Immunotherapy: effective treatments gathering on the horizon
EGFR mutations and ALK rearrangements are well-established therapeutic targets in NSCLC, with EGFR TKIs and ALK TKIs representing the first-line standard of care for these molecular subsets of patients.
However, resistance to first-generation inhibitors invariably develops, which calls for strategies to improve upon the durability of any response. Moreover, targeted agents are available only for a limited group of patients, while chemotherapy with or without anti-angiogenic treatments still remains the cornerstone in the majority of cases. Docetaxel is the standard second-line therapy in NSCLC, but its use is hampered by important toxicity.
Two anti-angiogenic agents have recently been shown to enhance docetaxel efficacy and to improve patient survival. Ramucirumab proved beneficial in the REVEL trial in patients with all histologies [1]. In the LUME-Lung 1 study, nintedanib demonstrated activity in patients with adenocarcinoma [2]. In 2015, immunotherapeutic agents targeting the PD-L1/PD-1 pathway are about to change the management of NSCLC.
CheckMate 057: nivolumab in non-squamous NSCLC
Nivolumab, a fully human IgG4 PD-1 immune checkpoint inhibitor antibody, is active in pre-treated patients with advanced or metastatic NSCLC across different histologies. PD-1 expression on tumour-infiltrating lymphocytes is associated with decreased cytokine production and effector function. Nivolumab binds PD-1 receptors on T- cells and disrupts the negative signalling triggered by PD-L1/PD-L2, to thus restore T-cell antitumour function.
The randomised, global, phase III CheckMate 057 trial assessed the efficacy and safety of nivolumab 3 mg/kg every 2 weeks until progression (n = 292) compared to docetaxel 75 mg/m2 every 3 weeks until progression (n = 290) in patients with advanced non-squamous NSCLC (stage IIIb/IV), after failure of one platinum-based doublet chemotherapy [3]. PD-L1 expression was measured using IHC. The rationale for this trial was based on the concept that patients with advanced non-squamous NSCLC who progress after platinum-based doublet chemotherapy only have limited effective options.
Outcomes according to PD-L1 expression
The CheckMate 057 trial showed that nivolumab-treated patients benefited significantly in terms of OS, compared to the docetaxel arm (12.2 vs. 9.4 months; HR, 0.73; p = 0.0015). The 1-year OS rates were 51 % and 39 % for nivolumab and docetaxel, respectively. The OS analysis favoured nivolumab in almost all of the predefined subgroups. Nivolumab is the first PD-1 inhibitor to significantly improve OS over docetaxel in previously treated patients with advanced non-squamous NSCLC.
There was also significant advantage for the ORR in the experimental arm (19 % vs. 12 %; p = 0.0246), and the responses lasted conspicuously longer in the nivolumab-treated group (17.2 vs. 5.6 months). The PFS did not differ significantly between the two arms. Also, nivolumab showed a more favourable safety profile than its comparator.
According to the biomarker analysis, both OS and PFS were significantly prolonged in the nivolumab patients with marked PD-L1 expression. This correlation was evident already at the lowest expression levels. The median OS nearly doubled with nivolumab versus docetaxel across the PD-L1 expression continuum. On the other hand, no differences in OS were seen in patients whose tumours did not express PD-L1. The ORR almost tripled in the PD-L1 expressors.
However, when compared indirectly to pooled data of ramucirumab plus docetaxel and nintedanib plus docetaxel, these results are not robust enough to be able to state that nivolumab can outperform the combination of docetaxel and the anti-angiogenics. Only a head-to-head trial will answer this question fully.
CheckMate 017: nivolumab in patients with squamous histology
As compared to patients with non-squamous tumours, those with squamous cell histology have a worse prognosis. Indeed, the therapeutic options after front-line chemotherapy are meagre in this group. Second-line therapy with docetaxel has only modest clinical activity and is associated with significant toxicity.
The phase II, single-arm trial CheckMate 063 demonstrated clinical activity of nivolumab in refractory squamous-cell carcinoma with a median OS of 8.2 months, a 1-year OS rate of 41 %, and an ORR of 15 % [4]. In the head-to-head setting, the randomised global phase III study CheckMate 017 tested the efficacy and safety of nivolumab compared to docetaxel in patients with advanced squamous-cell carcinoma after failure of platinum-based chemotherapy [5]. Patients with stage IIIb/IV squamous NSCLC who had already received one previous platinum-doublet-based chemotherapy were randomised to either nivolumab 3 mg/kg every 2 weeks until progression (n = 135) or docetaxel 75 mg/m2 every 3 weeks until progression (n = 137). Pre-treatment tumour samples were required for the PD-L1 analysis. 83 % of patients showed quantifiable PD-L1 expression.
