Rare mutations: HER2, RET, ALK, BRAF
DESTINY-Lung01: trastuzumab deruxtecan
Trastuzumab deruxtecan (T-DXd) is a novel antibody-drug conjugate containing a humanized anti-HER2 monoclonal antibody linked to a topoisomerase I inhibitor exatecan derivative. The open-label, multicenter, phase II DESTINY-Lung01 study tested T-DXd 6.4 mg/kg 3-weekly in patients with relapsed or refractory advanced NSCLC that expressed HER2 (Cohort 1; n = 42) or carried HER2-activating mutations (Cohort 2; n = 42). At the ASCO Congress, Smit et al. reported the interim results for Cohort 2 [1]. In terms of confirmed ORR according to independent central review, which was the primary endpoint, T-DXd demonstrated pronounced clinical activity. Almost 62 % of patients responded, and 2.4 % achieved complete remissions (Table). At the time of the analysis, the median duration of response had not been reached yet; this also applied to OS. Median PFS was 14.0 months.
The safety profile observed in this HER2-mutated cohort was generally consistent with previously reports. Nausea, alopecia, anemia, neutropenia and decreased appetite represented the most common treatment-emergent AEs. Fatigue and nausea primarily prompted dose reductions, while dose interruptions were predominantly due to neutropenia (19.0 %) and lung infection (7.1 %). Five patients developed grade-2 interstitial lung disease (ILD). The authors noted that ILD remains a concern and requires careful monitoring and management. Overall, these data show the potential of T-DXd as a new treatment option in the setting of HER2-mutated NSCLC. Meanwhile, enrolment in the HER2-mutated cohort has been expanded to better characterize the risk-benefit ratio of T-DXd.
CNS effects of selpercatinib in RET-positive lung cancer
RET fusions have been identified in approximately 2 % of NSCLC patients [2, 3]. The ongoing registrational, international, phase I/II LIBRETTO-001 trial is assessing the efficacy of the selective, CNS-active RET inhibitor selpercatinib (LOXO-292). LIBRETTO-001 is being conducted in patients with advanced RET-fusion–positive solid tumors; 253 of these have NSCLC. In the primary analysis set, the ORR was 68 %, and responses lasted for a median of 20.3 months [4].
Data from the NSCLC CNS population with measurable CNS disease (n = 22) presented by Subbiah et al. shed more light on the intracranial activity of selpercatinib [5]. Overall, the CNS ORR was 81.8 %, with CRs occurring in 22.7 %. Patients without prior irradiation to the brain fared somewhat better than those who had received radiotherapy (ORR, 85.7 % vs. 75.0 %; CR, 28.6 % vs. 12. 5%). Both patients with and without prior anti-PD-(L)1 treatment responded intracranially; also, this was not affected by the prior use of multi-targeted kinase inhibitor therapy. Median duration of CNS response was 9.4 in the total group. The authors concluded that selpercatinib shows marked and durable intracranial anti-tumor activity in patients with RET-fusion–positive NSCLC and CNS metastases. A randomized, global, phase III study of selpercatinib versus platinum-based chemotherapy with or without pembrolizumab in treatment-naïve RET-fusion–positive NSCLC including patients with asymptomatic brain metastases is ongoing.
RET kinase inhibitor pralisetinib
Another investigational, selective RET kinase inhibitor that is currently being developed is pralisetinib (BLU-667). The ongoing pivotal, global, phase I/II ARROW trial is testing pralisetinib in patients with advanced solid, RET-altered tumors including RET-fusion–positive NSCLC. Gainor et al. reported data for the intent-to-treat (ITT) efficacy population of 132 NSCLC patients that included 116 response-evaluable individuals [6]. In the ITT population, 92 and 29 patients had received prior platinum and were treatment-naïve, respectively.
Pralsetinib gave rise to rapid and durable responses. According to blinded independent centralized review, the ORR in the response-evaluable group was 65 %, with 6 % obtaining CRs. Disease control resulted in 93 %. The median duration of response had not been reached yet. All treatment-naïve patients in the evaluable cohort achieved tumor reductions, and 12 % experienced CRs. Furthermore, pralisetinib showed robust activity in the CNS, with intracranial ORR and CR rates of 56 % and 33 %, respectively.
