New applications of PARP inhibitors
Metastatic castration-resistant prostate cancer
Metastatic castration-resistant prostate cancer (mCRPC) that progresses after androgen-receptor(AR)–targeted therapy (i.e., enzalutamide or abiraterone) and taxane-based chemotherapy is associated with a poor prognosis [1]. Only few treatment options are available for these patients. Up to 25 % of men with mCRPC harbor deleterious germline and/or somatic alterations in BRCA1, BRCA2, ATM or other DNA damage repair (DDR) genes, including those with direct or indirect roles in homologous recombination repair (HRR) [2-4]. These alterations are associated with sensitivity to PARP inhibition [5]. Emerging data suggest clinical activity of PARP inhibitors in patients with mCRPC and DDR gene anomalies [6-8]. The PROfound, GALAHAD and TRITON2 studies investigated the clinical benefit of PARP inhibition with olaparib, niraparib and rucaparib, respectively, in advanced mCRPC.
Olaparib: primary analysis of PROfound
Patients whose disease had progressed on enzalutamide or abiraterone and who had alterations in any of 15 predefined genes that play a direct or indirect role in HRR were enrolled into randomized, open-label, phase III PROfound trial that evaluated olaparib compared to AR-targeted therapy. Cohort A included 245 patients with alterations in BRCA1, BRCA2 or ATM, while Cohort B included 142 patients with one of 12 other HRR alterations ranging from BRIP1 to RAD54L. Both cohorts were randomized in a 2:1 ratio to either olaparib or AR-targeted therapy according to physician’s choice. Patients progressing on physician’s choice treatment were allowed to cross over to olaparib. The primary endpoint was radiographic progression-free survival (rPFS) in Cohort A as assessed by blinded independent central review.
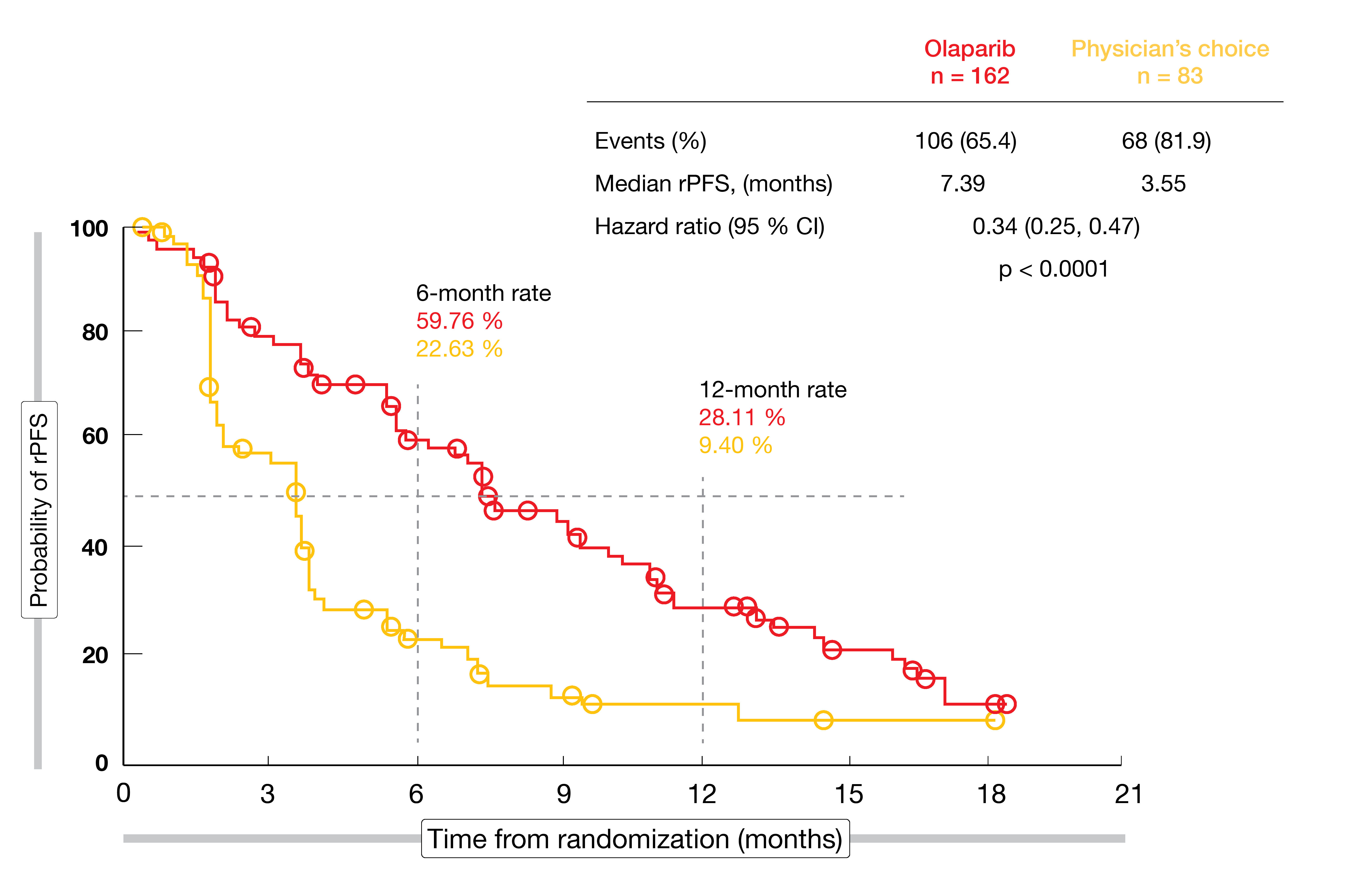
Hussain et al. reported that in Cohort A, olaparib indeed provided a statistically significant rPFS improvement compared to enzalutamide or abiraterone (7.39 vs. 3.55 months; HR, 0.34; p < 0.0001; Figure 1) [9]. This was also observed in the overall population including both cohorts with alterations in any qualifying gene (5.82 vs. 3.52 months; HR, 0.49; p < 0.0001). In cohort A, the objective response rate (ORR) in patients who had measurable disease was 33.3 % with olaparib compared to 2.3 % with enzalutamide or abiraterone (odds ratio, 20.86; p < 0.0001). Median time to pain progression had not been reached with olaparib versus 9.92 months for the hormonal agents, representing a 56 % reduction (HR, 0.44; p = 0.0192). Olaparib showed a favorable trend with respect to overall survival (OS) for patients with alterations in BRCA1, BRCA2 or ATM (18.5 vs. 15.11 months; HR, 0.64; p = 0.0173) and in the overall population (17.51 vs. 14.26 months; HR, 0.67; p = 0.0063). This was achieved despite a cross-over of more than 80 %. However, OS data are still immature.
