Optimal strategy for the treatment of EGFR-mutant lung cancer
The emergence of EGFR TKIs has changed the standard of care in EGFR-mutant NSCLC patients. IPASS was the first open-label randomized study to compare the first-generation EGFR TKI gefitinib with platinum-based chemotherapy in Chinese patients with EGFR-mutant NSCLC. It showed that the disease-free survival (DFS) rate was remarkably higher in the gefitinib group compared to chemotherapy. Therefore, IPASS laid the foundation for the use of first-generation EGFR TKIs in advanced EGFR-mutant NSCLC [1].
At CSCO 2017, discussions on EGFR-targeted treatment were mainly focused on three topics. First, first-, second- and third-line standard treatment strategies as well as optional treatment strategies for advanced EGFR-mutant NSCLC patients; second, combination of targeted therapy and radiotherapy for EGFR-mutant NSCLC patients; third, latest data and novel insights from studies focused on EGFR-targeted treatment in Chinese NSCLC patients.
Optimal strategy for the treatment of EGFR-mutant positive lung cancer: first-line treatment
In the guidelines on lung cancer (LC) treatment recently published by CSCO, first-generation EGFR TKIs (erlotinib or gefitinib) and the second-generation EGFR TKI afatinib were recommended as the standard first-line treatment in patients with advanced EGFR-mutant NSCLC [2].
In China, afatinib altered the first-line treatment recommendations for patients with stage IV, EGFR-mutant, advanced NSCLC according to the 2017 CSCO guidelines. Data from the LUX-Lung (LL) 3, 6, and 7 trials demonstrated that afatinib can prolong patient PFS and OS and increase objective response rate (ORR) compared to first-generation EGFR TKI treatment or chemotherapy [3, 4]. At CSCO 2017, updated data focusing on Chinese patients were presented. LL6 compared afatinib (n = 217) 40mg/d to 6 cycles of gemcitabine/cisplatin (G/C) (n = 110). PFS was longer for afatinib compared to G/C (median, 11.0 vs. 5.6 months; HR, 0.30; p < 0.0001). In addition, afatinib improved OS vs. G/C in patients with deletion 19 (median, 31.6 vs. 16.3 months; HR, 0.61; p = 0.0146). No unexpected AEs were observed with afatinib treatment in Chinese patients [5]. LL7, which compared afatinib to gefitinib as first-line treatment for EGFR-mutant NSCLC patients, showed that PFS, ORR and time to treatment (TTF) were remarkably higher in the afatinib group than in the gefitinib group. In-depth analyses revealed improved OS (27.9 vs. 24.5 months) and increased TTF (13.7 vs. 11.5 months; p = 0.007) for afatinib compared to gefitinib irrespective of the EGFR mutation type. Safety results showed that afatinib dose reduction decreased the incidence/severity of treatment-related AEs [6,7]. Thus, the authors concluded that EGFR-mutant NSCLC patients can benefit from first-line afatinib treatment with regard to PFS, OS, TTF and safety.
Additionally, long-term survival analyses of LL3, LL6 and LL7 were reported at CSCO 2017. According to these, long-term responders (LTRs; treatment with afatinib for ≥ 3 years) occurred in 10 %, 10 % and 12 % in LL3, LL6 and LL7, respectively. In contrast, the respective number for gefitinib was only 4 % in LL7. Finally, Professor Yi-Long Wu from the Guangdong Lung Cancer Institute, Guangdong General Hospital and Guangdong Academy of Medical Sciences, Guangzhou (China), indicated that afatinib 30mg or 40mg might be the optimal choice for Chinese patients with EGFR-mutant NSCLC [8].
