Extensive-disease small-cell tumors: signals of activity
CheckMate 451: immunotherapeutic maintenance
Most of the patients with small-cell lung cancer (SCLC) are diagnosed in the extensive-disease stage (ED-SCLC). They generally respond well to first-line platinum-based chemotherapy; however, responses are not durable, and prognosis is poor [1, 2]. In the second line and beyond, the NCCN guidelines recommend patient inclusion in a clinical trial, systemic therapy depending on the patient performance status and the duration of the relapse-free interval, or palliative symptom management [1]. No maintenance treatment is currently available to prolong the effects obtained with first-line chemotherapy. The CheckMate 451 trial was designed to assess an immunotherapeutic approach in this setting [3]. A total of 834 patients with ED-SCLC who had ongoing responses in the form of CR, PR or SD following 4 cycles of platinum-based first-line chemotherapy were randomized to either nivolumab plus ipilimumab (n = 279), nivolumab 240 mg Q2W (n = 280), or placebo (n = 275). In the combination arm, nivolumab was administered in a weight-based manner (1 mg/kg Q3W) together with ipilimumab 3 mg/kg Q3W for a maximum of 4 doses, followed by nivolumab 240 mg Q2W. The treatment continued until progression or unacceptable toxicity for a maximum of 2 years. CheckMate 451 failed to show a significant OS improvement for nivolumab plus ipilimumab compared to placebo, which was defined as the primary endpoint (median OS, 9.2 vs. 9.6 months; HR, 0.92). This was also true for the landmark analysis at 12 months (41 % vs. 40 %).
Benefit after short treatment-free interval
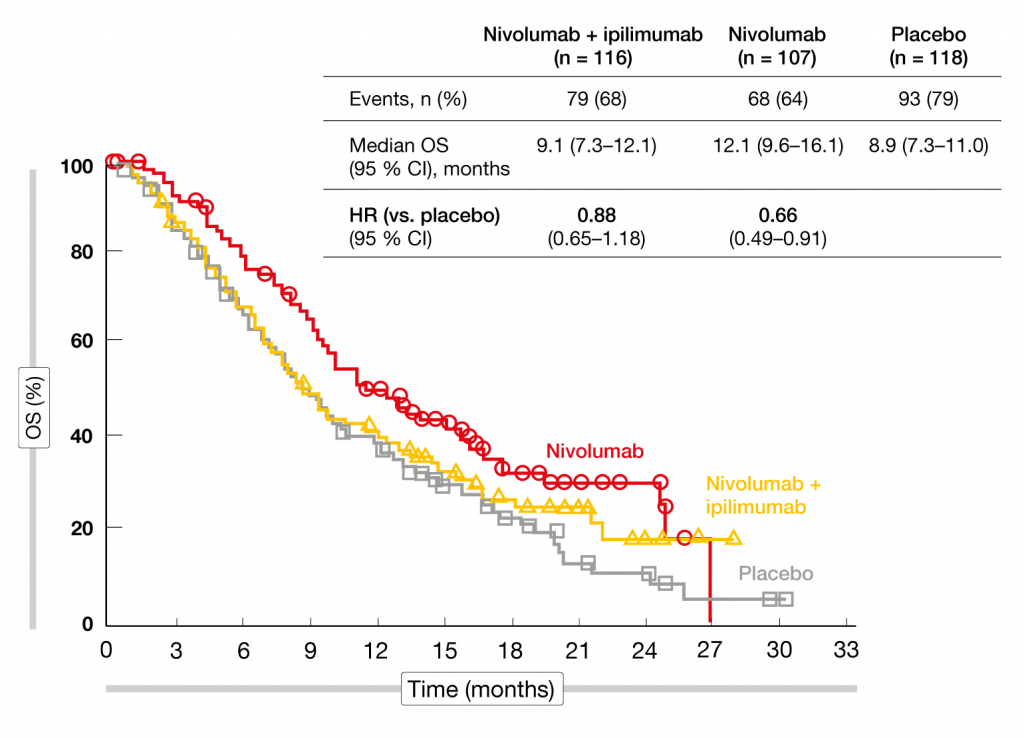
Due to the hierarchical testing requirement of the study, the other endpoints were not tested for statistical significance but only analyzed for descriptive purposes. With nivolumab monotherapy vs. placebo, as for the combination, there was no OS difference in the total population (10.4 vs. 9.6 months; HR, 0.84). However, the subgroup analysis implied a significant benefit compared to placebo in patients who started immunotherapy within 5 weeks after their last doses of frontline chemotherapy (median OS, 12.1 vs. 8.9 months; HR, 0.66; Figure). Nivolumab plus ipilimumab did not elicit any OS advantage in this cohort (HR, 0.88). Patients who received immunotherapy after an interval of more than 5 weeks, on the other hand, showed no survival benefit with either nivolumab alone or the combination (HR for both, 0.96). The PFS analysis hinted at improved outcomes compared to placebo with both nivolumab plus ipilimumab (HR, 0.72; 6-month PFS rates, 20 % vs. 10 %) and nivolumab alone (HR, 0.67; 6-month PFS rates, 21 % vs. 10 %). Likewise, responses were more favorable in the experimental arms. Clinical benefits (i.e., CR plus PR plus SD) occurred in 45 %, 47 %, and 35 % of patients treated with the combination, nivolumab, and placebo, respectively. Median duration of response was 10, 11, and 8 months, respectively. The safety profiles of the immunotherapeutic regimens corresponded to previous reports for the same doses and schedules. Nivolumab monotherapy proved more tolerable than the combination. The authors concluded that PFS and response rate data suggest activity of immunotherapy in the maintenance setting of ED-SCLC. Nivolumab might provide improved OS in patients with a shorter interval from the last dose of first-line chemotherapy to initiation of treatment.
