Guidelines for the treatment of NSCLC in China: progress and controversies
Lung cancer (LC) is the leading cause of tumor deaths worldwide. Per year, 1.8 million people are diagnosed with LC, and the annual death toll amounts to 1.6 million. Non–small-cell lung cancer (NSCLC) is the most common type, which accounts for 85% of LC cases. Squamous cell carcinoma (SqCC), adenocarcinoma (AC), and large-cell carcinoma are subtypes of NSCLC. The 5-year survival rate of NSCLC is only approximately 15%, and nowadays almost 70% of NSCLC patients are in advanced stages at the time of diagnosis [1].
The guidelines of the Chinese Society of Clinical Oncology (CSCO) for LC are based on evidence-based medicine and precision medicine. However, the availability of health resources should be considered simultaneously. China is an unequally developed country, and implementing the guidelines for LC treatment should consider imbalanced development of regions, accessibility to drugs and treatment methods as well as the value of cancer treatment. Thus, for each clinical problem, the Chinese guidelines give a standard recommendation for cancer care as a basic strategy, as well as a cost-effective therapeutic strategy for regions with low accessibility to drugs as an optional strategy. At CSCO 2017, which was held in Xiamen from September 26th to 30th, 2017, the objective was to introduce these guidelines for the treatment of NSCLC and to discuss the main controversies these recommendations bring.
Guidelines for stage IA-IB NSCLC – controversy: stereotactic ablative radiotherapy or sublobectomy?
For stage IA-IB NSCLC patients who are suitable for surgery, the 2017 CSCO guidelines recommend that pulmonary lobectomy plus systematic mediastinal lymph node dissection by minimally invasive technique should be the basic strategy (IIA). An additional recommendation is the participation in a clinical trial which compares surgery to stereotactic ablative radiotherapy (SABR) (level 3 evidence).
For stage IA-IB NSCLC patients who are not suitable for surgery, stereotactic body radiation therapy (SBRT)/SABR should be the basic strategy (IIA), and SBRT/SABR by other advanced radiotherapy (RT) technology, such as RT with photons, protons and carbon-ions, should be the optional strategy (IIA) [2].
SBRT or SABR has been the standard of care (SOC) for NSCLC patients who are not suitable for surgery. However, as for early-stage NSCLC patients who are suitable for surgery, the controversy whether SABR/SBRT should be an alternative treatment option besides the SOC (i.e., pulmonary lobectomy and lymph node dissection) still exists.
The studies STARS and ROSEL compared SABR to surgery in operable early-stage NSCLC. Histological confirmation of NSCLC by biopsy or cytological evaluation was required in the STARS trial but was not mandatory in ROSEL. In ROSEL, Dutch patients were enrolled. Furthermore, patients for whom no pathological confirmation of diagnosis was available were eligible if they had a new or growing pulmonary lesion with radiological features consistent with malignant disease and avidity on 18F-fluorodeoxyglucose (18F-FDG-PET). Patients in these two studies were randomly assigned in a 1:1 ratio to receive either SABR or surgery. In total, 58 patients were enrolled in the two studies, as 36 patients participated in STARS and 22 in ROSEL. The median follow-up times were 40.2 and 35.4 months for the group that received SABR and the group that underwent surgery, respectively. Estimated overall survival (OS) at 3 years was 95 % in the SABR group compared to 79 % in the surgery group (p = 0.037), and recurrence-free survival (RFS) at 3 years was 86 % and 80 % for the SABR and the surgery groups, respectively (p = 0.54) [3]. Six patients in the surgery group died, compared to one patient in the SABR group.