Superiority across all endpoints
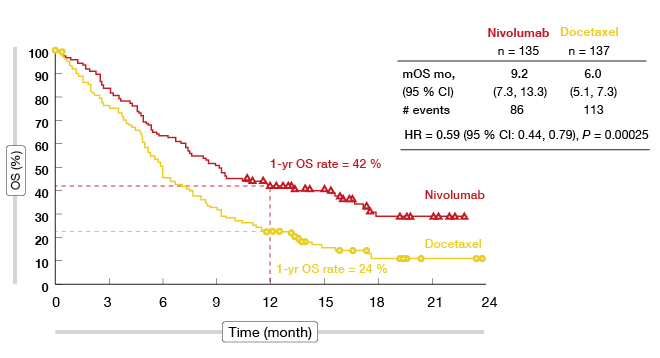
Regarding the OS, which was defined as the primary endpoint of the CheckMate 017 trial, nivolumab did significantly better than docetaxel. Survival was 9.2 months versus 6.0 months with nivolumab and docetaxel, respectively, which translated to a reduction in the risk of death of 41 % (HR, 0.59; p = 0.00025; Figure 1). At 1 year, the OS rates were 42 % and 24 %, respectively (Figure 1). Nivolumab demonstrated superiority over docetaxel across all of the secondary efficacy endpoints. The PFS was significantly longer with nivolumab (3.5 vs. 2.8 months; HR, 0.62; p = 0.0004). 21 % of patients were progression-free in the nivolumab arm at 1 year, while this proportion was much lower in the control arm (6.4 %). Objective response rates were 20 % and 9 %, respectively (p = 0.0083).
Figure 1: Overall survival in CheckMate 017
The OS and PFS were also investigated according to PD-L1 expression. This analysis showed, however, that the survival benefit obtained with nivolumab was independent of PD-L1 expression levels. This was also true for the ORR, which was also consistently higher with nivolumab than with docetaxel. Moreover, according to the Lung Cancer Symptom Scale (LCSS) Average Symptom Burden Index, the symptoms were reduced more efficiently with nivolumab treatment.
Treatment-related AEs occurred more frequently with docetaxel than with nivolumab (any grade: 86 % vs. 58 %). Correspondingly, AEs predominantly led to discontinuation in the docetaxel arm (10 % vs. 3 %). Nivolumab showed a favourable safety profile that was consistent with prior studies. Fatigue, decreased appetite, and asthenia were the most common AEs reported in the nivolumab arm. Docetaxel, on the other hand, gave rise to fatigue, neutropenia, anaemia, nausea, diarrhoea, and alopecia. Among the select AEs, however, pneumonitis occurred in 5 % of the nivolumab-treated patients (docetaxel: 0 %), and was severe in 1 %. Nivolumab is the first PD-1 inhibitor to demonstrate a survival benefit compared to standard-of-care docetaxel therapy in previously treated patients with advanced squamous NSCLC.
However, as for CheckMate 057, the follow-up period is still relatively short. An indirect comparison of the REVEL and CheckMate 017 trials shows a larger risk reduction between the two arms regarding OS with nivolumab versus docetaxel than with docetaxel plus ramucirumab versus docetaxel monotherapy, which suggests that nivolumab could outperform not only docetaxel, but also the combination of docetaxel and ramucirumab. Nevertheless, a randomised trial is called for to clarify this.
POPLAR: favourable results with atezolizumab
Atezolizumab (MPDL3280A) is a humanised anti-PD-L1 antibody that inhibits the binding of PD-L1 to PD-1 and B7.1. The inhibition of the PD-L1/PD-1 and PD-L1/B7.1 interactions can restore antitumour T-cell activity and enhance T-cell priming. Targeting PD-L1 leaves the PD-L2/PD-1 interaction intact, thereby potentially preserving peripheral immune homeostasis. PD-L2 expression on tumour cells and immune cells is potentially predictive for the activity of atezolizumab in NSCLC.
In the POPLAR randomised, all-comer, phase II study, 287 patients with metastatic or locally advanced NSCLC who showed disease progression on a prior platinum therapy were randomised to either atezolizumab 1,200 mg IV every 3 weeks until loss of clinical benefit or docetaxel 75 mg/m2 IV every 3 weeks until progression [6]. PD-L1 expression (0 vs. 1 vs. 2 vs. 3) was one of the stratification factors. The primary study objective was the estimated OS in the PD-L1-selected and the intention-to-treat (ITT) patient populations.