Figure: Reduction in mortality with nivolumab vs. placebo in patients who started immunotherapy within 5 weeks after frontline chemotherapy
Rova-T: third-line setting
To date, no drug has been approved for the third-line treatment of ED-SCLC. The antibody-drug conjugate rovalpituzumab tesirine (Rova-T) targets the delta-like protein 3 (DLL3), which is highly expressed in SCLC and neuroendocrine carcinoma (NEC) [4]. Single-agent Rova-T has shown encouraging anti-tumor activity and a manageable safety profile in SCLC patients treated in phase I and II studies [5, 6]. A retrospective analysis presented at ELCC 2019 evaluated DLL3 test results and clinical experience with Rova-T in patients after failure of at least two cycles of systemic treatment in a real-life setting [7]. DLL3 immunohistochemistry was performed in 68 patients with high-grade NEC including 61 SCLC and 7 large-cell NEC cases. Most of the samples showed DLL3 staining. Forty-nine specimens (72.1 %) were classified as highly positive, 10 (14.7 %) as positive, and 9 (13.2 %) as negative. Sixteen patients who had no other treatment options left received at least one of two planned cycles of Rova-T 0.3 mg/kg. In this group, 2 patients were DLL3-negative, 4 were DLL3-positive, and 10 were highly DLL3-positive. Both treatment cycles were administered in 7 patients, while 9 received only one cycle due to disease progression or the occurrence of AEs. Four patients (25 %) experienced partial responses, while 4 (25 %) had stable disease, and 8 developed disease progression (50 %). Common drug-related AEs included fatigue, photosensitivity, pleural effusion, peripheral edema, and thrombocytopenia. AEs were generally manageable. The authors concluded that Rova-T gave rise to clinical benefit in selected patients and therefore appears to be an option for ED-SCLC in later lines. A large proportion of SCLC patients tested DLL3-positive, although further studies are needed to assess the feasibility of DLL3 as a biomarker in this setting.
REFERENCES
- National Comprehensive Cancer Network. Small Cell Lung Cancer. V 1.2019
- Hurwitz JL et al., New advances in the second-line treatment of small cell lung cancer. Oncologist 2009; 14(10): 986-994
- Owonikoko TK et al., Nivolumab plus ipilimumab, nivolumab, or placebo as maintenance therapy in patients with extensive disease small cell lung cancer after first-line platinum-based chemotherapy: results from the double-blind, randomized phase 3 CheckMate 451 study. ELCC 2019, abstract LBA1_PR
- Deng SM et al., The Notch ligand delta-like 3 promotes tumor growth and inhibits Notch signaling in lung cancer cells in mice. Biochem Biophys Res Commun 2017; 483(1): 488-494
- Carbone DP et al., Efficacy and safety of rovalpituzumab tesirine in patients with DLL3-expressing, ≥ 3rd line small cell lung cancer: results from the phase 2 TRINITY study. J Clin Oncol 2018; 36 (suppl; abstr 8507)
- Lashari BH et al., Rovalpituzumab tesirine: a novel DLL3-targeting antibody-drug conjugate. Drugs R D. 2018; 18(4): 255-258
- Hochmair M et al., Real-life experience with the implementation of DLL3 staining and the subsequent treatment with rovalpituzumab tesirine in heavily pretreated SLC patients. ELCC 2019, abstract 63P