Immunotherapy: novel biomarkers on the horizon & news from pivotal trials
Is tumour mutation burden relevant in SCLC?
Only limited treatment options are available for patients with recurrent small-cell lung cancer (SCLC). The CheckMate 032 trial evaluated the anti-PD-1 antibody nivolumab with or without the anti-CTLA-4 antibody ipilimumab in a PD-L1–unselected cohort of SCLC patients who had received at least one prior platinum-based chemotherapy regimen. Both nivolumab alone and the combination showed impres-sive activity in this setting: 2-year OS rates were 26 % and 14 %, respectively [1]. Responses occurred regardless of PD-L1 status.
As PD-L1 expression is uncommon in SCLC, improved biomarkers are needed for immunotherapy in this tumour type. The randomised, phase III CheckMate 026 trial that compared frontline nivolumab with chemotherapy has identified tumour mutation burden (TMB) as a predictive biomarker for the use of nivolumab [2]. Antonia et al. therefore conducted an exploratory TMB analysis of the CheckMate 032 trial data with the aim of assessing if this observation holds true for SCLC [3].
The TMB-evaluable group included 211 patients, 133 and 78 of whom received single-agent nivolumab and nivolumab plus ipilimumab, respectively. TMB was determined by whole-exome sequencing, and was calculated as the total number of missense mutations in the tumour. For the analysis, patients were divided into 3 subgroups based on the TMB tertile. The TMB-evaluable patients were representative of the overall population, with comparable PFS and OS outcomes in both treatment arms.
Improved activity with high TMB
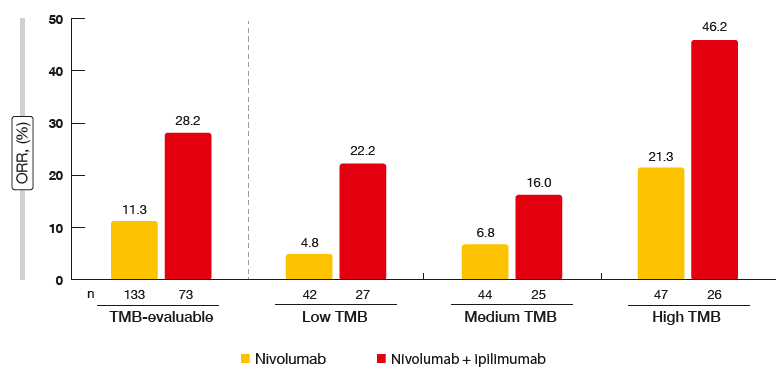
According to the ORR analysis by TMB subgroup, an incremental increase was observed for nivolumab treatment, ranging from 4.8 % to 21.3 % across the groups with low, medium and high TMB (Figure 1). The combination of nivolumab and ipilimumab, on the other hand, gave rise to similar response rates in the low and medium TMB cohorts, while the high TMB group showed an impressive ORR of 46.2 %.
In a similar vein, there was a differential benefit for PFS, with the high TMB cohort experiencing considerably longer progression-free intervals with both treatments. At 1 year, PFS rates were 21.2 % and 30.0 % for nivolumab and the combination, respectively. As opposed to this, patients with low and medium TMB showed PFS rates in the single-digit range. The OS analysis revealed an incremental survival effect of rising TMB for nivolumab monotherapy; here, 1-year OS rates were 22.1 %, 26.0 % and 35.2 % for patients with low, medium, and high TMB, respectively. In contrast, patients receiving both nivolumab and ipilimumab fared considerably better when they had high TMB (1-year OS rate, 62.4 %) compared with low or medium TMB (23.4 % and 19.6 %, respectively).
Overall, as for the NSCLC setting, patients with SCLC were shown to respond differently to immunotherapeutic treatment according to their tumour mutation load. In those with high TMB, improved outcomes resulted for both nivolumab monotherapy and nivolumab plus ipilimumab, but the findings were clearly more robust for the combination. Optimisation of the TMB cut-off and prospective investigation of TMB are warranted. As the authors concluded, TMB might be a relevant predictive biomarker across all lung cancers.
