Present and future perspectives of anti-angiogenic therapy
The oral, triple angiokinase inhibitor nintedanib has been approved in the European Union and other countries in combination with docetaxel for the treatment of advanced adenocarcinoma of the lung after first-line chemotherapy. It works by targeting vascular endothelial growth factor (VEGF) receptors 1-3, platelet-derived growth factor (PDGF) receptors α/β and fibroblast growth factor (FGF) receptors 1-3, as well as RET [1, 2].
Given the changing treatment landscape in advanced NSCLC, the activity of nintedanib plus docetaxel is of particular interest in patients who have previously received immune checkpoint inhibitor (ICI) therapy. The optimal sequence after progression on this therapy has not yet been elucidated, although the underlying tumor biology can contribute to guiding the selection of treatment.
Angiogenesis plays a role in ICI resistance as excessive VEGF release can create an immunosuppressive tumor microenvironment [3, 4]. Therefore, anti-angiogenic strategies involving the inhibition of VEGF, PDGF and FGF might support vessel normalization and improve access of immune cells to the tumor. This might tip the balance towards an immunosupportive tumor microenvironment in the so-called angio-immunogenic switch.
Nintedanib plus docetaxel after ICI therapy
At the ASCO Congress, Grohé et al. reported updated results from 57 patients included in the Cohort B of the ongoing, non-interventional, prospective VARGADO trial [5]. In this cohort, patients with locally advanced, metastatic or locally recurrent adenocarcinoma of the lung received nintedanib plus docetaxel after first-line chemotherapy and second-line ICI treatment.
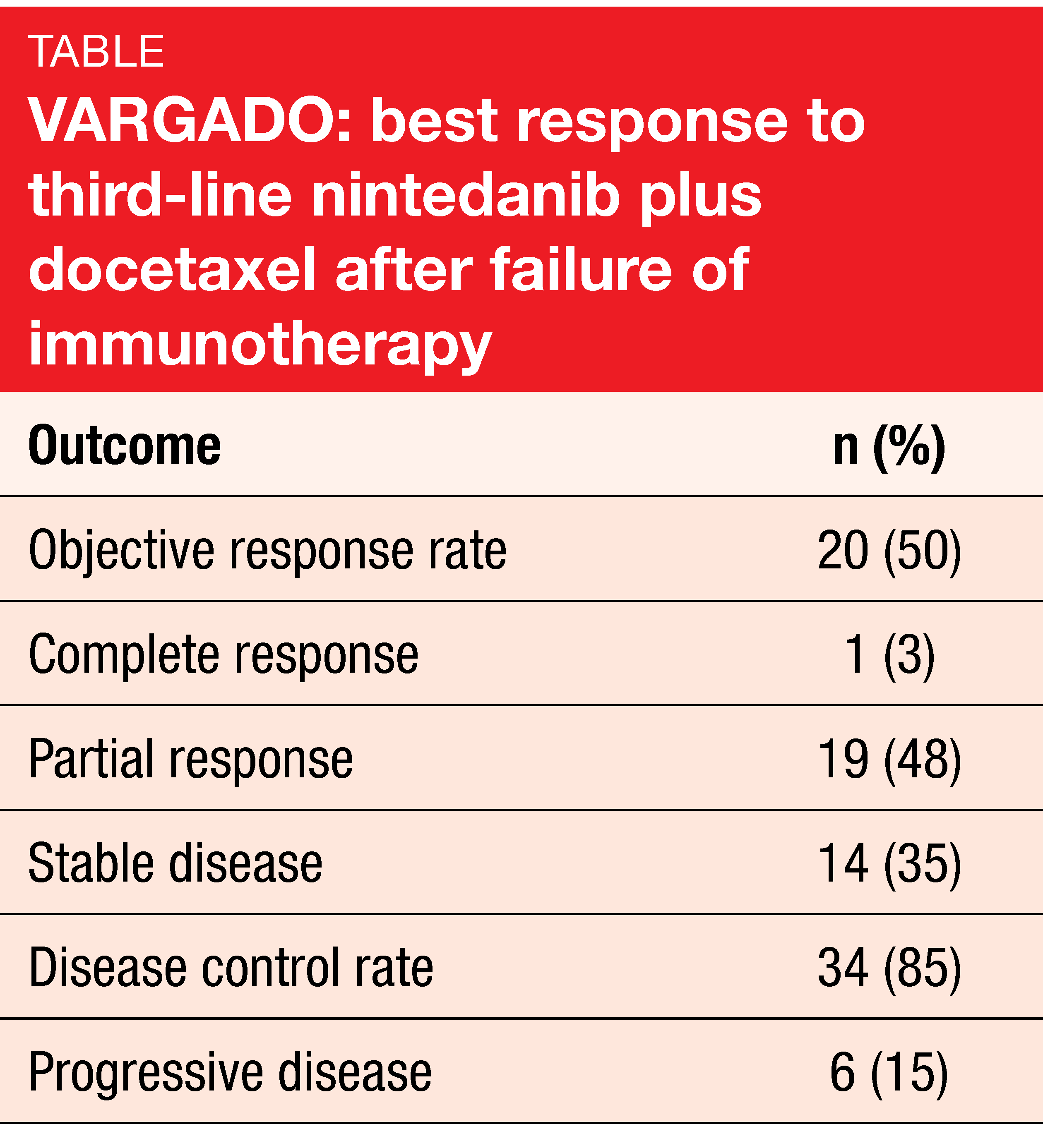
The updated analysis continued to demonstrate encouraging clinical benefit and a manageable safety profile of nintedanib plus docetaxel. From the start of third-line therapy, patients remained progression-free for a median of 6.5 months and survived for a median of 12.4 months. OS from the start of first-line therapy was 34.5 months. Twenty patients responded to the treatment, which translated to an ORR of 50 % (Table). One patient experienced a complete response. In 65 %, treatment-related AEs occurred, with the most common events being diarrhea (all grades, 37 %) stomatitis (12 %) and decreased white blood cell count (11 %). At least one dose reduction was performed in 26 % and 19 % for nintedanib and docetaxel, respectively. Treatment-emergent AEs led to discontinuation of study treatment in 30 % of patients.