Olaparib was well tolerated. The most common adverse events (AEs) included anemia (46.1 % vs. 15.4 %), nausea (41.4 % vs. 19.2 %), fatigue (41.0 % vs. 32.3 %), and decreased appetite (30.1 % vs. 17.7 %). The authors concluded that PROfound is the first positive biomarker-selected phase III study evaluating a molecularly targeted therapy in men with mCRPC. Also, these findings highlight the importance of genomic testing in this population.
Figure 1: Radiographic progression-free survival with olaparib vs. enzalutamide or abiraterone in mCRPC patients with BRCA1, BRCA2, or ATM alterations
GALAHAD: interim data on niraparib
The ongoing, open-label, single-arm, phase II GALAHAD study is assessing the safety and efficacy of niraparib in patients with mCRPC and DNA repair defects (DRD) who have progressed on ≥ 1 line of AR-targeted therapy and ≥ 1 line of taxane-based chemotherapy. At ESMO 2019, Smith et al. presented results from a pre-specified interim analysis [10]. Overall, 223 patients were screened for eligibility, and 165 patients with DRD (defined as pathogenic mutation in BRCA1/2 [BRCA], ATM, FANCA, PALB2, CHEK2, BRIP1 or HDAC2 [non-BRCA]; monoallelic or biallelic) were enrolled.
The findings indicate that niraparib has high clinical activity in patients with mCRPC, particularly in those with biallelic BRCA DRD (n = 46). This group showed an ORR of 41 %, and objective responses lasted for 5.6 months. The composite response rate, which was defined as ORR by RECIST 1.1, or conversion of circulating tumor cells from ≥ 5/7.5 mL to < 5/7.5 mL of blood, or ≥ 50 % decline in PSA, amounted to 63 %. Patients with measurable disease showed a composite response rate of 66 %; for those with non-measurable disease, this was 59 %. Median rPFS and OS were 8.2 and 12.6 months, respectively. On the other hand, patients with non-BRCA biallelic DRD (n = 35) responded only in 9 %, and the composite response rate was 17 %. Median rPFS and OS for this group were 5.3 and 14.0 months, respectively. Declines in PSA levels of ≥ 50 % occurred in 50 % and 3 % of patients with BRCA and non-BRCA biallelic DRD, respectively.
Overall, niraparib treatment has shown a manageable safety profile with no new safety signals identified. The most common grade 3/4 AEs comprised anemia (29 %), thrombocytopenia (15 %), and neutropenia (7 %). Niraparib in patients with mCRPC and DRD will continue to be evaluated in ongoing trials including GALAHAD, MAGNITUDE and QUEST.