Figure 1: CheckMate 032: response rates according to tumour mutation burden
OAK: analysis according to Teff gene expression
High expression of the T-effector (Teff) gene signature, which is a marker of pre-existing immunity, has been demonstrated to correlate with improved survival in patients treated with the anti-PD-L1 antibody atezolizumab in the phase II POPLAR study [4]. Therefore, a retrospective, exploratory analysis of the phase III OAK study was conducted to assess the association between Teff gene expression and the clinical benefit achieved with atezolizumab in this trial [5]. Patients included in the OAK trial had received either atezolizumab or docetaxel in the second-line or later-line settings. The primary analysis showed significantly improved median OS with atezolizumab compared to docetaxel (13.8 vs. 9.6 months; HR, 0.73; p = 0.0003), but a PFS benefit with atezolizumab was only observed in patients with high PD-L1 expression [6].
In the primary OAK population, 753 patients had tumour tissue sufficient for the evaluation of Teff gene expression. For the purposes of this analysis, the Teff signature was defined by mRNA expression of three genes (PDL1, CXCL9, IFNG). There was a partial overlap between the Teff gene signature and PD-L1 expression according to immunohis-tochemistry (IHC), but at the same time, the Teff gene signature identified a unique patient subset within the PD-L1–negative population.
Higher accuracy of PFS prediction
The results of the study imply that the Teff gene signature is a more sensitive biomarker of PFS than PD-L1 expression. Three different Teff gene expression levels were assessed. The analysis showed a significant association between higher expression levels and the atezolizumab-mediated PFS benefit. PFS HR was 0.73 for patients with high Teff gene expression (≥ 50 %), but 1.30 for those with low expression (< 50 %). Compared to PD-L1 status according to IHC, the Teff signature identified a larger number of patients who experienced a significant PFS benefit with ate-zolizumab therapy at a comparable population prevalence of the two biomarkers (Table). For OS, the atezolizumab-mediated benefit resembled that observed in the entire biomarker-evaluable population, although the Teff signature also enriched for improved results at all expression cut-offs.
These findings suggest that pre-existing immunity could be an important biological aspect determining the efficacy of immunotherapeutic agents in lung cancer patients. Ongoing studies are designed to further validate a role for the Teff gene signature as a potential predictive biomarker of immunotherapy efficacy in first-line NSCLC treatment.
PACIFIC study: function and quality of life
The double-blind, placebo-controlled, international, phase III PACIFIC trial compared the anti-PD-L1 antibody durvalumab 10 mg/kg every 2 weeks for up to 12 months (n = 476) with placebo (n = 237) in patients with stage III, lo-cally advanced, unresectable NSCLC who had not progressed following definitive platinum-based concurrent chemoradiation. Patients were not selected according to PD-L1 expression status. The interim PFS analysis yielded significantly superior findings with durvalumab compared to placebo (median PFS, 16.8 vs. 5.6 months; HR, 0.52; p < 0.0001) [7].
At the WCLC, Hui et al. presented patient-reported outcomes (PROs), which were a prespecified secondary endpoint of the PACIFIC study [8]. Symptoms, physical function and global health status/ quality of life were evaluated using the EORTC QLQ-C30 v3 questionnaire and its lung cancer module, QLQ-LC13. According to this, the scores for key symptoms as well as functioning and global health status remained stable throughout the study with both durvalumab and placebo. There were no significant differences between arms with regard to changes from baseline. Clinically relevant improvements within each arm from baseline were observed at week 48 for dysphagia and alopecia, which suggests resolution of toxicities related to the concurrent chemoradiation therapy that all patients received. Odds of improvement of the item ,appetite loss’ were greater with durvalumab, while no between-arm differences in improvement rates existed for functioning or other symptoms. For time to deterioration of functioning and symptoms, the analysis showed no differences between durvalumab and placebo with regard to most of the items. Only time to deterioration of ,other pain’ was longer with durvalumab (HR, 0.72). This difference, however, was not reflected in any additional ,pain’ terms.
Overall, this analysis showed that adding durvalumab for 12 months after chemoradiation did not compromise quality of life in patients with locally advanced, unresectable NSCLC. Alongside the positive efficacy and safety data from PACIFIC, these findings further support the clinical value of durvalumab in the early-stage setting.
First-line pembrolizumab: update of KEYNOTE-024
Brahmer et al. presented the updated analysis of the international, randomised, open-label, phase III KEY-NOTE-024 trial that compared pembrolizumab and platinum-based chemotherapy in 305 untreated patients with stage IV NSCLC expressing PD-L1 (tumour proportion score [TPS] ≥ 50 %) [9]. The primary analysis has revealed significant superiority of pembrolizumab over chemotherapy with respect to PFS (HR, 0.50) and OS (HR, 0.60), but median OS in the pembrolizumab arm had not been reached at that time [10].