In summary, these results showed longer OS, better RFS and fewer deaths in the SABR group compared to the surgery group in early-stage NSCLC. The data suggests that SABR could be a better choice for early-stage NSCLC compared to surgery. Surprisingly, another large study, NCDB, was found to contradict these results. Data from this study showed that SABR was associated with a significantly reduced 5-year survival rate compared to surgery in both the unmatched analysis (30.9 % vs. 55.2 %; p < 0.001) and the analysis adjusted for covariates (31.0 % vs. 49.9 %; p < 0.001). In this study, SABR was associated with worse OS compared to the surgery group according to 2 subgroup analyses in propensity-matched patients (both p < 0.05) [4].
In operable stage IA-IB NSCLC patients, surgery is still the standard treatment, with SABR as an optional treatment. In inoperable stage IA-IB NSCLC patients, stereotactic ablative radiotherapy is the preferred treatment for LC care.
Guidelines for stage IIIA-N2 NSCLC – controversy 1: neoadjuvant therapy plus surgery or concurrent chemoradiotherapy
Stage IIIA NSCLC is a heterogeneous disease. The treatment strategies differ depending on the stage of the tumor (T), the presence of lymph node metastasis (N) and resectability of the tumor (Table 1).
The main controversies regarding stage IIIA NSCLC relate to whether neoadjuvant therapy plus surgery could substitute concurrent chemoradiotherapy and whether post-operative RT is necessary in operable patients with clinical stage IIIA-N2 after surgery. The opinions uttered in various studies on whether neoadjuvant therapy should be added to the basic strategy or substitute the concurrent radiochemotherapy are conflicting.
In the INT0139 trial, which enrolled 429 patients with stage IIIA NSCLC, all patients were randomized into surgery or radical RT groups after treatment with concurrent chemoradiotherapy. The study revealed increased progression-free survival (PFS; 12.8 months vs. 10.5 months; p = 0.017) and OS (33.6 months vs. 21.7 months; p = 0.002) for neoadjuvant treatment plus surgery compared to neoadjuvant treatment plus concurrent RT in operable stage IIIA-N2 NSCLC patients [5]. In contrast, other studies showed no PFS or OS differences between neoadjuvant therapy plus surgery and concurrent RT. In the EORTC08941 study that enrolled 579 patients, all patients were randomized to either the surgery group or the RT group after 3 weeks of neoadjuvant therapy, and received relevant treatment. No statistically significant difference in OS (16.4 months vs. 17.5 months; p = 0.596) and PFS (9 months vs. 11.3 months; p = 0.605) was observed across the surgery and RT groups [6]. Similar results were reported from the SAKK, ESPATUE and GLCCG studies, which showed no significant difference between neoadjuvant therapy plus surgery and concurrent RT.
Thus, based on the current evidence for treatment of operable stage IIIA-N2 NSCLC patients, concurrent RT is still the basic strategy. Additional studies on neoadjuvant therapy for the treatment of NSCLC patients with stage IIIA-N2 are required.
Guidelines for stage IIIA-N2 NSCLC – controversy 2: post-operative radiotherapy
Another controversy in the treatment of stage IIIA-N2 NSCLC patients is whether post-operative radiation therapy (PORT) should be applied or not. Precise RT technologies, such as three-dimensional conformal radiation therapy and intensity-modulated radiation therapy, have been widely used for the treatment of LC. This treatment strategy decreases non-cancer-related deaths caused by cardiotoxicity.
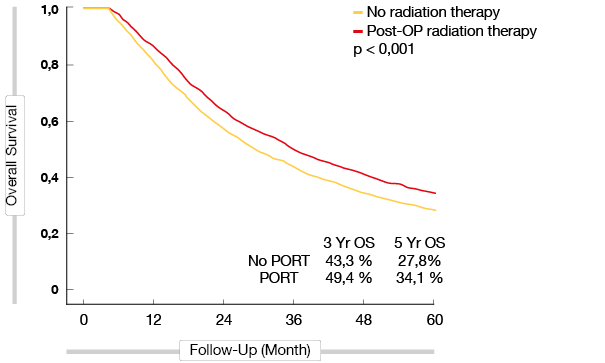
Corso et al. conducted a retrospective case-control study, which included 6,979 stage II-III-N2 NSCLC patients. The results showed that the 5-year OS rate in the PORT group was significantly higher than in the control group (34.1% vs. 27.8 %; p < 0.001), with a 6.3% OS increase in the PORT group [7] (Figure 1). According to Urban et al., who analyzed data of 4,773 N2 patients from the SEER database who underwent surgery, the risk of death was notably decreased in the PORT group (HR,0.9; p = 0.026) [8]. Mikell et al. and Robison et al. reported similar results in pN2 NSCLC patients that favored PORT [9, 10]. However, Wisnivesky et al. reported that PORT did not improve survival in old (≥65 years) pN2 NSCLC patients, (HR, 1.1; p = 0.3) [11].