As the authors noted, these data are consistent with the ICI-pretreated subgroup analysis of the LUME-BioNIs study evaluating nintedanib plus docetaxel [6] and previous data from the nintedanib named patient use program [7]. Rational sequencing of an anti-angiogenic agent after ICI therapy might be a promising approach that warrants further investigation.
RELAY+: ramucirumab combined with gefitinib
Preclinical and clinical studies have demonstrated activity of dual inhibition of the VEGF and EGFR pathways in EGFR-mutant NSCLC [8-11]. In the phase III RELAY study, the anti-VEGFR2 antibody ramucirumab in combination with the EGFR TKI erlotinib gave rise to prolonged PFS compared to erlotinib alone (19.4 vs. 12.4 months; HR, 0.59) [12]. As gefitinib is more commonly used in Japan than erlotinib, the exploratory open-label RELAY+ cohort evaluated ramucirumab 10 mg/kg every 2 weeks plus gefitinib 250 mg daily in 80 patients with previously untreated, EGFR-mutant, stage IV NSCLC [13].
The 1-year PFS rate, which constituted the primary exploratory objective, was similar between the ramucirumab plus gefitinib regimen (65 %) and the ramucirumab plus erlotinib regimen in RELAY (71.9 %) [12]. This also applied to ORR (71 % and 76 %, respectively) and DCR (99 % and 95 %, respectively). Moreover, the 1-year PFS rates were similar when viewed according to EGFR mutation subtype within the RELAY+ group (63 % and 67 % for L858R mutations and exon 19 deletions, respectively). Ramucirumab in combination with gefitinib proved tolerable, and the overall safety profile was consistent with the established safety profiles for each agent in EGFR-mutation–positive metastatic NSCLC.
NEJ026: no OS effect with bevacizumab plus erlotinib
The randomized, open-label, phase III NEJ026 trial tested bevacizumab plus erlotinib against single-agent erlotinib in patients with non-squamous, stage IIIB/IV or postoperatively recurrent NSCLC harboring activating EGFR mutations. PFS was significantly in favor of bevacizumab plus erlotinib (16.9 vs. 13.3 months; HR, 0.605; p = 0.016) [10]. However, the superiority of the combined strategy waned over time. The combination showed only slightly improved activity in terms of PFS2, which was defined as the time on second-line therapy after the first progression (28.6 vs. 24.3 months; HR, 0.773; p = 0.205) [14]. Finally, regarding OS, the addition of bevacizumab to erlotinib provided no further benefit (50.7 vs. 46.2 months; HR, 1.007). No subgroup appeared to derive a significant survival benefit from the regimen. These observations coincide with those from the phase II JO25567 study that revealed significant PFS improvement with bevacizumab plus erlotinib over erlotinib but showed comparable results for the two arms with respect to OS [15].