The RET inhibitor therapy was well tolerated. Treatment-related AEs included mainly transaminase elevations, cytopenia, constipation and hypertension, and were predominantly grades 1 and 2. In their summary, the authors emphasized that pralisetinib has the potential to change the standard of care for patients with RET-fusion–positive NSCLC.
Survival update of ALEX
Previous analyses of the global, randomized, phase III ALEX study have established the superiority of alectinib over crizotinib in patients with untreated, advanced ALK-positive NSCLC. The mature PFS data confirmed significant improvement for this endpoint (34.8 vs. 10.9 months) [7], while the OS results remained immature. After a further 12 months of follow-up, Peters et al. presented updated OS and other outcomes [8].
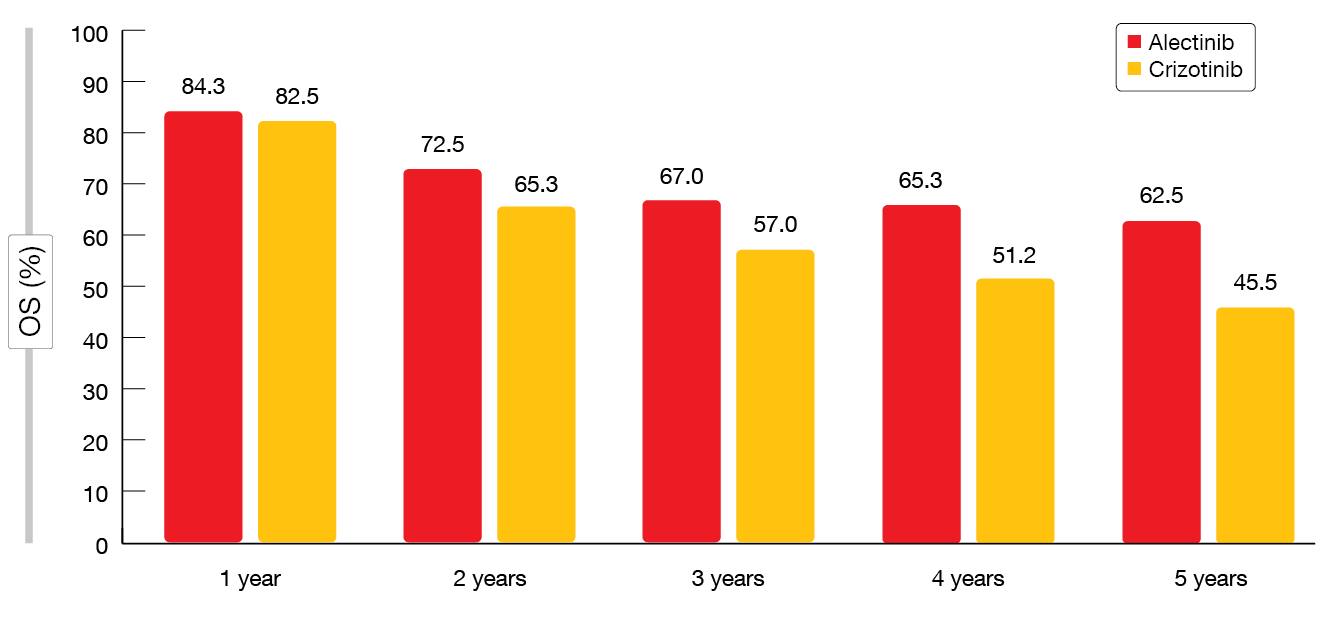
The OS data were still immature at that time, with five-year OS rates of 62.5 % vs. 45.5 % for alectinib and crizotinib, respectively (HR, 0.67; p = 0.0376; Figure). Among patients who experienced disease progression, subsequent therapy was administered in more than 60 % in both arms. Follow-up treatment with other ALK TKIs was prescribed to 38.1 % and 53.5 % of alectinib- and crizotinib-treated patients with progressive disease. No new safety signals occurred after almost three-times longer median treatment duration with alectinib (28.1 months) than crizotinib (10.8 months). The investigators concluded that ALEX is the first global, randomized trial of a next-generation ALK TKI to demonstrate a clinically meaningful OS improvement compared to crizotinib in treatment-naïve, advanced ALK-positive NSCLC.