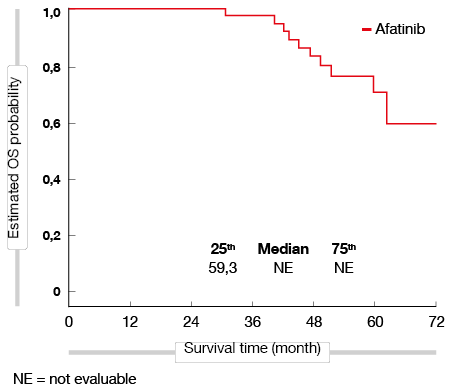
Analyses of different second-line treatments in patients who received first-line afatinib showed promising results for osimertinib: A total of 37 patients who discontinued afatinib received subsequent osimertinib, mostly in the third-line setting and beyond. For these patients, median time on osimertinib in any treatment line was long at 20.2 months, and after a median follow-up of more than 4 years, OS had not yet been reached (Figure 1) [9]. Thus, these findings support that treatment with first-line afatinib, followed by subsequent therapy including osimertinib, may be an optional strategy for patients with EGFR-mutant advanced NSCLC.
Figure 1: Exploratory OS analysis in patients starting on afatinib who received subsequent osimertinib in any line
Osimertinib – upgrade from second-line to first-line treatment?
Osimertinib is a third-generation EGFR TKI that was tested in the studies AURA, AURAext, AURA2, AURA3, AURA17 and AURA18, which showed an ORR of approximately 60 %–70 % and PFS of 9.6–10.1 months in patients with advanced NSCLC who were resistant to first- and second-generation EGFR TKIs. The AURAext and AURA2 studies demonstrated that, once patients have become resistant to first-generation EGFR TKIs, osimertinib may be the optional choice instead of platinum-based chemotherapy for EGFR T790M-positive NSCLC [10]. However, it should be noted that AURAext and AURA2 were single-arm studies, which provided insufficient evidence that osimertinib could be used instead of chemotherapy as second-line treatment in patients who are resistant to first-generation EGFR TKIs. AURA3 was a head-to-head study that compared osimertinib to platinum-pemetrexed as a second-line treatment for patients with EGFR T790M-positive, advanced NSCLC who had previously received EGFR TKI treatment. AURA3 demonstrated improved PFS in the osimertinib treatment group compared to the platinum-pemetrexed group (10.1 vs. 4.4 months; HR, 0.3; p < 0.001) [11]. Thus, the 2017 NCCN guideline suggests that osimertinib should be the standard of care (SOC) as second-line treatment for EGFR T790M-positive advanced NSCLC.
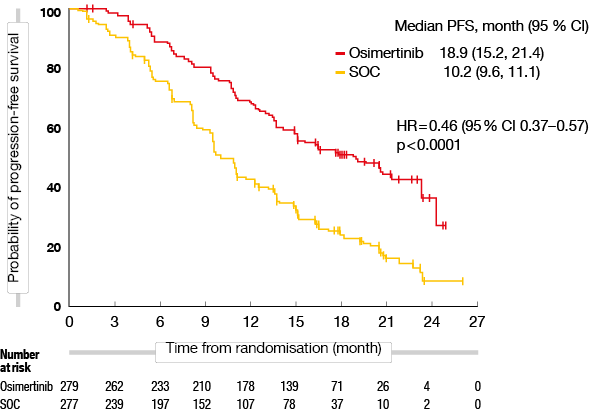
FLAURA was the first head-to-head study that compared osimertinib with first-generation TKIs as first-line treatment for EGFR-mutant (i.e., exon 19 deletion or L858R mutation) advanced NSCLC. Patients were randomized into two groups and were treated with osimertinib (80 mg once daily) or first-generation EGFR TKIs (gefitinib 250 mg or erlotinib 150 mg once daily), respectively. The primary endpoint was disease progression according to RECIST 1.1. Median PFS was significantly longer in the osimertinib group compared to SOC (18.9 vs. 10.2 months; p < 0.0001) (Figure 2). The ORR was slightly higher in the osimertinib group (80 % vs. 76 %; p = 0.2335), and median duration of response (DoR) was also higher with osimertinib (17.2 vs. 8.5 months). Safety profiling indicated that patients in the osimertinib group experienced fewer AEs than those who received SOC. Moreover, the occurrence of serious AEs (grade > 3) was also reduced with osimertinib (34 % vs. 45 %). The interim analysis of OS (data maturity, 25 %) did not reach formal statistical significance with regard to osimertinib superiority, but encouraging data indicated a trend for a survival benefit (HR for death, 0.63) [12]. If osimertinib is chosen as first-line treatment, other hurdles, such as limitations of detection of the T790M mutation, heterogeneous mechanisms of osimertinib resistance and cross-resistance among the first-, second- and third-generation EGFR TKIs, should also be considered. Thus, Professor James Chih-Hsin Yang from the National Taiwan University Hospital, Taipei (Taiwan), indicated that the designation of the third-generation EGFR TKI osimertinib as a first-line treatment is still uncertain and that its efficacy in EGFR-mutant NSCLC is not completely understood yet [13].