The authors surmised that the loss of efficacy of the combined regimen might be related to the long post-progression survival and the initiation of second-line osimertinib at the end of study treatment. Osimertinib was superior to treatment without osimertinib in both arms of the trial. According to liquid biopsy studies, bevacizumab plus erlotinib elicited T790M resistance mutations as frequently as first-generation EGFR TKIs did.
Meta-analysis: anti-angiogenesis plus erlotinib vs. erlotinib
Landre et al. performed a meta-analysis of five randomized trials comparing VEGF inhibition plus erlotinib with erlotinib alone in the first-line treatment of advanced NSCLC harboring EGFR mutations [16]. The analysis included the NEJ026, ARTEMIS, RELAY, and J025567 studies, as well as the US-based trial by Stinchcombe et al. RELAY was conducted with ramucirumab in addition to erlotinib, while all of the others used bevacizumab as the anti-angiogenic partner of the EGFR TKI. For all studies, PFS had been defined as the primary endpoint.
According to the results, the combined inhibition of VEGF and EGFR is associated with significantly improved PFS and duration of response compared to erlotinib alone. However, mature data for OS are required to confirm the benefit of this strategy. Moreover, the outcomes suggested that the combination might slow the emergence of resistance to EGFR TKIs. In the ARTEMIS trial, at the time of progression, patients in the combination arm showed fewer acquired resistance mutations, such as T790M, than the patients in the monotherapy arm. The same trend was observed in RELAY.
VEGR inhibition with anlotinib
The TKI anlotinib is an anti-angiogenic drug that targets multiple receptor tyrosine kinases including VEGFR2 and VEGFR3. Due to its oral route of administration, anlotinib offers advantages over bevacizumab and ramucirumab, which are administered intravenously. Huang et al. conducted a single-arm study to investigate the activity of anlotinib combined with the oral EGFR TKI icotinib in untreated patients with EGFR-mutated, IIIB, IIIC or IV non-squamous NSCLC [17]. Thirty-five patients were evaluated for toxicity, and 30 of these were evaluable for efficacy.
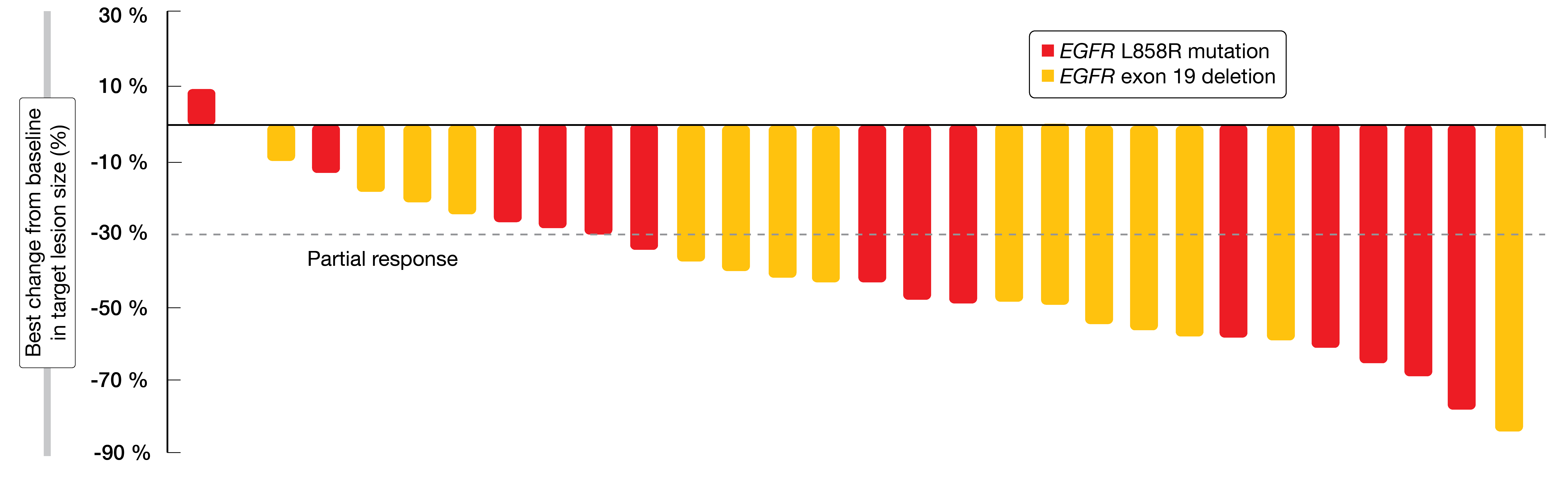
Anlotinib plus icotinib demonstrated encouraging efficacy in the first-line setting. Overall, 21 patients (70.0 %) responded, with all of them achieving partial remission. PRs were obtained by 11 patients with exon 19 deletions (73.3 %) and 10 of those with L858R mutations (66.7 %; Figure). Disease control resulted in 96.7 %, as another eight patients experienced disease stabilization. Eighteen patients harbored additional aberrations regarding oncogenic drivers (PIK3CA or AKT1) and/or tumor suppressors (TP53, RB1, PTEN); here, the ORR was 83.3 %. Anlotinib plus icotinib was well tolerated, and AEs proved manageable. The most common AEs included hypercholesterolemia, hypertriglyceridemia, hypertension, diarrhea, and rash. Among grade-3 AEs, hypertension (17 %) and hypertriglyceridemia (6 %) occurred most frequently. The only grade-4 event was hypertriglyceridemia (6 %). PFS and OS outcomes are awaited as longer follow-up is required for further evaluation.