Figure: Overall survival rates obtained in the ALEX study with alectinib and crizotinib over time
Impact of biomarkers in ALTA-1L
The open-label, randomized, multicenter, phase III ALTA-1L study evaluated brigatinib in patients with ALK-TKI–naïve, ALK-positive advanced NSCLC, demonstrating superior efficacy compared to crizotinib with acceptable tolerability at the second interim analysis [9]. Camidge et al. evaluated the impact of EML4-ALK fusion variants and other baseline variables on the activity of brigatinib vs. crizotinib in the ALTA-1L trial [10].
Brigatinib was superior to crizotinib with respect to ORR and PFS regardless of EML4-ALK fusion variant or TP53 mutation status. EML4-ALK fusion variant 3 (V3) appeared to be prognostic, as patients with this variant had worse PFS than those harboring V1 or V2, irrespective of treatment. Brigatinib demonstrated superior PFS particularly in this poor-prognosis group of patients with V3 (HR, 0.30). Also, there was a trend indicating that TP53 mutations are an independent biomarker of poor prognosis which persisted in multivariate analyses and warrants further investigation in a larger sample size. Defining higher-risk ALK-positive advanced NSCLC might impact future clinical trial designs and treatment options.
BRAFV600E-mutant disease: dabrafenib & trametinib
Dabrafenib as a monotherapy and combined with trametinib was assessed in a non-randomized, multicenter, open-label phase II trial in patients with BRAFV600E-mutant metastatic NSCLC. The primary analysis has revealed robust clinical activity for dabrafenib plus trametinib with a manageable safety profile [11]. At the ASCO Congress, Planchard et al. presented the updated OS and genomic analysis data for the combination therapy population in Cohorts B (pretreated patients) and C (treatment-naïve patients) [12].
Dabrafenib plus trametinib provided combined CR and PR rates of 68.4 % and 63.9 % in Cohorts B and C, respectively. Reponses lasted for 9.8 and 10.2 months, respectively. OS was 18.2 months in Cohort B and 17.3 months in Cohort 3. The genomic analysis suggested that co-occurring genetic alterations influence clinical outcomes, as patients with PI3K pathway alterations showed a trend towards decreased OS. Toxicities of the combined treatment were manageable; the safety profile matched that reported for patients with melanoma who receive dabrafenib and trametinib. Overall, the combination provided durable clinical benefit with a favorable risk/benefit ratio regardless of prior treatment.
REFERENCES
- Smit E et al., Trastuzumab deruxtecan in patients with HER2-mutated metastatic non-small cell lung cancer: interim results of DESTINY-Lung01. J Clin Oncol 38: 2020 (suppl; abstr 9504)
- Takeuchi K, Discovery stories of RET fusions in lung cancer: a mini-review. Frontiers Physiol 2019; 10: 216
- Tsuta K et al., RET-rearranged non-small-cell lung carcinoma: a clinicopathological and molecular analysis. Brit J Cancer 2014; 110(6): 1571-1578
- Drilon A et al., Registrational results of LIBRETTO-001: a phase 1/2 trial of selpercatinib (LOXO-292) in patients with RET-fusion–positive lung cancer. WCLC 2019, abstract #PL02.08
- Subbiah V et al., Intracranial activity of selpercatinib (LOXO-292) in RET fusion-positive non-small cell lung cancer (NSCLC) patients on the LIBRETTO-001 trial. J Clin Oncol 38: 2020 (suppl; abstr 9516)
- Gainor JF et al., Registrational dataset from the phase 1/2 ARROW trial of pralisetinib (BLU-667) in patients with advanced RET fusion+ non-small cell lung cancer (NSCLC). J Clin Oncol 38: 2020 (suppl; abstr 9515)
- Mok T et al., Updated overall survival and final progression-free survival data for patients with treatment-naïve advanced ALK-positive non-small-cell lung cancer in the ALEX study. Ann Oncol 2020; S0923-7534(20)39796-9
- Peters S et al., Updated overall survival and safety data from the randomized, phase III ALEX study of alectinib vs crizotinib in untreated advanced ALK+ NSCLC. J Clin Oncol 38: 2020 (suppl; abstr 9518