Promising activity of rucaparib in TRITON2
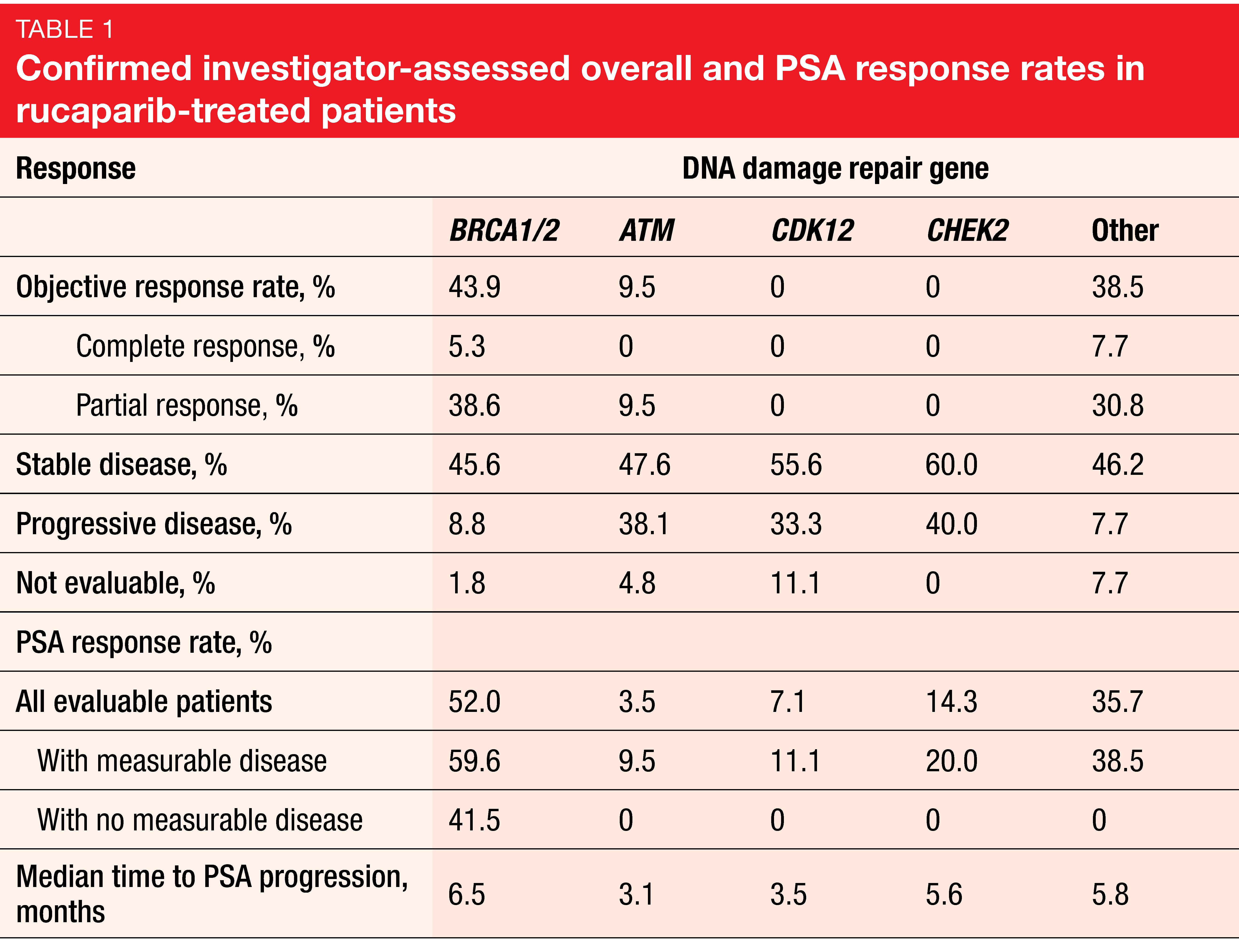
Another ongoing phase II trial testing a PARP inhibitor in the mCRPC setting is TRITON2. Here, patients with mCRPC and DDR alterations including BRCA1/2, ATM, CDK12 and CHEK2, who have progressed on AR-targeted therapy and chemotherapy, are being treated with rucaparib. According to the analysis presented at ESMO 2018, patients with deleterious BRCA1/2 alterations showed confirmed ORR and PSA responses in 44.0 % and 51.1 %, respectively [11]. At ESMO 2019, Abida et al. presented an update from TRITON2 after a median follow-up of 13.1 months for a total of 190 patients [12].
In keeping with prior reports, rucaparib demonstrated promising efficacy. Among evaluable patients with BRCA1/2 alterations, 43.9 % experienced confirmed investigator-assessed radiographic responses. Fifty-two percent of all men with BRCA1/2 mutations had confirmed PSA responses (i. e., ≥ 50 % decreases). Patients with germline and somatic BRCA1/2 alterations responded to a similar extent. Also, confirmed radiographic and PSA responses occurred in the group with alterations in other DDR genes, including ATM, CDK12, and CHEK2 (Table 1). Among patients with BRCA1/2 alterations who demonstrated confirmed radiographic responses, the majority (60.0 %) responded for more than 24 weeks.
The safety profile of rucaparib was consistent with prior reports from TRITON2 and the experience obtained in patients with ovarian cancer and other solid tumors [7, 13-15]. At 17.9 %, the most common grade ≥ 3 treatment-emergent AE was anemia. In addition to ongoing enrolment into TRITON2, the randomized, phase III TRITON3 study is evaluating rucaparib versus second-line AR-directed therapy or docetaxel in chemotherapy-naïve mCRPC patients with deleterious alterations in BRCA1, BRCA2, or ATM, who progressed on one prior AR-targeted therapy. Here, rPFS has been defined as the primary objective.
