Checkpoint inhibition in non-small-cell lung cancer: expanding the range of options
IMpower110: interim OS findings
The randomized phase III IMpower110 trial is evaluating the PD-L1 inhibitor atezolizumab as first-line treatment in patients with stage IV, PD-L1–positive non-small-cell lung cancer (NSCLC) independent of tumor histology. Patients in the experimental arm are treated with atezolizumab until disease progression, while the platinum-based chemotherapy regimens administered in the control arm for 4 to 6 cycles depend on histology. Patients were stratified according to their PD-L1 expression on tumor-infiltrating immune cells (IC1/2/3) and tumor cells (TC1/2/3). Approximately one third showed the highest PD-L1 expression (TC3 or IC3).
At the ESMO 2019 Congress, Spigel et al. reported the interim results for overall survival (OS) in the wildtype population, i.e., the patients without EGFR and/or ALK aberrations, which was defined as the primary endpoint [1]. OS assessments followed hierarchical testing, with the analysis of the TC3 or IC3 subgroup preceding evaluation of the TC2/3 or IC2/3 group, which again preceded the assessment of the TC1/2/3 or IC1/2/3 population (i.e., the total population).
Benefits across histologies
Patients in the TC3 or IC3 subgroup (n = 205) derived clinically meaningful benefits from atezolizumab compared to chemotherapy. Median OS was 20.2 vs. 13.1 months (HR, 0.59; p = 0.0106; Figure 1), and survival rates at 12 months amounted to 64.9 % vs. 50.6 %. Objective responses occurred in 38.3 % and 28.6 %, respectively. While the median duration of response had not yet been reached in the experimental arm, it was 6.7 months in the control arm. In the TC2/3 or IC2/3 subgroup (n = 328), the pre-specified OS boundary had not been crossed at the time of the analysis, although the results favored atezolizumab (18.2 vs. 14.9 months). Therefore, the TC1/2/3 or IC1/2/3 population (n = 554) was not formally tested.
Likewise, progression-free survival (PFS) will only be formally assessed once the primary endpoint is positive for all three populations. The current analysis showed median PFS of 8.1 vs. 5.0 months in the TC3 or IC3 subgroup (HR, 0.63) as well as superiority of the atezolizumab treatment for PFS in the TC2/3 or IC2/3 subgroup and the total population. Response rates did not differ across treatment arms in the TC2/3 or IC2/3 and TC1/2/3 or IC1/2/3 populations. Additional biomarker analyses will be presented at a future congress. The authors noted that atezolizumab represents a promising first-line treatment option in NSCLC patients with high PD-L1 expression.
Figure 1: Overall survival benefit with atezolizumab compared to chemotherapy in the TC3 or IC3 subgroup of the IMpower110 trial
Final analysis of B-F1RST
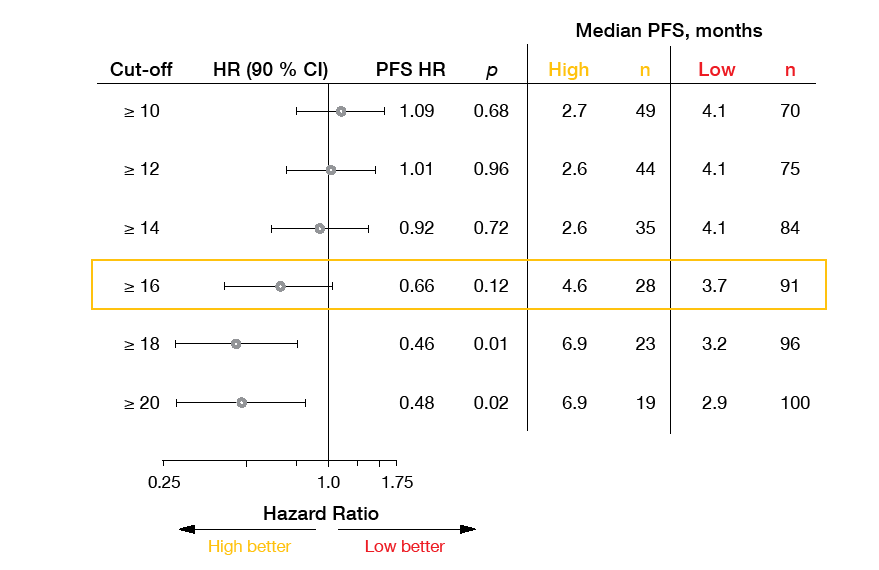
The phase II B-F1RST trial was the first prospective evaluation of blood tumor mutational burden (bTMB) as a predictive biomarker for patients with metastatic NSCLC receiving first-line atezolizumab monotherapy. According to the primary analysis presented in 2018 [2], the pre-specified bTMB cutoff score of 16 correlated with numerical clinical benefits. Socinski et al. presented the final analysis of the B-F1RST trial after a follow-up period of ≥ 18 months at ESMO 2019 [3].
