Small-cell lung cancer: established and novel approaches
Small-cell lung cancer (SCLC) accounts for 14 % of all lung cancers. It is initially sensitive to chemotherapy and radiation therapy, but resistance tends to develop rapidly, which leads to high recurrence rates. Outcomes with second-line treatments are poor, with 5-year survival rates of only 5 %. Topotecan is the only approved agent in the second-line setting.
The phase I/II CheckMate 032 trial evaluated nivolumab as monotherapy and in combination in various tumour types, including SCLC. The updated results for the SCLC cohort that were presented at the ECC showed that both nivolumab monotherapy and the combination of nivolumab and ipilimumab have anti-tumour activity in patients who have progressed after at least one prior therapy, including a platinum-based regimen as first line [1]. The patients were unselected in terms of PD-L1 expression. Nivolumab monotherapy was administered at a dose of 3 mg/kg very 2 weeks (n = 80). The combination was applied every three weeks for 4 cycles at three different doses (nivolumab 1 mg/kg plus ipilimumab 1 mg/kg [n = 3]; nivolumab 1 mg/kg plus ipilimumab 3 mg/kg [n = 47]; nivolumab 3 mg/kg plus ipilimumab 1 mg/kg [n = 53]). Approximately one third of patients was platinum-resistant/ refractory.
Benefits irrespective of platinum sensitivity
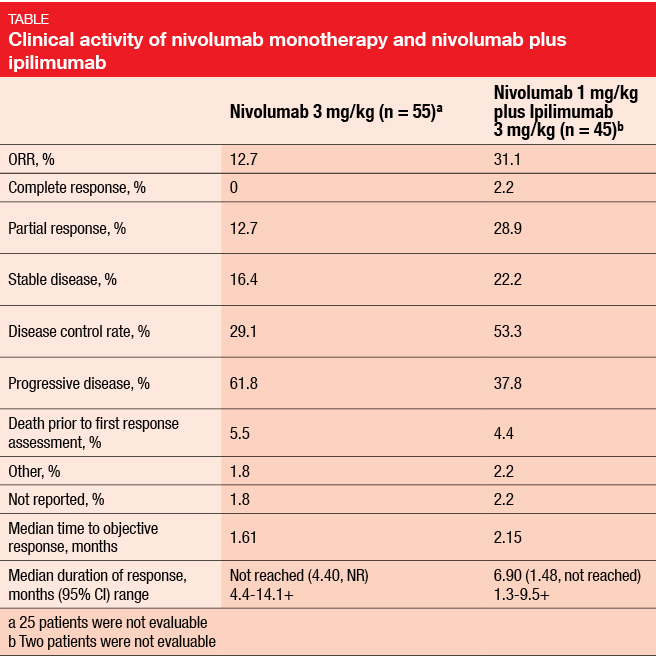
Responses were durable and occurred early on. The ORR, which was defined as the primary outcome, was 12.7 % in the nivolumab monotherapy arm and 31.1 % in the nivolumab 1 mg/kg plus ipilimumab 3 mg/kg arm (Table). Objective responses were observed in second-line patients with both platinum-sensitive and platinum-resistant disease. Median OS were 3.55 months and 7.75 months, with 27.1 % and 47.5 % of patients alive at 1 year. For PFS, median estimates were 1.38 months and 3.35 months for the two regimens. The 9-month PFS rates were 10.2 % and 30.4 %. In both treatment arms, tumour responses were observed in patients with < 1 % and ≥ 1 % PD-L1 expression, according to the preliminary analysis.
Treatment-related AEs occurred more frequently with the combination regimen. Grade 3–4 AEs were seen in 11.3 % of patients in the nivolumab monotherapy arm and 31.9 % of patients in the nivolumab 1 mg/kg plus ipilimumab 3 mg/kg arm. Fatigue and diarrhoea constituted the most frequent AEs in both groups. Grade-2 limbic encephalitis occurred in two patients, which resolved with immunosuppressive treatment. One patient had grade 4 limbic encephalitis, which did not resolve with immunosuppressive treatment. The authors therefore recommended close monitoring for early signs or symptoms of paraneoplastic syndromes (e. g., limbic encephalitis) and autoimmune disease (e. g., myasthenia gravis). Pneumonitis was diagnosed in two patients in the monotherapy arm and in one patient in the combination arm. The management of toxicity followed established safety guidelines.
Phase III studies in SCLC patients with extensive stage disease in the first line and second line are presently being initiated; CheckMate 331 is assessing nivolumab versus chemotherapy in relapsed SCLC, and CheckMate 451 is testing nivolumab alone and in combination with ipilimumab as maintenance therapy after platinum-based first-line therapy in SCLC.
Rova-T: single-agent activity of an antibody drug conjugate
To date, no targeted therapy has shown proven benefit in patients with SCLC. Encouraging phase I data are now available for rovalpituzumab tesirine (Rova-T), a delta-like protein (DLL)3-targeted antibody drug conjugate [2]. DLL3, which is overexpressed in SCLC tumour-initiating cells, is directly targeted by the humanised monoclonal antibody present in Rova-T.
This trial included 73 patients with relapsed and refractory SCLC who had experienced disease progression after first-line or second-line treatment. Escalating doses of Rova-T were administered once every 3 weeks. Confirmed responses were noted at 0.2 mg/kg, 0.3 mg/kg and 0.4 mg/kg. Subsequently, the phase Ib expansion cohorts received 0.2 mg/kg every 3 weeks or 0.3 mg/kg every 6 weeks.
In the group of patients evaluable for response assessment (n = 53), a total of 23 % achieved ORR, with a clinical benefit rate (CBR) of 68 %. Forty-nine samples were obtained for the assessment of DLL3 expression, which was high in approximately 70 % of patients. In this group, ORR was 44 % and CBR 78 %. Importantly, responses were similar regardless of whether Rova-T was administered as second line or third line. Patients experiencing stable disease, on the other hand, showed variable DLL3 expression.
These responses were durable. At the 0.3 mg/kg every 6 weeks dosing schedule, patients had an ongoing response for 189 days after their confirmatory computed tomography. The survival remained prolonged in these patients. Therefore, the 0.3 mg/kg every 6 weeks schedule was chosen as the randomised phase II dose.
Rova-T showed a manageable safety profile. Toxicity was comparable between the two dosing cohorts. Fatigue occurred most frequently, at 28 %, followed by peripheral oedema, rash, thrombocytopenia, pleural effusion, and nausea. Also, photosensitivity reactions occurred in 12 %. Overall, the benefits achieved with Rova-T are exceptional in the second-line and third-line SCLC setting. These results support biomarker-guided phase II studies, as DLL3 might be the first predictive biomarker associated with drug efficacy in SCLC.
REFERENCES
- Calvo E et al., Nivolumab monotherapy or in combination with ipilimumab for treatment of recurrent small cell lung cancer (SCLC). ECC 2015, abstract 3098
- Pietanza MC et al., Phase I study of a DLL3-targeted antibody drug conjugate, rovalpituzumab tesirine, in patients with relapsed and refractory small cell lung cancer (SCLC). ECC 2015, abstract 7LBA