Pancreatic cancer: analyses of the POLO trial
POLO is the first phase III trial to evaluate maintenance therapy with a PARP inhibitor in metastatic pancreatic cancer. In this randomized, international study, pancreatic cancer patients with germline BRCA1 and/or BRCA2 mutations whose disease had not progressed during ≥ 16 weeks of first-line platinum-based chemotherapy received either olaparib or placebo. Indeed, olaparib treatment resulted in a statistically significant and clinically meaningful PFS benefit compared with placebo (7.4 vs. 3.8 months; HR, 0.53; p = 0.004) [16]. At ESMO 2019, results for prespecified secondary efficacy analyses of the study and data on health-related quality of life were presented.
Time to treatment discontinuation and subsequent therapy
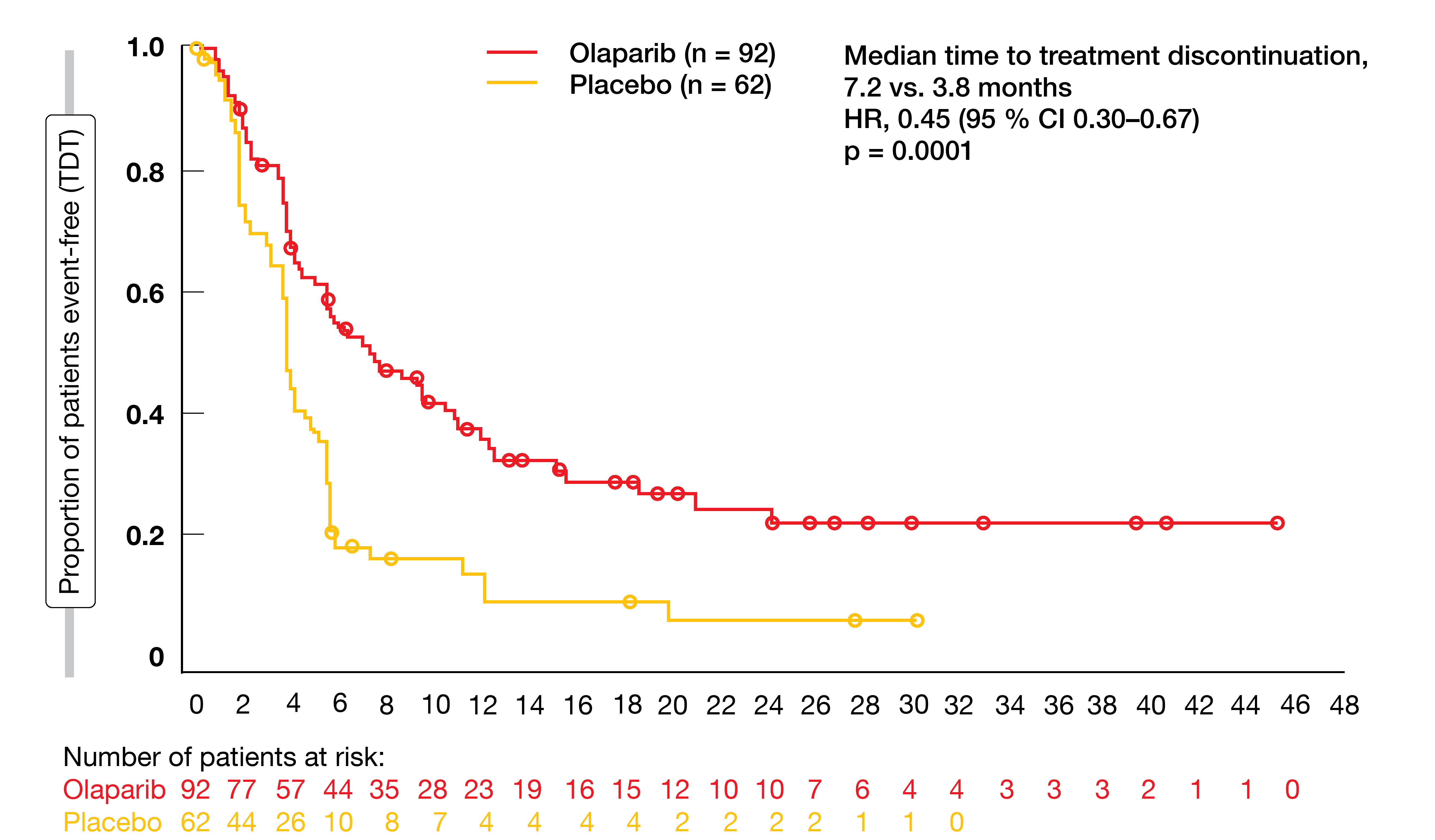
Secondary endpoints of the POLO trial included time to treatment discontinuation as well as time to first and second subsequent therapies. Since maintenance treatment with olaparib provided a PFS benefit, patients in the experimental arm were less likely to require a subsequent therapy than those in the placebo arm. Van Cutsem et al. demonstrated that olaparib maintenance led to a meaningful prolongation of time to treatment discontinuation compared to placebo, translating to a 55 % risk reduction (7.2 vs. 3.8 months; HR, 0.45; p = 0.0001) (Figure 2) [17]. Furthermore, there were meaningful increases in time to initiation of both first (8.6 vs. 5.7 months; HR, 0.50; p = 0.0013) and second subsequent treatments (13.2 vs. 9.2 months; HR, 0.68; p = 0.083).