These data showed that single-agent atezolizumab provides outcome improvements in a real-world, unselected population. Objective response rates (ORRs) in the intent-to-treat (n = 152) and biomarker-evaluable (n = 119) populations were 17.1 % and 12.6 %, respectively. Median duration of response in the ITT population was 16.3 months. Median OS will continue to be followed. Also, the analysis corroborated the predictive potential of the bTMB cutoff score of 16. This applied to PFS (5.0 vs. 3.5 months for bTMB-high vs. bTMB-low cohorts; HR, 0.80; p = 0.35), ORR (35.7 % vs. 5.5 %; p < 0.0001) and OS (23.9 vs. 13.4 months; HR, 0.66; p = 0.18). Exploratory analyses demonstrated improved clinical benefit with increasing bTMB cutoff.
The scientists concluded that a blood-based assay can measure TMB in the absence of adequate tissue and predict numerical PFS and OS benefits if a chemotherapy-free first-line option is wished for. Another exploratory endpoint of the trial was serum C-reactive protein (CRP), which was measured at baseline and on day 1 of cycle 3 while on atezolizumab treatment. Decreases in CRP during this time were shown to be associated with improved OS.
TMB as a biomarker: pembrolizumab monotherapy …
Based on the observation that a relationship between high TMB levels and improved OS has not been unequivocally established for treatment with the PD-1 inhibitor pembrolizumab, Herbst et al. assessed the predictive power of tissue (tTMB) according to whole exome sequencing in the open-label KEYNOTE-010 and KEYNOTE-042 trials [4]. These had tested pembrolizumab monotherapy against chemotherapy in the second-line and first-line settings, respectively, in patients with PD-L1–expressing (TPS ≥ 1 %) advanced NSCLC [5, 6].
In KEYNOTE-010, the tTMB-evaluable population comprised 253 individuals. According to the exploratory analysis, tTMB was associated with OS, PFS and ORR for pembrolizumab as a continuous variable, but not with chemotherapy. The phase III KEYNOTE-042 trial included 793 tTMB-evaluable patients. As for KEYNOTE-010, this analysis demonstrated an association of tTMB with outcomes for pembrolizumab as a continuous variable, but not with response to chemotherapy in general. In both trials, the samples of tTMB-evaluable patients were deemed representative due to comparability of baseline characteristics and outcomes across these cohorts and the overall populations.
For both KEYNOTE-010 and -042, the tTMB cutpoint of ≥ 175 mutations per exome showed clinical utility. This cutpoint provided a distinct separation for the results obtained with pembrolizumab versus chemotherapy with respect to OS, PFS, and ORR (Figure 2). Neither analysis revealed a relationship between tTMB and PD-L1 expression for pembrolizumab or chemotherapy. In their conclusion, the authors noted that tTMB may provide additional information regarding the clinical benefit of pembrolizumab monotherapy in patients with PD-L1–positive advanced NSCLC in the first-line and previously treated settings. This might represent another step in the process of personalization of immunotherapy.