Figure 2: PFS with osimertinib compared to first-generation EGFR TKI (gefitinib or erlotinib) as first-line treatment of EGFR-mutant, advanced NSCLC
3.1.3. Dacomitinib in Chinese patients – ARCHER 1050
At ASCO 2017, data from the ARCHER 1050 trial was reported. This trial tested another EGFR-targeted drug, dacomitinib, a second-generation EGFR TKI. In the first-line treatment of EGFR-mutant advanced NSCLC, dacomitinib was associated with longer PFS than the first-generation EGFR TKI (gefitinib) (14.7 vs. 9.2 months; HR, 0.59; p < 0.0001). Interestingly, among the 452 patients who were enrolled in this study, 231 patients were Chinese. Thus, at CSCO 2017, the data was presented with a focus on these patients.
The main inclusion criterion for ARCHER 1050 was advanced NSCLC and the presence of one EGFR mutation (deletion 19 or Leu858Arg mutation). Patients were randomized into two groups and received either oral dacomitinib (45 mg/day, in 28-day cycles) or oral gefitinib (250 mg/day, in 28-day cycles) until disease progression or until another discontinuation criterion was met. The primary endpoint was PFS assessed by masked independent review. In the subgroup analysis of Chinese patients, PFS was significantly higher in the dacomitinib group compared to gefitinib (16 vs. 9.2 months according to masked independent review and 18.4 vs. 11.1 months according to investigator analysis). The ORR in Chinese patients was similar to that in the total population, and DoR was higher for dacomitinib than for gefitinib (15.6 vs. 8.3 months).
The data of ARCHER 1050 showed favorable results for dacomitinib as first-line treatment for EGFR-mutant advanced NSCLC. However, the authors pointed out that even though the data on PFS was promising, it remained unclear whether satisfactory results would be obtained in terms of OS. Furthermore, several other important questions remained unanswered: how effective is dacomitinib for the treatment of CNS metastases in LC patients? As a broad-spectrum TKI, what is the resistance mechanism to dacomitinib? Are there any alternative treatment strategies if patients become resistant to dacomitinib? Thus, further studies investigating the efficacy of dacomitinib as first-line treatment of EGFR-mutant advanced NSCLC are still needed [14].
Third-line treatment of EGFR-mutant NSCLC – ALTER0303
In the 2017 CSCO guidelines for LC treatment, first- and second-line therapies for advanced NSCLC patients were defined as follows: most patients should receive EGFR TKI therapy, anaplastic lymphoma kinase (ALK) TKI treatment or chemotherapy as first-line treatment depending on their driver oncogene status. In case of resistance to first-line treatment (afatinib), patients should receive osimertinib or chemotherapy as second-line treatment. An increasing number of patients have the chance to receive third-line treatment and further therapy. However, to date, there is no SOC for the third-line treatment setting in NSCLC. The ALTER0303 trial provided useful information on how to treat NSCLC patients after failure of second-line treatment.
ALTER0303, a phase III study investigating anlotinib, was a randomized, double-blind, placebo-controlled study. Anlotinib is a multi-targeted TKI that inhibits VEGFR, PDGFR and FGFR, among others. The main inclusion criterion for ALTER0303 was advanced-stage NSCLC in patients who previously failed first- and second-line therapy. Patients were randomized into an anlotinib arm or a placebo arm. The primary endpoint analysis showed that OS was significantly higher in patients treated with anlotinib than in those receiving placebo (9.6 vs. 6.3 months; HR, 0.68 [95 % CI 0.54, 0.87]; p = 0.0018). Also, ORR and DCR analyses showed better outcomes for anlotinib (ORR, 9.18 % vs. 0.7 %; p < 0.0001; DCR, 80.95 % vs. 37.06 %; p < 0.0001). All AEs were predictable, including fatigue, hypertension, dermal toxicity, TSH elevation and hypertriglyceridemia.