Among the patients who did receive subsequent treatment, the majority in both arms were treated with cytotoxic chemotherapy. Hence, maintenance olaparib can meaningfully delay the need of second-line treatments for metastatic pancreatic cancer, and data suggest that this effect may be maintained up to the third line. The authors concluded that maintenance olaparib following first-line platinum-based treatment may provide the opportunity to delay the subsequent use of standard cytotoxic chemotherapies with their associated toxicities.
Figure 2: Superior time to treatment discontinuation with olaparib maintenance compared to placebo
Health-related quality of life
In addition to efficacy benefits, preservation of patient health-related quality of life (HRQoL) is a major therapeutic goal in the maintenance setting. A prespecified secondary objective of the POLO study was the effect of maintenance olaparib on HRQoL, specifically evaluating the adjusted mean change from baseline in global health status using the EORTC QLQ-C30 questionnaire [18]. Analyses were conducted in 89 of 92 patients in the olaparib arm and 58 of 62 patients in the placebo arm with evaluable baseline forms (overall compliance was 96.6 % and 94.8 %, respectively).
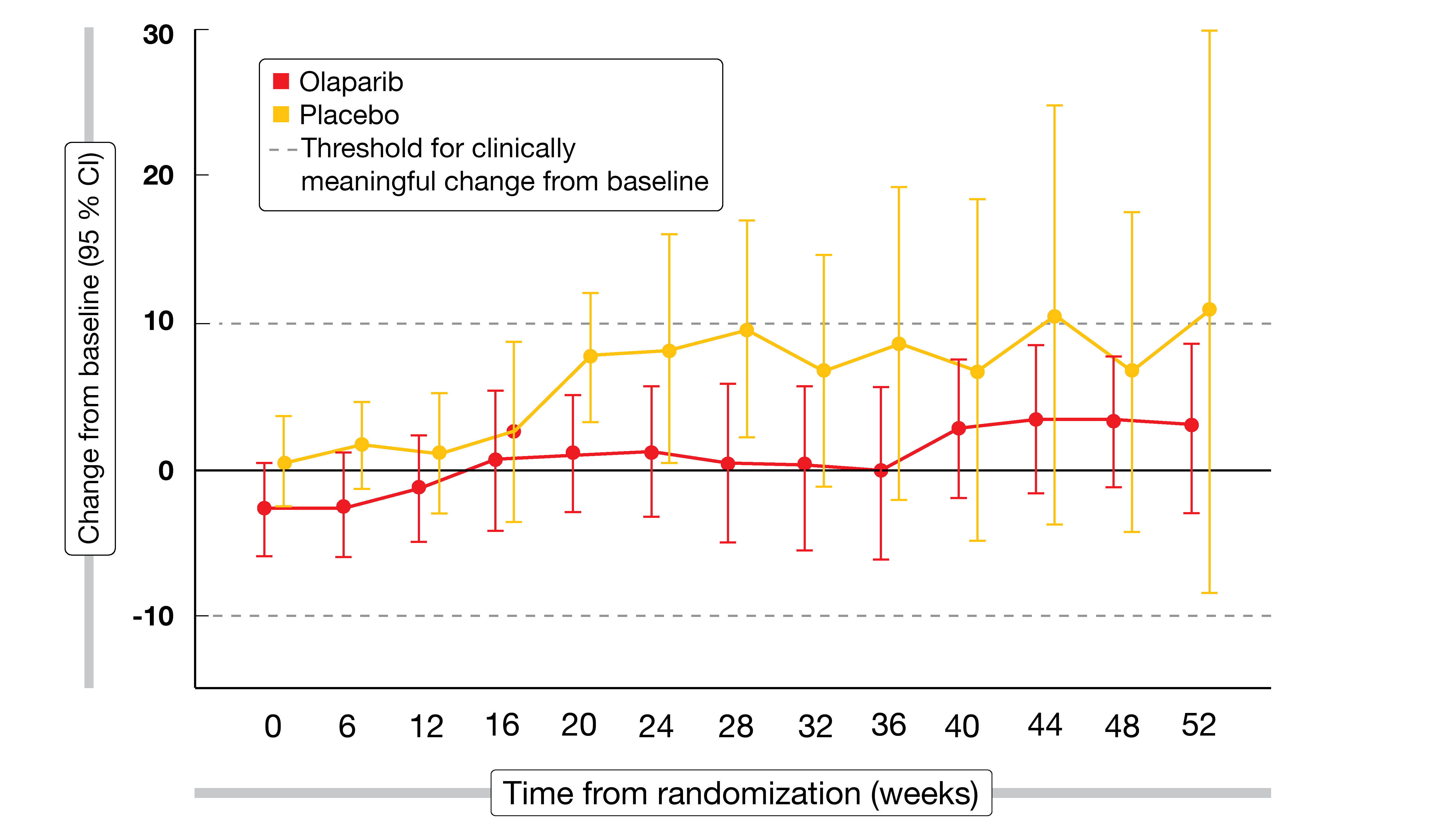
Patients treated in both arms of the POLO study had high baseline global health status scores (70.4 vs. 74.3) and physical functioning scores (83.3 vs. 84.9) following successful first-line chemotherapy. Global health status remained relatively stable over time for both treatment arms without a statistically significant or clinically meaningful difference in the overall between-group adjusted mean change from baseline. For physical functioning, the scores improved over time in both arms (Figure 3), although the adjusted mean change from baseline between groups did not reach the threshold considered to be clinically meaningful. There was no difference in time to sustained clinically meaningful deterioration for olaparib versus placebo for global HRQoL (21.2 vs. 6.0 months; HR, 0.72; p = 0.25). Overall, olaparib maintenance treatment was shown to preserve the HRQoL benefits achieved with first-line chemotherapy.