Thus, the treatment with post-operative radiotherapy revealed a notably increased efficacy in the treatment of stage III-N2 NSCLC patients. However, it is still not clear whether PORT is suitable for older patients. Therefore, additional studies are called for.
Figure 1: Overall survival of stage IIIA-N2 patients with and without PORT
Guidelines for stage IV EGFR-mutant NSCLC: afatinib as first-line recommendation
In the 2017 CSCO guidelines, the standard first-line strategy for patients with stage IV epidermal growth factor receptor (EGFR)-mutant NSCLC includes the tyrosine kinase inhibitors (TKIs) afatinib, gefitinib, icotinib and erlotinib. Optional strategies include erlotinib or gefitinib plus chemotherapy, platinum-based chemotherapy or platinum-based chemotherapy plus the VEGF inhibitor bevacizumab (performance status [PS], 0–1; IIA). For stage IV patients with EGFR-mutant NSCLC after resistance to first-line EGFR TKI, treatment decisions should be made based on the status of progression. The guidelines recommend continuing EGFR TKI plus local therapy (IIA) for localized progression, and continuation of initial EGFR TKI therapy (IIA) for slow progression as basic strategies. In patients with rapid progression, EGFR mutation testing should be performed. Osimertinib or platinum-based chemotherapy should be considered as a basic strategy for EGFR T790M-positive patients (grade I evidence), while platinum-based chemotherapy is indicated in T790M-negative patients (grade I evidence). Participation in clinical trials based on the resistance mutation should be considered as an optional strategy for stage IV patients with EGFR-mutant NSCLC after resistance to first-line EGFR TKI [12].
Guidelines for advanced SqCC
According to the 2017 CSCO guidelines, the treatment strategy for SqCC patients should be selected based on the PS score and patient tolerance to platinum. For the first-line treatment of patients with a PS of 0-1 who are suitable for platinum treatment, the basic strategy is platinum-based chemotherapy, including cis-platinum-based chemotherapy (cis-platinum plus gemcitabine/docetaxel/paclitaxel/vinorelbine) (grade I evidence) or carboplatin-based chemotherapy (carboplatin plus gemcitabine/docetaxel/paclitaxel/vinorelbine) (Grade I evidence). The optional strategy is the participation in clinical trials. For patients with a PS of 2 who are not suitable for platinum treatment, single-agent chemotherapy should be the basic strategy, and best support care should be the optional strategy [12].
For the second-line treatment of SqCC patients, single-agent chemotherapy should be the basic strategy (grade I evidence), and afatinib should be the optional strategy (IB).