Figure 2: Clinical utility of the tissue tumor mutation burden cutpoint of 175 mutations per exome for objective response rates in KEYNOTE-010
… and pembrolizumab-chemotherapy combination
Different results were reported for another exploratory analysis that related to the association of tTMB with the outcomes obtained in patients who received pembrolizumab together with chemotherapy. Data were obtained from the KEYNOTE-21 cohorts C and G, as well as the KEYNOTE-189 and -407 trials [7]. All of these had been conducted in the first-line setting and had tested pembrolizumab plus platinum-based chemotherapy. Cohort C of the KEYNOTE-21 study assessed two different pembrolizumab doses [8], while Cohort G [9, 10], KEYNOTE-189 [11] and KEYNOTE-407 [12] contained chemotherapy-only control arms. Patients included in KEYNOTE-407 had squamous histology, whereas all of the other trial participants had been diagnosed with non-squamous NSCLC. Whole exome sequencing was used to quantify tTMB. Overall, 675 tTMB-evaluable patients representative of the total populations of their respective trials provided data for this analysis.
None of the studies showed a significant association between tTMB and the efficacy of pembrolizumab plus chemotherapy or platinum-based chemotherapy alone. OS, PFS, and ORR benefits occurred with pembrolizumab plus histology-specific chemotherapy in both tTMB-high and tTMB-low subgroups. Again, there was no relationship between tTMB and PD-L1 expression. These data suggest that tTMB has limited clinical utility in the setting of first-line pembrolizumab plus platinum-based chemotherapy for both metastatic squamous and non-squamous NSCLC.
Durable improvements in KEYNOTE-407
As noted above, the phase III KEYNOTE-407 trial investigated pembrolizumab plus chemotherapy compared to chemotherapy alone in patients with advanced, previously untreated metastatic squamous NSCLC. The addition of the PD-1 inhibitor has been shown to induce significant OS and PFS benefits [12]. According to the protocol-specified final analysis reported at ESMO 2019, pembrolizumab plus carboplatin and paclitaxel/nab-paclitaxel continued to demonstrate improved outcomes compared to chemotherapy [13]. After a median follow-up of 14.3 months, median OS was 17.1 vs. 11.6 months (HR, 0.71), and median PFS amounted to 8.0 vs. 5.1 months (HR, 0.57). Responses differed by 24.1 % (62.6 % vs. 38.4 %), and they lasted much longer in the combination arm (8.8 vs. 4.9 months). The results were consistent across PD-L1 expression groups, including in patients with PD-L1 TPS < 1 %.
Moreover, the authors estimated PFS2, which is defined as the time from randomization to objective tumor progression on next-line treatment or death from any cause, whichever occurs first. Here, the analysis demonstrated substantial improvement for patients treated with pembrolizumab (13.8 vs. 9.1 months; HR, 0.59). Overall, the KEYNOTE-407 findings continue to support pembrolizumab plus carboplatin and paclitaxel or nab-paclitaxel as a standard-of-care first-line regimen in patients with metastatic squamous NSCLC, regardless of PD-L1 expression status.
CheckMate 227: a potential first-line option
The first-line combination of the PD-1 inhibitor nivolumab and the anti-CTLA-4 antibody ipilimumab was investigated by the randomized, open-label, phase III CheckMate 227 study conducted in patients with advanced NSCLC who showed PD-L1 expression of ≥ 1 % (Part 1a) or < 1 % (Part 1b). In Part 1a, patients were randomized to either nivolumab plus low-dose ipilimumab, chemotherapy, or single-agent nivolumab. Part 1b compared nivolumab plus low-dose ipilimumab with chemotherapy and with nivolumab plus chemotherapy. CheckMate 227 had two independent co-primary endpoints that included PFS in the TMB-high (i. e., ≥ 10 mut/Mb) population on one hand and OS in the PD-L1 ≥ 1 % population on the other; both comparisons related to nivolumab plus ipilimumab versus chemotherapy.
Peters et al. presented the final results for the OS endpoint at the conference [14]. Irrespective of PD-L1 expression, 583 patients had been randomized to the combination and to chemotherapy each. In Part 1a, 396 individuals had received nivolumab monotherapy.