Figure 3: Improvement of physical functioning over time in both arms of the POLO trial
Novel PARP inhibition
Pamiparib monotherapy in advanced solid tumors
Pamiparib is a potent and selective oral PARP1/2 inhibitor that has demonstrated promising antitumor activity in ovarian cancer in a phase Ia study [19]. Single-agent recommended phase II dose was defined as 60 mg twice daily in the dose-escalation part. The dose-expansion component of the study was conducted in patients with ovarian, breast, prostate, gastric, and small-cell lung cancer. Voskoboynik et al. reported updated safety data from the study and updated efficacy data from the cohorts with ovarian cancer and associated tumors [20].
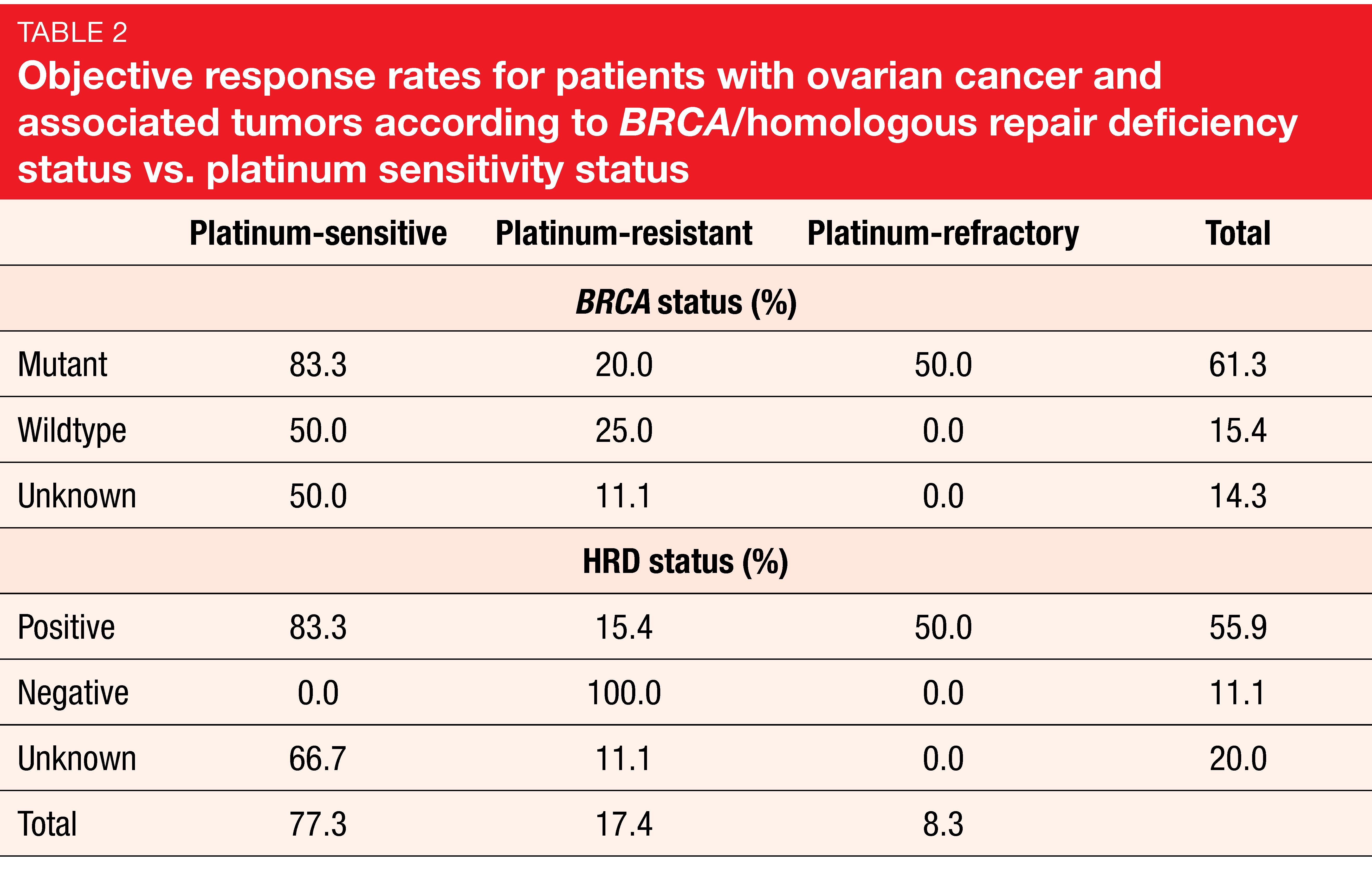
As of June 2019, 101 patients were enrolled in the dose-escalation (n = 64) and dose-expansion (n = 37) cohorts. Out of these, 63 patients had ovarian, fallopian, or peritoneal cancer, and 28 received pamiparib at the recommended phase II dose. Confirmed complete or partial responses were observed in 23 of 58 evaluable ovarian and associated cancer patients (39.7 %). The median duration of response was 14.9 months. Among the 58 evaluable patients, 31 had germline or somatic BRCA mutations, while 27 had either germline or somatic BRCA wildtype or unknown BRCA status. Pamiparib treatment elicited higher ORR in the mutated group compared to the other cohorts (61.3 % vs. 14.8 %). ORRs by platinum-sensitivity status were 77.3 %, 17.4 % and 8.3 % for the platinum-sensitive, platinum-resistant and platinum-refractory populations, respectively. In the platinum-sensitive group, higher ORR was achieved in BRCA-mutant patients than in the groups with BRCA wildtype or unknown status (83.3 % vs. 50.0 %; Table 2).
