(11): 9661-9671
- Liu SV et al., Targeting NRG1 fusions in multiple tumour types: afatinib as a novel potential treatment option. ESMO 2019, abstract 1969P
More posts
Even infrequent actionable drivers are important
NRG1 fusions are oncogenic events, i.e., transforming events that occur in all tumor types, although in fairly low frequencies. Their prevalence is less than 1 % throughout all tumor types. Some reports have estimated the NRG1 fusion prevalence at approximately 0.2 %. Although NRG1 fusions are not a common event, they represent an important actionable driver.
CNS disease does not preclude successful treatment
Brain metastases occur in approximately 35 % of patients with metastatic NSCLC and are associated with a variety of neurological symptoms, as well as poor prognosis. However, little is known about the prognostic impact of the symptomatic burden of CNS lesions at the time of diagnosis. This was assessed by an analysis based on a real-life cohort of 1,608 NSCLC patients from the Vienna Brain Metastasis Registry with newly diagnosed brain metastases.
Innovations in the setting of rare mutations: ALK, ROS1, NTRK, NRG1
Oncogene-directed treatment requires molecular testing, but, as is known, limitations related to tissue collection and tissue-based testing can represent a serious obstacle in clinical practice. Blood-based next generation sequencing (NGS) has the potential to overcome some of these limitations.
Exploring synergy between anti-angiogenic drugs and immunotherapy
In the setting of non-squamous advanced NSCLC without actionable driver mutations, the advent of immune checkpoint inhibitor therapy has led to the implementation of new standards. Synergistic effects can be expected from anti-angiogenic treatment. The vascular endothelial growth factor (VEGF) has been shown to create an immunosuppressive tumor microenvironment by modifying immune cell function besides promoting angiogenesis.
Frontline combinations of EGFR- and angiogenesis-targeted agents
In patients with untreated EGFR-mutant tumors, it has been shown that the addition of the anti-VEGF antibody bevacizumab to first-generation EGFR TKIs induces PFS benefits with an acceptable toxicity profile. The open-label, randomized, multicenter, phase III ARTEMIS (CTONG 1509) study is the first phase III trial to test bevacizumab plus erlotinib in Chinese NSCLC patients.
EGFR-positive tumors: the issue of optimal therapy across several lines
In patients with advanced, EGFR-positive NSCLC, EGFR tyrosine kinase inhibitors (TKIs) represent the frontline treatment standard. Three generations of TKIs are widely available, but the ideal sequence is currently unknown. The phase III, double-blind, randomized FLAURA trial compared the third-generation EGFR TKI osimertinib with the first-generation agents gefitinib and erlotinib in the frontline setting.