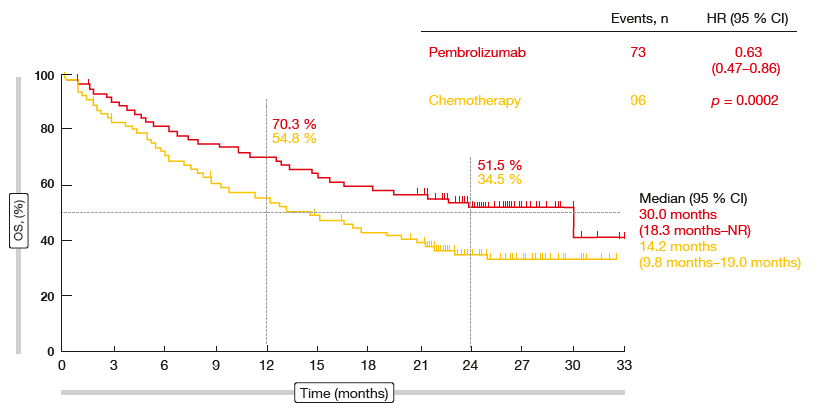
After a median follow-up of 25.2 months, the updated OS analysis showed a significant benefit for pembrolizumab with a remarkable median survival outcome of 30.0 months (vs. 14.2 months in the chemotherapy arm; HR, 0.63; p = 0.002; Figure 2). 24-month OS rates were 51.5 % and 34.5 % for patients treated with pembrolizumab and chemotherapy, respectively. This improvement was maintained in spite of a significant effective crossover rate to anti-PD-1 treatment in the chemotherapy arm that amounted to 63 %.
ORRs were 45.5 % vs. 29.8 % for pembrolizumab and chemotherapy, respectively (p = 0.0031). Patients who crossed over to pembrolizumab experienced an ORR of 20.7 %. In all pembrolizumab-treated patients, median duration of response had not been reached yet (vs. 7.1 months in the chemotherapy arm). After a median exposure of 7.9 months, which was more than double that in the chemotherapy arm, pembrolizumab continued to demonstrate a favourable safety profile. The authors concluded that pembrolizumab remains a standard of care for the first-line therapy of patients with NSCLC and high PD-L1 expression (TPS ≥ 50 %).
Figure 2: Updated overall survival outcomes in the KEYNOTE-024 trial
Nivolumab in patients with brain metastasis
An Italian expanded access programme offered the opportunity to evaluate nivolumab 3 mg/kg every 2 weeks for a maximum of 24 months in patients with stage IIIB/IV, nonsquamous NSCLC and CNS metastases outside of a controlled clinical trial [11]. Patients with brain lesions were eligible if they had no neurologic symptoms at least 2 weeks before enrolment and did not require systemic corticosteroid treatment or were on a stable or decreasing dose of ≤ 10mg/day of prednisone or prednisone equivalent. Among the total population of 1,588 patients who participated at 153 centres, 409 (26 %) had asymptomatic and controlled brain metastases. Twenty-nine percent were receiving steroid therapy at baseline, and 18 % had concomitant radiotherapy.
Efficacy and safety of nivolumab therapy in this group appeared similar to that observed in the overall cohort and the CheckMate 057 trial population [12]. ORR and DCR were 17 % and 40 %, respectively. CR occurred in four patients (1 %), PR in 64 patients (16 %), and SD in 96 patients (23 %). Median OS was 8.6 months for patients with CNS metastasis, compared with 11.3 months for all patients. At 1 year, 43 % and 48 % of patients in the CNS metastasis and overall cohorts, respectively, were alive. Both the CNS cohort and the overall population showed a median PFS of 3.0 months. PFS 1-year rates were 20 % and 22 %. These results suggested that patients with CNS metastasis could benefit from immunotherapy with nivolumab.