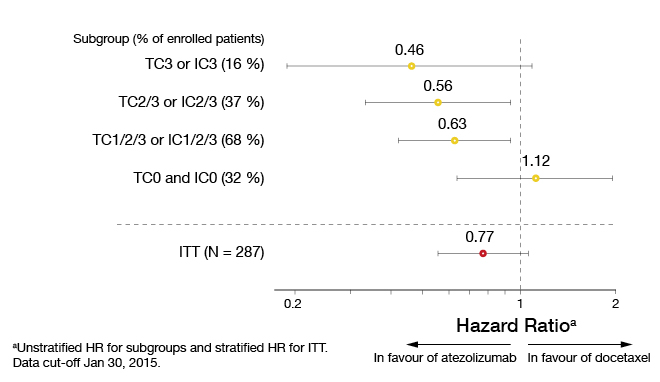
After a minimum of 10 months of follow-up, an interim analysis was performed according to which the OS favours atezolizumab in most of the PD-L1 expression subgroups (Figure 2). The pattern of improved survival correlated with increasing PD-L1 expression. Whereas the patients with the highest PD-L1 expression (i.e., tumour cell ‘TC3’ or immune cell ‘IC3’) experienced a reduction in mortality of 54 %, those in the ‘TC0’ and ‘IC0’ cohorts, with PD-L1 expression < 1 %, did not derive any benefit relative to docetaxel. This correlation between PD-L1 expression and risk reduction also applies to the PFS analysis. In the ITT population, the OS was longer in the atezolizumab-treated arm than in the docetaxel-treated arm, although not significantly so (11.4 vs. 9.5 months; HR, 0.77; p = 0.11). Also, the response rates achieved with atezolizumab were highest in the ‘TC3’ and ‘IC3’ subgroups (38 %). In the ITT population, there was no difference between the two treatment arms regarding the response rates (15 % each). The median duration of response had not been reached in the experimental arm at the time of the analysis, while it was 7.8 months for the control arm.
Figure 2: POPLAR: Increasing overall survival benefit with atezolizumab treatment relative to PD-L1 expression
Atezolizumab was well tolerated, and its safety profile was consistent with previous studies. Compared with docetaxel, the atezolizumab AE rates were lower, which also applied to grade 3/4 AEs. The median treatment duration with atezolizumab exceeded that in the docetaxel group (3.7 vs. 2.1 months), and the proportion of patients treated beyond progression was greater by far (38 % vs. 2 %). This trial also demonstrated that the highly sensitive and specific IHC SP142 assay that measures PD-L1 on both tumour cells and immune cells is a predictive diagnostic biomarker for the efficacy of atezolizumab treatment in NSCLC. A phase III randomised study of atezolizumab monotherapy in second-line and third-line NSCLC patients is ongoing at present.
Preliminary evidence on pembrolizumab: KEYNOTE-021
Robust antitumour activity and manageable toxicity in multiple tumour types has been shown for pembrolizumab, a potent, humanised monoclonal antibody against PD-1. In the KEYNOTE-001 trial, pembrolizumab was effective in treatment-naïve patients with advanced NSCLC [7]. The KEYNOTE-021 trial tested the combination of pembrolizumab and the CTLA-4 antibody ipilimumab [8]. Anti-PD-1 and anti-CTLA-4 combination therapy is feasible because of the complementary mechanisms of action: CTLA-4 functions at the activation stage of the anticancer immune response, while PD-1 shows activity at the effector stage.
Patients with advanced or metastatic NSCLC of any histology participated in KEYNOTE-021. They had to have received at least one prior therapy that included ≥ 1 platinum-doublet chemotherapy. Any PD-L1 status was allowed, as well as any EGFR and ALK status. In the dose-finding part of this study, pembrolizumab 2 mg/kg plus ipilimumab 1 mg/kg was shown to have a manageable toxicity profile. The most frequent treatment-related AEs were fatigue, decreased appetite, myalgia, pruritus and rash.
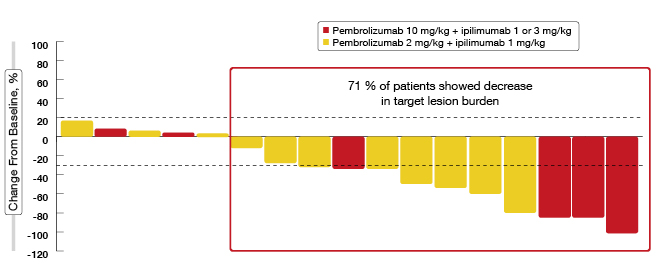
The preliminary evidence on efficacy has indicated robust, durable antitumour activity in an unselected population. In the pembrolizumab 2 mg/kg plus ipilimumab 1 mg/kg dose group, an ORR of 33 % was achieved. Partial responses were 33 %, and disease stabilisation for at least 6 weeks was achieved in 42 %. The DCR was 75 %. 71 % of patients showed decreases in target-lesion burden (Figure 3). All of the responses were ongoing at the time of data cut-off. The combination will be further characterised in a currently enrolling dose-expansion cohort of 32 patients.