VEGF/Ang-2 blockade and checkpoint inhibition
Another potential combination strategy based on anti-angiogenesis consists in the inhibition of VEGF/Angiopoietin-2 (Ang-2) together with an immunotherapeutic approach. A phase Ib trial established preliminary antitumor activity of the VEGF/Ang2-blocking nanobody® BI 836880 combined with the anti-PD-1 antibody BI 754091 at doses of 720 mg and 240 mg, respectively, three-weekly [18]. Ten of 12 patients with locally advanced or metastatic non-squamous NSCLC achieved partial remissions or disease stabilization. Two thirds of them had already received ICI therapy prior to enrollment. Changes in target lesions were observed in both ICI-pretreated and ICI-naïve patients.
The combination showed a manageable safety profile. All-grade AEs comprised hypertension, vomiting, nausea, and asthenia. No grade-4 events occurred. Expansion cohorts are ongoing, and further results can be expected.
REFERENCES
- Hilberg F et al., BIBF 1120: Triple angiokinase inhibitor with sustained receptor blockade and good antitumor efficacy. Cancer Res 2008; 68(12): 4774-4782
- Hilberg F et al., Triple angiokinase inhibitor nintedanib directly inhibits tumor cell growth and induces tumor shrinkage via blocking oncogenic receptor tyrosine kinases. J Pharmacol Exp Ther 2018; 364(3): 494-503
- Fukumura D et al., Enhancing cancer immunotherapy using antiangiogenics: Opportunities and challenges. Nat Rev Clin Oncol 2018; 15(5): 325-340
- van der Woude LL et al., Migrating into the tumor: A roadmap for T cells. Trends Cancer 2017; 3(11): 797-808
- Grohé C et al., Nintedanib plus docetaxel in lung adenocarcinoma patients following treatment with immune checkpoint inhibitors: updated efficacy and safety results of the ongoing non-interventional study VARGADO (NCT02392455). J Clin Oncol 38: 2020 (suppl; abstr 9604)
- Reck M et al., Nintedanib + docetaxel after immunotherapy in adenocarcinoma non-small cell lung cancer: first results from the non-interventional LUME-BioNIS study. Ann Oncol 2019; 30(Suppl. 11): abstract 180
- Corral J et al., Efficacy of nintedanib and docetaxel in patients with advanced lung adenocarcinoma treated with first-line chemotherapy and second-line immunotherapy in the nintedanib NPU Program. Clin Transl Oncol 2019; 21(9): 1270-1279
- Seto T et al., Erlotinib alone or with bevacizumab as first-line therapy in patients with advanced non-squamous non-small-cell lung cancer harbouring EGFR mutations (JO25567): An open-label, randomised, multicentre, phase 2 study. Lancet Oncol 2014; 15(11): 1236-1244
- Ichihara E et al., Phase II trial of gefitinib in combination with bevacizumab as first-line therapy for advanced non-small cell lung cancer with activating EGFR gene mutations: The Okayama Lung Cancer Study Group Trial 1001. J Thorac Oncol 2015; 10(3): 486-491
- Saito H et al., Erlotinib plus bevacizumab versus erlotinib alone in patients with EGFR-positive advanced non-squamous non-small-cell lung cancer (NEJ026): Interim analysis of an open-label, randomised, multicentre, phase 3 Trial. Lancet Oncol 2019; 20(5): 625-635
- Byers LA, Heymach JV, Dual targeting of the vascular endothelial growth factor and epidermal growth factor receptor pathways: Rationale and clinical applications for non-small-cell lung cancer. Clin Lung Cancer 2007; 8(suppl 2): S79-S85
- Nakagawa K et al., Ramucirumab plus erlotinib in patients with untreated, EGFR-mutated, advanced non-small-cell lung cancer (RELAY): A randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol 2019; 20(12): 1655-1669
- Nishio M et al., RELAY+: Exploratory study of ramucirumab plus gefitinib in untreated patients with epidermal growth factor receptor (EGFR)-mutated metastatic non-small cell lung cancer (NSCLC). J Clin Oncol 38: 2020 (suppl; abstr 9564
- Maemondo M et al., NEJ026: final overall survival analysis of bevacizumab plus erlotinib treatment for NSCLC patients harboring activating EGFR mutations. J Clin Oncol 38: 2020 (suppl; abstr 9506