REFERENCES
- Hellmann MD et al., Nivolumab (nivo) ± ipilimumab (ipi) in advanced small-cell lung cancer (SCLC): First report of a randomized expansion cohort from CheckMate 032. J Clin Oncol 35, 2017 (suppl; abstr 8503)
- Carbone DP et al., First-line nivolumab in stage IV or recurrent non-small-cell lung cancer. N Engl J Med 2017; 376: 2415-2426
- Antonia SJ et al., Impact of tumor mutation burden on the efficacy of nivolumab or nivolumab + ipilimumab in small cell lung cancer: an exploratory analysis of Checkmate 032. WCLC 2017, OA 07.03a
- Fehrenbacher L et al., Atezolizumab versus docetaxel for patients with previously treated non-small-cell lung cancer (POPLAR): a multicentre, open-label, phase 2 randomised controlled trial. Lancet 2016; 387(10030): 1837-1846
- Kowanetz M et al., Pre-existing immunity measured by Teff gene expression in tumor tissue is associated with atezolizumab efficacy in NSCLC. WCLC 2017, MA 05.09
- Rittmeyer A et al., Atezolizumab versus docetaxel in patients with previously treated non-small-cell lung cancer (OAK): a phase 3, open-label, multicentre randomised controlled trial. Lancet 2017; 389(10066): 255-265
- Antonia SJ et al., Durvalumab after chemoradiotherapy in stage III non-small-cell lung cancer. N Engl J Med 2017 Sep 8. doi: 10.1056/NEJ-Moa1709937. [Epub ahead of print]
- Hui R et al., Patient-reported outcomes with durvalumab after chemoradiation in locally advanced, unresectable NSCLC: data from PA-CIFIC. WCLC 2017, PL 02.02
- Brahmer JR et al., Updated analysis of KEY-NOTE-024: pembrolizumab versus platinum-based chemotherapy for advanced NSCLC with PD-L1 TPS ≥ 50%. WCLC 2017, OA 17.06
- Reck M et al., Pembrolizumab versus chemotherapy for PD-L1-positive non-small-cell lung cancer. N Engl J Med 2016; 375: 1823-1833
- Crinò L et al., Italian nivolumab expanded access programme (EAP): data from patients with advanced non-squamous NSCLC and brain metastasis. WCLC 2017, P1.01-053
- Borghaei H et al., Nivolumab versus docetaxel in advanced nonsquamous non-small-cell lung cancer. N Engl J Med 2015; 373: 1627-1639
More posts
Chemotherapy: new approaches, new settings
Current guidelines recommend postoperative platinum-based chemotherapy in completely resected NSCLC with nodal involvement (stage II-IIIA). However, survival outcomes remain limited, and compliance is lower than for adjuvant therapy in other neoplasms. There are no direct comparisons between different chemotherapy regimens.
Malignant mesothelioma: recent data on nintedanib and checkpoint inhibitors
Malignant pleural mesothelioma (MPM) is an aggressive tumour that, if left untreated, shows a median survival of 7–9 months. The front-line standard treatment for patients with unresectable MPM consists of combination doublet therapy with cisplatin and pemetrexed, which yields a median OS of approximately 1 year.
Approaching squamous-cell carcinoma in a targeted manner
The EGFR mutation status is not routinely examined in NSCLC patients with squamous cell cancer (SCC) histology due to the low incidence of EGFR mutations in these tumours and poor clinical response to first-generation EGFR TKI treatment. Taniguchi et al. retrospectively reviewed 441 consecutive patients in 23 of whom the EGFR mutation status was assessed, in order to explore the clinical features of SCC with sensitive EGFR mutation, and to select the optimal indications for afatinib treatment.
Immunotherapy: novel biomarkers on the horizon & news from pivotal trials
Only limited treatment options are available for patients with recurrent small-cell lung cancer (SCLC). The CheckMate 032 trial evaluated the anti-PD-1 antibody nivolumab with or without the anti-CTLA-4 antibody ipilimumab in a PD-L1–unselected cohort of SCLC patients who had received at least one prior platinum-based chemotherapy regimen.
Taking anti-EGFR drug treatment further: later lines
Acquired resistance usually follows first-line EGFR TKI therapy, with the gatekeeper T790M mutation being the most common mechanism. The third-generation irreversible EGFR TKI osimertinib has been licensed for the treatment of patients whose tumours have been shown to carry this mutation. Retrospective data presented by Tan et al. demonstrated the activity of later-line osimertinib in 52 patients who participated in an early access program in Singapore.
“We are making steady progress toward better lung cancer control”
In a way, synergy is another expression for the multidisciplinary team approach, but the term ,multidisciplinary’ is not necessarily restricted to medical doctors. It also includes nursing staff and others such as the supportive care team, including the rehabilitation team and patient advocates. At the same time, the bottom line of that concept is having the patient at the centre of the overall care plan.