Figure 3: Reductions in target lesions with pembrolizumab plus ipilimumab in KEYNOTE-021
REFERENCES
- Garon EB et al., Ramucirumab plus docetaxel versus placebo plus docetaxel for second-line treatment of stage IV non-small-cell lung cancer after disease progression on platinum-based therapy (REVEL): a multicentre, double-blind, randomised phase 3 trial. Lancet Oncol 2014; 23; 384(9944): 665-73
- Reck M et al., Docetaxel plus nintedanib versus docetaxel plus placebo in patients with previously treated non-small-cell lung cancer (LUME-Lung 1): a phase 3, double-blind, randomised controlled trial. Lancet Oncol 2014; 15(2): 143-55
- Paz-Ares L et al., Phase III, randomized trial (CheckMate 057) of nivolumab (NIVO) versus docetaxel (DOC) in advanced non-squamous cell (non-SQ) non-small cell lung cancer (NSCLC). J Clin Oncol 33, 2015 (suppl; abstr LBA109)
- Rizvi NA et al., Activity and safety of nivolumab, an anti-PD-1 immune checkpoint inhibitor, for patients with advanced, refractory squamous non-small-cell lung cancer (CheckMate 063): a phase 2, single-arm trial. Lancet Oncol 2015 16(3): 257-65
- Spigel DR et al., A phase III study (CheckMate 017) of nivolumab (NIVO; anti-programmed death-1 [PD-1]) vs docetaxel (DOC) in previously treated advanced or metastatic squamous (SQ) cell non-small cell lung cancer (NSCLC). J Clin Oncol 33, 2015 (suppl; abstr 8009)
- Spira AI et al., Efficacy, safety and predictive biomarker results from a randomized phase II study comparing MPDL3280A vs docetaxel in 2L/3L NSCLC (POPLAR). J Clin Oncol 33, 2015 (suppl; abstr 8010)
- Garon EB et al., Pembrolizumab for the treatment of non-small-cell lung cancer. N Engl J Med 2015; 372: 2018-20
- Patnaik A et al., Phase 1 study of pembrolizumab (pembro; MK-3475) plus ipilimumab (IPI) as second-line therapy for advanced non-small cell lung cancer (NSCLC): KEYNOTE-021 cohort D. J Clin Oncol 33, 2015 (suppl; abstr 8011)
More posts
Interview: “These findings will change the standard-of-care”
Will findings presented here at ASCO change the future of NSCLC therapy?
Further analyses of biomarkers
Immune checkpoint inhibitors have become established as standard therapy for certain subsets of NSCLC patients. Efforts are ongoing to optimise the benefits gained through these drugs, by identification of reliable prognostic and predictive biomarkers, such as PD-L1 expression in tumour cells or infiltrating immune cells, CD8-positive tumour-infiltrating lymphocytes, smoking status, and mutation burden.
Novel approaches in small-cell lung cancer
Small-cell lung cancer (SCLC) accounts for approximately 15 % of all lung cancers, and it is associated with poor outcomes. 70 % of these patients present with extensive disease. Their treatment remains a significant challenge for oncologists. The median survival in the extensive disease stage is 7 months to 9 months, and only 2 % of patients survive for 5 years
Statement: “Select the patients who will be able to benefit”
At this ASCO Congress, the biggest news in the field of NSCLC was the effects of the checkpoint inhibitors in the treatment of this disease. This applies especially to the results of the CheckMate 057 trial. The PD-1 inhibitor nivolumab is one of the first checkpoint inhibitors to be approved in lung cancer; it was approved in the United States for the treatment of squamous NSCLC early in 2015.
Immunotherapy: effective treatments gathering on the horizon
EGFR mutations and ALK rearrangements are well-established therapeutic targets in NSCLC, with EGFR TKIs and ALK TKIs representing the first-line standard of care for these molecular subsets of patients. However, resistance to first-generation inhibitors invariably develops, which calls for strategies to improve upon the durability of any response.
Interview: “EGFR testing is a reality in European countries”
Which of the recent advances in the field of NSCLC would you deem most important from the clinical point of view?