- Camidge DR et al., Brigatinib vs. crizotinib in patents with ALK inhibitor-naïve advanced ALK+ NSCLC: updated results from the phase III ALTA-1L trial. Ann Oncol 2019; 30 (suppl_9): ix183-ix202
- Camidge DR et al., Correlation of baseline molecular and clinical variables with ALK inhibitor efficacy in ALTA-1L. J Clin Oncol 38: 2020 (suppl; abstr 9517)
- Planchard D et al., Dabrafenib plus trametinib in patients with previously untreated BRAFV600E-mutant metastatic non-small-cell lung cancer: an open-label, phase 2 trial. Lancet Oncol 2017; 18(10): 1307-1316
- Planchard D et al., The updated overall survival and genomic analysis from a single-arm phase 2 study of dabrafenib plus trametinib in patients with BRAFV600E mutant metastatic non-small cell lung cancer. J Clin Oncol 38: 2020 (suppl; abstr 9593)
More posts
Rare mutations: HER2, RET, ALK, BRAF
Trastuzumab deruxtecan (T-DXd) is a novel antibody-drug conjugate containing a humanized anti-HER2 monoclonal antibody linked to a topoisomerase I inhibitor exatecan derivative. The open-label, multicenter, phase II DESTINY-Lung01 study tested T-DXd 6.4 mg/kg 3-weekly in patients with relapsed or refractory advanced NSCLC that expressed HER2 (Cohort 1; n = 42) or carried HER2-activating mutations (Cohort 2; n = 42).
COVID-19 in patients with thoracic cancers: TERAVOLT
The global consortium TERAVOLT was established to determine factors that place patients with thoracic malignancies who develop COVID-19 at risk for hospitalization and death, to elucidate the clinical course of these patients and to identify therapeutic strategies that might impact survival. Thoracic cancer patients with a COVID-19 diagnosis, i.e. cases of confirmed infection according to RT-PCR techniques and suspected COVID-19 cases, are being entered into the database.
Present and future perspectives of anti-angiogenic therapy
The oral, triple angiokinase inhibitor nintedanib has been approved in the European Union and other countries in combination with docetaxel for the treatment of advanced adenocarcinoma of the lung after first-line chemotherapy. It works by targeting vascular endothelial growth factor (VEGF) receptors 1-3, platelet-derived growth factor (PDGF) receptors α/β and fibroblast growth factor (FGF) receptors 1-3, as well as RET.
Improving outcomes in the early-stage setting with (neo)adjuvant strategies
Approximately 30 % of NSCLC patients present with resectable disease at diagnosis. Surgery is the primary treatment for early-stage NSCLC; after resection, adjuvant cisplatin-based chemotherapy is recommended for patients with stage II/IIIA lung cancer and select patients with stage IB disease. However, the rates for disease recurrence or death following surgery and adjuvant chemotherapy remain high, ranging from 45 % in stage IB to 76 % in stage III.
EGFR-mutated disease: early combinations and new approaches in exon 20 insertion-positive lung cancer
Oligometastatic disease is generally defined by one to five metastatic lesions. As progression occurs most frequently in sites of the original disease, it is surmised that aggressive local treatment might prevent further dissemination. Based on this rationale, the open-label, randomized, phase III SINDAS trial conducted in China explored the use of concurrent stereotactic body radiotherapy (SBRT) and EGFR TKI therapy in patients with oligometastatic, EGFR-mutant NSCLC.
Immune checkpoint inhibition: comprehensive benefits, but not devoid of risks
First-line nivolumab plus ipilimumab (NI) was shown to significantly prolong OS compared to chemotherapy in patients with advanced NSCLC irrespective of tumor PD-L1 expression in the randomized, phase III CheckMate 227 study. At the ASCO Congress, Ramalingam et al. presented the updated 3-year efficacy and safety results from Part 1 of the trial.