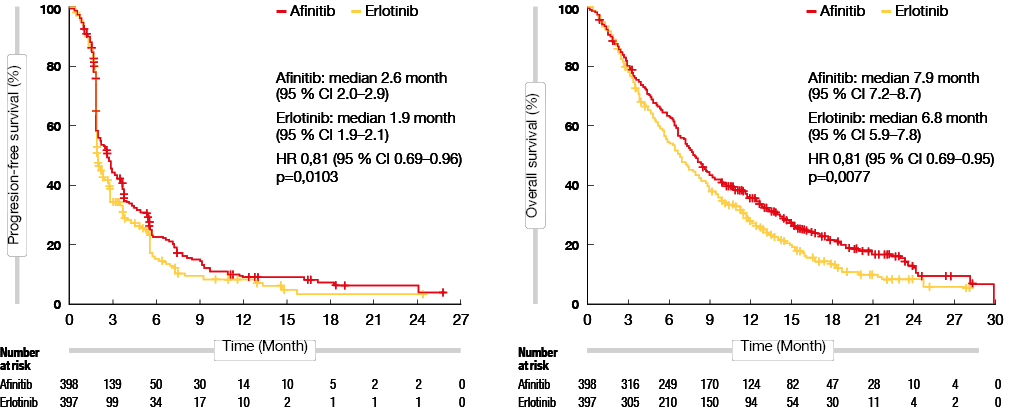
As previously established, SqCC is a heterogeneous disease, and there has hardly been any progress with respect to the treatment of this entity, especially for advanced SqCC. However, in the 2017 CSCO guidelines for LC, the recommendation for the second-line treatment of advanced SqCC was updated. This update was mainly based on evidence from the LUX Lung 8 (LL8) trial. LL8 compared afatinib with erlotinib in advanced SqCC after first-line chemotherapy. Results showed improved PFS (median 2.6 [95 % CI 2.0-2.9] vs. 1.9 months [1.9-2.1]; HR 0.81 [95 % CI 0.69-0.96], p = 0.0103) and OS (median 7.9 months [95 % CI 7.2-8.7] vs. 6.8 months [5.9-7.8]; HR 0.81 [95 % CI 0.69-0.95], p = 0.0077) in the afatinib group compared to the erlotinib group [13] (Figure 2). Thus, CSCO 2017 recommends afatinib as a second-line treatment for advanced SqCC patients. (please see additional information on SqCC here)
Figure 2: PFS (left) and OS (right) with afatinib compared to erlotinib in advanced SqCC
Guidelines for HER2-positive NSCLC: Afatinib as a recommended treatment in China
HER2 is a driver gene identified in NSCLC, with a prevalence of the HER2 mutation of about 1.92 % in China [14]. However, there is no SOC for HER2-mutant NSCLC, which constitutes a problem in clinical practice. The activity of afatinib was demonstrated in preclinical HER2-mutant LC models and in clinical studies with HER2-mutant NSCLC patients. At CSCO 2017, the study design of afatinib for the treatment of HER2-mutant NSCLC patients in China (NCT02597946) was introduced, which was initiated in March 2016 [15].
REFERENCES
- Goldstraw P et al., Non-small-cell lung cancer. Lancet 378:1727-1740.
- Fan Y et al., Guidelines for I-IIIa LC CSCO 2017
- Chang JY et al., Stereotactic ablative radiotherapy versus lobectomy for operable stage I non-small-cell lung cancer: a pooled analysis of two randomised trials. Lancet Oncol. 2015 Jun;16(6):630-7.
- Yerokun BA et al., A national analysis of wedge resection versus stereotactic body radiation therapy for stage IA non-small cell lung cancer. J Thorac Cardiovasc Surg. 2017 Aug; 154 (2):
675-686.e4. - Albain KS et al., Radiotherapy plus chemotherapy with or without surgical resection for stage III non-small-cell lung cancer: a phase III randomised controlled trial. Lancet. 2009 Aug 1; 374(9687): 379-86.
- Jan P et al., Randomized controlled trial of resection versus radiotherapy after induction chemotherapy in stage IIIA-N2 non – small-cell lung cancer. J Natl Cancer Inst 2007; 99: 442-50
- Corso CD et al., Re-evaluation of the role of postoperative radiotherapy and the impact of radiation dose for non-small-cell lung cancer using the National Cancer Database. J Thorac Oncol. 2015 Jan;10 (1):148 – 55.