In the safety population (n = 101), treatment-emergent AEs observed in 10 % or more of patients included nausea, fatigue, anemia, diarrhea, vomiting, and decreased appetite. The most common treatment-emergent ≥ grade 3 AE was anemia. Pamiparib plasma exposure increased linearly with increasing dose, with a median terminal half-life of approximately 13 hours. Hence, pamiparib can be administered independent of food intake. The authors concluded that pamiparib continued to be generally well-tolerated and demonstrated promising antitumor activity in patients with ovarian and associated cancer.
Combination with temozolomide
A dose-escalation/expansion study is evaluating pamiparib together with oral low-dose temozolomide in patients with locally advanced and metastatic tumors. The rationale for this combination is based on the hypothesis that DNA damage caused by low-dose temozolomide synergizes with PARP inhibition. This synergy might lead to increased antitumor activity via enhanced PARP-dependent tumor cell killing. PARP inhibition results in the accumulation of highly cytotoxic adducts, leading to cell death.
During dose escalation, patients received pamiparib 60 mg twice daily plus escalating doses of temozolomide daily on days 1-7 (Arm A; pulsed) or continuously (Arm B; continuous flat) for each 28-day cycle. According to a preliminary analysis, pamiparib 60 mg twice daily combined with pulsed or continuous-flat–dosed temozolomide showed antitumor activity and was generally well tolerated, with the expected toxicity of bone marrow suppression [21].
Stradella et al. presented updated results of this phase Ib study including data on the recommended phase II dose and schedule of the combination, which was determined to be pamiparib 60 mg twice daily on days 1-28 and pulsed temozolomide 60 mg daily on days 1-7 [22]. A total of 113 patients with solid tumors had been enrolled, with 66 and 47 patients included in the dose-escalation and dose-expansion cohorts, respectively.