Successful dual approach
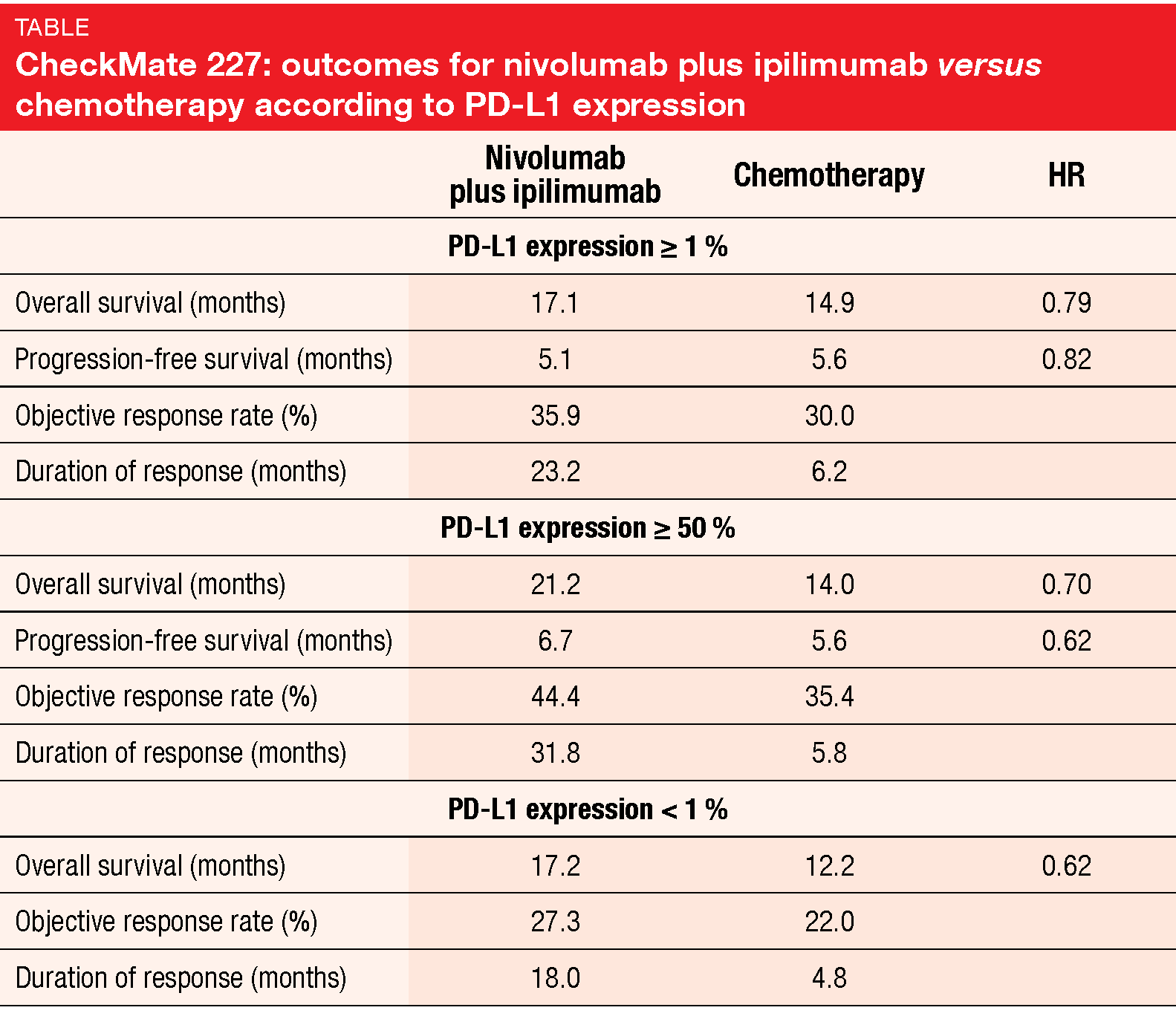
CheckMate 227 met its primary endpoint of OS in patients with PD-L1 ≥ 1 % and is the first phase III study to demonstrate that PD-1 plus CTLA-4 inhibition is effective in NSCLC. Median OS was 17.1 vs. 14.9 months with nivolumab plus ipilimumab and chemotherapy, respectively (HR, 0.79; p = 0.007; Table). At 24 months, 40 % vs. 33 % of patients were alive. The risk of progression and death decreased by 18 % (HR, 0.82), with 22 % vs. 7 % remaining progression-free at 24 months. Nivolumab plus ipilimumab gave rise to deep and durable responses. ORRs amounted to 35.9 % vs. 30.0 %, and responses lasted longer in the combination arm (23.2 vs. 6.2 months).