- Urban D et al., Lymph node ratio may predict the benefit of postoperative radiotherapy in non-small-cell lung cancer. J Thorac Oncol. 2013 Jul; 8 (7):940 – 6. 7.
- Mikell JL et al., Postoperative radiotherapy is associated with better survival in non-small cell lung cancer with involved N2 lymph nodes: results of an analysis of the National Cancer Data Base. J Thorac Oncol. 2015 Mar;10(3):462-71.
- Robinson CG et al., Postoperative radiotherapy for pathologic N2 non–small-cell lung cancer treated with adjuvant chemotherapy: A review of the National Cancer Data Base. J Clin Oncol. 2015 Mar 10; 33(8): 870–876.
- Wisnivesky JP et al., Postoperative radiotherapy for elderly patients with stage III lung cancer. Cancer. 2012 Sep 15;118 (18):4478 – 85.
- Zhefeng L et al., Guidelines for advanced LC therapy CSCO 2017
- Soria JC et al., Afatinib versus erlotinib as second -line treatment of patients with advanced squamous cell carcinoma of the lung (LUX lung 8): an open-label randomised controlled phase 3 trial. Lancet Oncol, 2015, 16 (8):897 – 907.
- Zhangzhou H et al., Mutational subtypes and prognosis of non-small-cell lung cancer harboring HER2 mutations. CSCO 2017
- Caicun Z et al., Afatinib in patients with advanced HER2 mutation-positive NSCLC previously treated with chemotherapy. CSCO 2017
More posts
Determination of clinical responses to immunotherapy
In the past 13 years, an earthshaking change has occurred in treatment of non–small-cell lung cancer (NSCLC). The emergence of two new treatment methods – targeted treatment and immunotherapy – has overturned doctors’ and patients’ perception of standard of care for NSCLC. However, not all of the driver oncogenes in NSCLC have been identified.
New insights into the treatment of ALK-mutant-positive NSCLC patients
Anaplastic lymphoma kinase (ALK) is a fusion oncogene, and the prevalence of ALK mutations in NSCLC patients is similar across different races. At CSCO 2017, the main progress for the treatment of ALK-mutant-positive NSCLC patients related to the new recommendations for first-line and second-line treatments, and the optimal strategies to manage patients before and after resistance to ALK tyrosine kinase inhibitors (TKIs).
Optimal strategy for the treatment of EGFR-mutant lung cancer
The emergence of EGFR TKIs has changed the standard of care in EGFR-mutant NSCLC patients. IPASS was the first open-label randomized study to compare the first-generation EGFR TKI gefitinib with platinum-based chemotherapy in Chinese patients with EGFR-mutant NSCLC. It showed that the disease-free survival (DFS) rate was remarkably higher in the gefitinib group compared to chemotherapy.
Different therapies for treatment of squamous cell carcinoma
Squamous cell carcinoma (SqCC) is one of the histopathological subtypes of non–small-cell lung cancer (NSCLC), and it accounts for 20 % to 30 % of these patients [1]. Unfortunately, few studies have explored the treatment options for patients with SqCC, and progress in SqCC treatment lags behind other histopathological and/or molecular subtypes of NSCLC [2].
Diagnosis of EGFR-mutated NSCLC: from guidelines to reality
Over the last decade, the increasing understanding of critical molecular and cellular mechanisms which drive tumor initiation, maintenance, and progression in non-small-cell lung cancer (NSCLC) have contributed to the discovery of various novel drug targets and the development of new treatment strategies.
Guidelines for the treatment of NSCLC in China: progress and controversies
Lung cancer (LC) is the leading cause of tumor deaths worldwide. Per year, 1.8 million people are diagnosed with LC, and the annual death toll amounts to 1.6 million. Non–small-cell lung cancer (NSCLC) is the most common type, which accounts for 85% of LC cases. Squamous cell carcinoma (SqCC), adenocarcinoma (AC), and large-cell carcinoma are subtypes of NSCLC.