Thus, Professor Baohui Han from the Shanghai Chest Hospital, Jiaotong University, Shanghai (China), indicated that additional studies with larger sample sizes will be necessary to evaluate the safety profile of anlotinib in depth. However, considering the current data, anlotinib is expected to become the standard NSCLC treatment in the third-line setting [15].
Treatment strategy after resistance to EGFR TKI
Precision medicine has changed the therapeutic landscape for LC patients. However, as reported based on previous studies including IPASS, First-SIGNAL, WJTOG3405, NEJ002, OPTIMAL, ENSURE, LL3 and LL6, PFS was 8–14 months in EGFR-mutant patients that received EGFR TKIs. This means that acquired drug resistance will ultimately occur after treatment with EGFR TKIs. What is the optimal treatment strategy after the development of EGFR TKI resistance?
Mok TS et al. showed that treatment with osimertinib significantly increased PFS, ORR and DCR compared to platinum-based chemotherapy in patients positive for T790M mutation who were resistant to first-line EGFR TKIs. Patients negative for T790M should be treated differently, according to the type of progression.
The ASPIRATION trial included patients with slow progression after resistance to first-line EGFR TKI treatment. Continued treatment with erlotinib prolonged PFS without the occurrence of new AEs [16]. However, IMPRESS enrolled patients with fast progression, and no benefit from prolonged gefitinib treatment was demonstrated [17]. Local therapy combined with EGFR TKIs also played a central role in the treatment of patients with resistance to first-line EGFR TKI treatment. Weickhardt AJ et al. reported that the PFS ranged from 10 to 13.8 months in patients treated with local therapy plus EGFR TKI.
Besides the most common mutation, i.e. T790M mutation which occurs in 61 %, Piotrowska et al. presented additional strategies for other relatively rare resistance mechanisms, such as the etoposide/platinum (EP) scheme for SCLC transformation (3 %) and chemotherapy or immunotherapy for unidentified resistance mechanisms (21 %) [18].
Thus, the author concluded that patients with the T790M mutation should receive osimertinib, and if patients present with SCLC transformation, the EP strategy should be applied. However, there are no specific treatment strategies in patients with unidentified resistance mechanisms, and additional studies are required [19].
Targeted therapy and radiotherapy in EGFR-mutant lung cancer
Radiotherapy (RT) and concurrent chemoradiotherapy are frequently used in stage I-III NSCLC. At CSCO 2017, results on the combination of radiotherapy and targeted therapy were reported.
The RTOG0617 study compared concurrent chemoradiotherapy with or without targeted treatment in stage IIIA or IIIB NSCLC patients. This trial showed no OS benefit due to the combination. Ongoing studies compare the efficacy of RT plus EGFR TKI to RT alone in locally advanced (LA), EGFR-mutant NSCLC patients. Even though in vitro data revealed promising results favoring the combination of EGFR TKI plus RT [20], the results of the Alliance 31101 and RTOG 1210 clinical trials, which compared RT plus EGFR TKI with RT alone in LA, EGFR-mutant NSCLC patients, were inconclusive.
In contrast, promising results were observed for combination therapies of RT plus EGFR TKIs as first-line and second-line treatment of patients with EGFR-mutant advanced NSCLC. Helena A et al. reported that for EGFR-mutant advanced NSCLC patients with resistance to first-line EGFR TKIs, PFS and OS were 10 and 41 months, respectively, which is an encouraging result in the second-line setting. Another retrospective study, which was reported by Magnuson WJ et al., showed OS outcomes of 46 and 30 months for patients treated with stereotactic radiosurgery (SRS) and whole-brain radiotherapy (WBRT), respectively, followed by EGFR TKI as first-line treatment .