Clinically meaningful survival improvement was observed with the combination regimen compared to chemotherapy regardless of PD-L1 expression, as both patients with scores ≥ 50 % and < 1 % benefited (Table). In all randomized patients, median OS was 17.1 vs. 13.9 months for nivolumab plus ipilimumab and chemotherapy, respectively (HR, 0.73). No consistent correlation existed between survival outcomes and PD-L1 expression or TMB alone or in combination. The CheckMate 227 trial revealed no new safety signals for nivolumab plus low-dose ipilimumab. In their conclusion, the authors pointed out that this dual immunotherapeutic regimen represents a potential new first-line option for patients with advanced NSCLC.
REFERENCES
- Spigel DR et al., IMpower110: interim OS analysis of a phase III study of atezolizumab vs platinum-based chemotherapy as 1L treatment in PD-L1–selected NSCLC. ESMO 2019, abstract LBA78
- Kim ES et al., Primary efficacy results from B-F1RST, a prospective phase II trial evaluating blood-based tumour mutational burden as a predictive biomarker for atezolizumab in 1L non-small cell lung cancer (NSCLC). ESMO 2018, abstract LBA55
- Socinski MA et al., Final efficacy results from B-F1RST, a prospective phase II trial evaluating blood-based tumor mutational burden as a predictive biomarker for atezolizumab in first-line non-small cell lung cancer. ESMO 2019, abstract LBA83
- Herbst RS et al., Association between tissue TMB and clinical outcomes with pembrolizumab monotherapy in PD-L1-positive advanced NSCLC in the KEYNOTE-010 and 042 trials. ESMO 2019, abstract LBA79
- Herbst RS et al., Pembrolizumab versus docetaxel for previously treated, PD-L1-positive, advanced non-small-cell lung cancer (KEYNOTE-010): a randomised controlled trial. Lancet 2016; 387(10027): 1540-1550
- Mok TSK et al., Pembrolizumab versus chemotherapy for previously untreated, PD-L1-expressing, locally advanced or metastatic non-small-cell lung cancer (KEYNOTE-042): a randomised, open-label, controlled, phase 3 trial. Lancet Oncol 2019; 393(10183): 1819-1830
- Paz-Ares L et al., Pembrolizumab plus platinum-based chemotherapy for metastatic NSCLC: tissue TMB and outcomes in KEYNOTE-021, 189, and 407. ESMO 2019, abstract LBA80
- Gageel SM et al., Pembrolizumab and platinum-based chemotherapy as first-line therapy for advanced non-small-cell lung cancer: Phase 1 cohorts from the KEYNOTE-021 study. Lung Cancer 2018; 125: 273-281
- Langer CJ et al., Carboplatin and pemetrexed with or without pembrolizumab for advanced, non-squamous non-small-cell lung cancer: a randomised, phase 2 cohort of the open-label KEYNOTE-021 study. Lancet Oncol 2016; 17(11): 1497-1508
- Borghaei H et al., 24-month overall survival from KEYNOTE-021 cohort G: pemetrexed and carboplatin with or without pembrolizumab as first-line therapy for advanced non-squamous non-small cell lung cancer. J Thorac Oncol 2019; 14(1): 124-129
- Gandhi L et al., Pembrolizumab plus chemotherapy in metastatic non-small-cell lung cancer. N Engl J Med 2018; 378(22): 2078-2092
- Paz-Ares et al., Pembrolizumab plus chemotherapy for squamous non-small-cell lung cancer. N Engl J Med 2018; 379(21): 2040-2051
- Paz-Ares L et al., Pembrolizumab plus chemotherapy in metastatic squamous NSCLC: final analysis and progression after the next line of therapy (PFS2) in KEYNOTE-407. ESMO 2019, abstract LBA82
- Peters S et al., Nivolumab + low-dose ipilimumab versus platinum-doublet chemotherapy as first-line treatment for advanced non-small cell lung cancer: CheckMate 227 part 1 final analysis. ESMO 2019, abstract LBA4_PR