- Yamamoto N et al. Erlotinib plus bevacizumab (EB) versus erlotinib alone (E) as first-line treatment for advanced EGFR mutation–positive non-squamous non–small-cell lung cancer (NSCLC): Survival follow-up results of JO25567. J Clin Oncol 36, 2018 (suppl; abstr 9007
- Landre T et al., Angiogenesis inhibitor plus erlotinib versus erlotinib alone as first-line for advanced non-small cell lung cancer harboring EGFR mutation. J Clin Oncol 38: 2020 (suppl; abstr 9569)
- Huang D et al., Study of anlotinib combined with icotinib as the first-line treatment in NSCLC patients harboring activating EGFR mutations (ALTER-L004). J Clin Oncol 38: 2020 (suppl; abstr 9573)
- Girard N et al., Phase Ib study of BI 836880, a VEGF/Ang2-blocking nanobody®, in combination with BI 754091, an anti-PD-1 antibody: initial results in patients with advanced non-small cell lung cancer. J Clin Oncol 38: 2020 (suppl; abstr 9566)
More posts
Rare mutations: HER2, RET, ALK, BRAF
Trastuzumab deruxtecan (T-DXd) is a novel antibody-drug conjugate containing a humanized anti-HER2 monoclonal antibody linked to a topoisomerase I inhibitor exatecan derivative. The open-label, multicenter, phase II DESTINY-Lung01 study tested T-DXd 6.4 mg/kg 3-weekly in patients with relapsed or refractory advanced NSCLC that expressed HER2 (Cohort 1; n = 42) or carried HER2-activating mutations (Cohort 2; n = 42).
COVID-19 in patients with thoracic cancers: TERAVOLT
The global consortium TERAVOLT was established to determine factors that place patients with thoracic malignancies who develop COVID-19 at risk for hospitalization and death, to elucidate the clinical course of these patients and to identify therapeutic strategies that might impact survival. Thoracic cancer patients with a COVID-19 diagnosis, i.e. cases of confirmed infection according to RT-PCR techniques and suspected COVID-19 cases, are being entered into the database.
Present and future perspectives of anti-angiogenic therapy
The oral, triple angiokinase inhibitor nintedanib has been approved in the European Union and other countries in combination with docetaxel for the treatment of advanced adenocarcinoma of the lung after first-line chemotherapy. It works by targeting vascular endothelial growth factor (VEGF) receptors 1-3, platelet-derived growth factor (PDGF) receptors α/β and fibroblast growth factor (FGF) receptors 1-3, as well as RET.
Improving outcomes in the early-stage setting with (neo)adjuvant strategies
Approximately 30 % of NSCLC patients present with resectable disease at diagnosis. Surgery is the primary treatment for early-stage NSCLC; after resection, adjuvant cisplatin-based chemotherapy is recommended for patients with stage II/IIIA lung cancer and select patients with stage IB disease. However, the rates for disease recurrence or death following surgery and adjuvant chemotherapy remain high, ranging from 45 % in stage IB to 76 % in stage III.
EGFR-mutated disease: early combinations and new approaches in exon 20 insertion-positive lung cancer
Oligometastatic disease is generally defined by one to five metastatic lesions. As progression occurs most frequently in sites of the original disease, it is surmised that aggressive local treatment might prevent further dissemination. Based on this rationale, the open-label, randomized, phase III SINDAS trial conducted in China explored the use of concurrent stereotactic body radiotherapy (SBRT) and EGFR TKI therapy in patients with oligometastatic, EGFR-mutant NSCLC.
Immune checkpoint inhibition: comprehensive benefits, but not devoid of risks
First-line nivolumab plus ipilimumab (NI) was shown to significantly prolong OS compared to chemotherapy in patients with advanced NSCLC irrespective of tumor PD-L1 expression in the randomized, phase III CheckMate 227 study. At the ASCO Congress, Ramalingam et al. presented the updated 3-year efficacy and safety results from Part 1 of the trial.