Thus, the authors concluded that in unselected NSCLC patients, the addition of RT to EGFR-targeted therapy did not improve clinical outcomes. The efficacy of the addition of RT to EGFR-targeted therapy was still unclear in LA, oncogene-positive NSCLC patients, and thus further studies will be needed. The efficacy of RT in advanced EGFR-mutant NSCLC patients looks promising but needs confirmation, and a prospective study is required [21].
Different mechanisms, differences in dose determination – lessons learned from developing the third-generation EGFR TKIs in China
The first preclinical experiment with osimertinib, a third generation EGFR TKI, started in 2011. After only 5 years, in August 2016, osimertinib was approved by the China Food and Drug Administration (CFDA) for clinical practice in China. This is in contrast to other new drugs whose approval may take more than 7.5 years. At CSCO 2017, Professor Yi-long Wu talked about lessons learned from the development of avitinib, another third-generation EGFR TKI, in China.
The phase I study evaluating avitinib was an open-label, dose-escalation study. The initial dose was 50mg twice a day, and doses were increased once patients developed partial response (PR). The final results showed no differences in safety across all dose groups, which differs from previous observations showing that the frequency of AEs increases with dose escalation. The highest ORR was observed at a dose of 300mg (52 %), which also indicated favorable pharmacokinetics with regard to avitinib. Thus, these data suggested that an avitinib dose of 300mg should be used for phase 2 studies [22].
REFERENCES
- Mok TS et al., Gefitinib or carboplatin-paclitaxel in pulmonary adenocarcinoma. N Engl J Med. 2009 Sep 3;361(10):947-57.
- Yang N et al., Upgrade of osimertinib to first line treatment? CSCO 2017
- Yang JC et al., Clinical activity of afatinib in patients with advanced non-small-cell lung cancer harbouring uncommon EGFR mutations: a combined post-hoc analysis of LUX-Lung 2, LUX-Lung 3, and LUX-Lung 6. Lancet Oncol. 2015 Jul;16(7):830-8.
- Park K et al., Afatinib versus gefitinib as first-line treatment of patients with EGFR mutation-positive non-small-cell lung cancer (LUX-Lung 7): a phase 2B, open-label, randomised controlled trial. Lancet Oncol. 2016 May;17(5):577-89.
- Xu CR et al., Afatinib vs cisplatin/gemcitabine for the first-line treatment of Chinese patients with advanced EGFR-mutation positive (EGFRm+) NSCLC: subgroup analysis of the LUX-Lung 6 trial. CSCO 2017
- Zhang L et al., Afatinib versus gefitinib in patients with EGFR mutation positive (EGFRm+) NSCLC: updated OS data from LUX-Lung 7. CSCO 2017
- Zhang L et al., Afatinib vs gefitinib for the first-line treatment of EGFR-mutation positive (EGFRm+) NSCLC (LUX-Lung 7): time to treatment failure and impact of afatinib dose adjustment. CSCO 2017
- Wu YL et al., Long-term response to first-line afatinib in patients with advanced EGFR mutation-positive (EGFRm+) NSCLC: analysis of LUX-Lung (LL) 3, 6 and 7. CSCO 2017
- Sequist L et al., Subsequent therapies post-afatinib among patients with EGFR mutation positive NSCLC in LUX-Lung (LL) 3, 6, 7. CSCO 2017
- Goss G et al., Osimertinib for pretreated EGFR Thr790Met-positive advanced non-small-cell lung cancer (AURA2): a multicentre, open-label, single-arm, phase 2 study. Lancet Oncol. 2016 Dec;17(12):1643-1652.
- Mok TS et al., Osimertinib or platinum-pemetrexed in EGFR T790M-positive lung cancer. N Engl J Med. 2017 Feb 16;376(7):629-640.
- Soria JC et al., Osimertinib in untreated EGFR-mutated advanced non–small-cell lung cancer. N Engl J Med. 2018 Jan 11;378(2):113-125. doi: 10.1056/NEJMoa1713137.
- Yang JC et al., Optimal strategy for treatment of EGFR mutation positive lung cancer Choice and Sequence. CSCO 2017
- Zhou Q et al., Dacomitinib vs gefitinib for first-line (1L) treatment of advanced EGFR+ non-small-cell lung cancer (NSCLC) in Chinese patients (ARCHER 1050). CSCO 2017
- Han B et al., Third-line treatment: A randomized, double-blind, placebo controlled phase III ALTER-0303 study—Efficacy and safety of anlotinib treatment in patients with refractory advanced NSCLC. CSCO 2017
- Park K et al., First-line erlotinib therapy until and beyond response evaluation criteria in solid tumors progression in Asian patients with epidermal growth factor receptor mutation-positive non-small-cell lung cancer: the ASPIRATION study. JAMA Oncol. 2016 Mar;2(3):305-12.
- Soria JC et al., Gefitinib plus chemotherapy versus placebo plus chemotherapy in EGFR-mutation-positive non-small-cell lung cancer after progression on first-line gefitinib (IMPRESS): a phase 3 randomised trial. Lancet Oncol. 2015 Aug;16(8):990-8.
- Piotrowska Z et al., Treatment of EGFR-mutant lung cancers after progression in patients receiving first-line EGFR tyrosine kinase inhibitors: a review. JAMA Oncol. 2016 Jul 1;2(7):948-54
- Zhao J et al., Treatment strategy after resistance in driver oncogene positive NCLCL. CSCO 2017
- Baumann M et al., Selective inhibition of the epidermal growth factor receptor tyrosine kinase by BIBX1382BS and the improvement of growth delay, but not local control, after fractionated irradiation in human FaDu squamous cell carcinoma in the nude mouse. Int J Radiat Biol. 2003 Jul;79(7):547-59.
- Fu X et al., Radiotherapy and target medicine for LC. CSCO 2017
- Wu YL et al., Lessons learned from developing the 3rd Generation EGFR inhibitors in China. CSCO 2017
More posts
Determination of clinical responses to immunotherapy
In the past 13 years, an earthshaking change has occurred in treatment of non–small-cell lung cancer (NSCLC). The emergence of two new treatment methods – targeted treatment and immunotherapy – has overturned doctors’ and patients’ perception of standard of care for NSCLC. However, not all of the driver oncogenes in NSCLC have been identified.
New insights into the treatment of ALK-mutant-positive NSCLC patients
Anaplastic lymphoma kinase (ALK) is a fusion oncogene, and the prevalence of ALK mutations in NSCLC patients is similar across different races. At CSCO 2017, the main progress for the treatment of ALK-mutant-positive NSCLC patients related to the new recommendations for first-line and second-line treatments, and the optimal strategies to manage patients before and after resistance to ALK tyrosine kinase inhibitors (TKIs).
Optimal strategy for the treatment of EGFR-mutant lung cancer
The emergence of EGFR TKIs has changed the standard of care in EGFR-mutant NSCLC patients. IPASS was the first open-label randomized study to compare the first-generation EGFR TKI gefitinib with platinum-based chemotherapy in Chinese patients with EGFR-mutant NSCLC. It showed that the disease-free survival (DFS) rate was remarkably higher in the gefitinib group compared to chemotherapy.
Different therapies for treatment of squamous cell carcinoma
Squamous cell carcinoma (SqCC) is one of the histopathological subtypes of non–small-cell lung cancer (NSCLC), and it accounts for 20 % to 30 % of these patients [1]. Unfortunately, few studies have explored the treatment options for patients with SqCC, and progress in SqCC treatment lags behind other histopathological and/or molecular subtypes of NSCLC [2].
Diagnosis of EGFR-mutated NSCLC: from guidelines to reality
Over the last decade, the increasing understanding of critical molecular and cellular mechanisms which drive tumor initiation, maintenance, and progression in non-small-cell lung cancer (NSCLC) have contributed to the discovery of various novel drug targets and the development of new treatment strategies.
Guidelines for the treatment of NSCLC in China: progress and controversies
Lung cancer (LC) is the leading cause of tumor deaths worldwide. Per year, 1.8 million people are diagnosed with LC, and the annual death toll amounts to 1.6 million. Non–small-cell lung cancer (NSCLC) is the most common type, which accounts for 85% of LC cases. Squamous cell carcinoma (SqCC), adenocarcinoma (AC), and large-cell carcinoma are subtypes of NSCLC.