Disease control in up to 80 % of SCLC patients
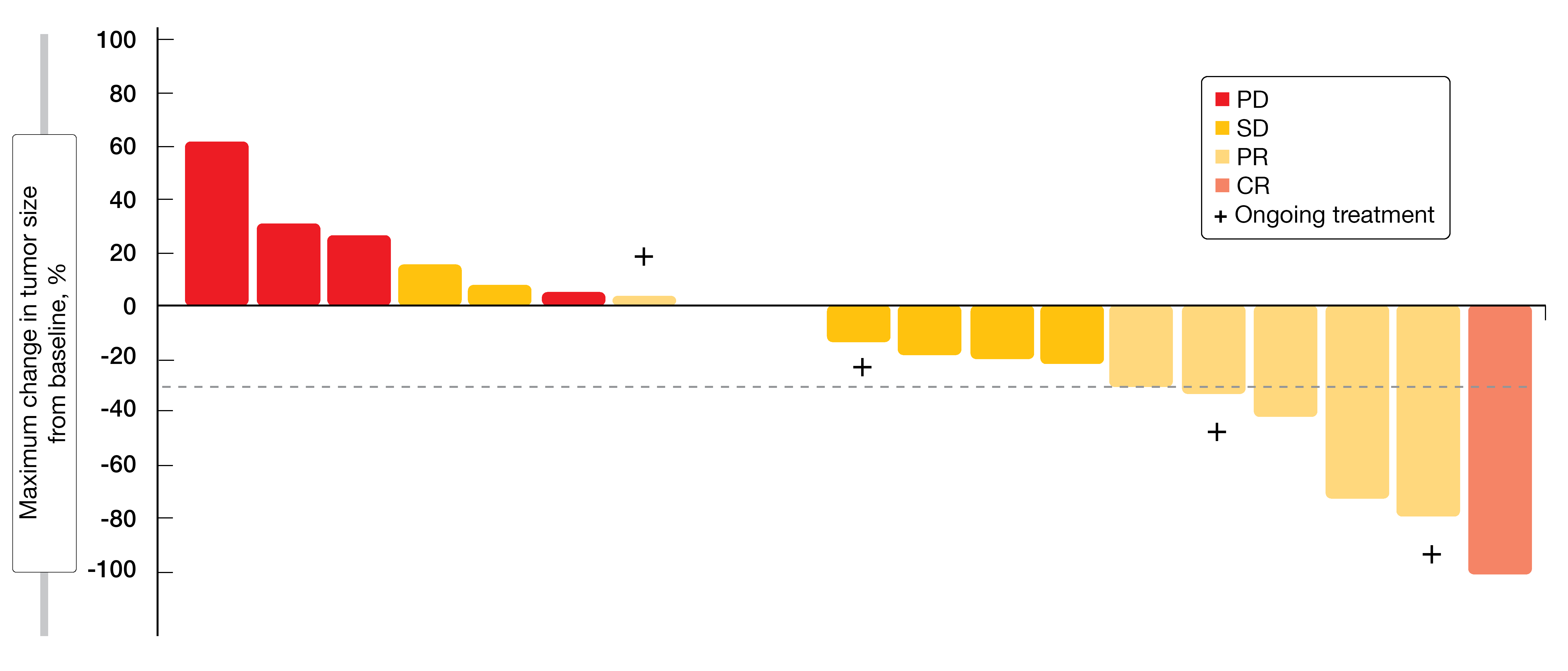
Promising preliminary efficacy was found in patients with extensive-stage small-cell lung cancer enrolled in the expansion phase (n = 22). In this group, out of 19 patients evaluable for response, 31.6 % showed responses to treatment, and the disease control rate was 78.9 % (Figure 4). Twenty patients with gastric/gastroesophageal junction cancer were also enrolled in the expansion phase, with 15 being evaluable for response. Disease control was achieved in 33.3 % of patients in this cohort.
The dose-escalation phase enrolled all comers, with 57 of 66 patients being evaluable for response (52 with measurable disease). For this group, ORR and disease control rate were 19.3 % and 64.9 %, respectively. Responses lasted for a median of 6.4 months.
Eight patients with homologous recombination deficient (HRD) tumors were included in the dose-escalation phase. Irrespective of BRCA mutational status, these showed a response rate of 62.5 %. According to the conclusion of the authors, HRD status may be a promising biomarker for sensitivity to treatment with pamiparib plus low-dose temozolomide, regardless of tumor type. The combination showed a manageable safety profile, with cytopenias representing the most frequent grade ≥ 3 events.
Figure 4: Maximum tumor reduction obtained with pamiparib plus temozolomide in evaluable patients with extensive-stage small-cell lung cancer
Fluzoparib in patients with advanced solid tumors
Fluzoparib (SHR3162) is a selective oral PARP1 inhibitor that has shown anti-tumor activity in both cell lines and xenograft models [23]. Li et al. presented results from the first-in-human, phase I trial investigating fluzoparib in patients with advanced solid tumors [24]. At five centers in China, 48 and 31 patients were enrolled into the dose-escalation and dose-expansion arms, respectively. Ovarian cancer represented the largest proportion of tumor types (59.5 %), followed by breast (20.3 %), colorectal (10.1 %), and other types of cancer (10.1 %). In the dose-escalation phase, fluzoparib was administered once or twice daily at 11 dose levels from 10 mg/day to 400 mg/day. The dose-expansion phase evaluated fluzoparib at 80 mg, 100 mg or 150 mg twice daily in patients with ovarian cancer. Dose-limiting toxicity was reported in two patients who received 400 mg/day. The maximum tolerated dose was 150 mg twice daily, which was determined to be the recommended phase II dose.
All patients experienced AEs during this study. Hematologic AEs of all grades included anemia (53.2 %), thrombocytopenia (17.7 %) and decreased neutrophil counts (24.1 %). The main non-hematologic AEs comprised fatigue (48.1 %), vomiting (17.7 %), nausea (34.2 %), and decreased appetite (29.1 %). Approximately 42 % of patients experienced grade 3/4 AEs, with the most common being anemia (8.9 %) and decreased neutrophil counts (5.1 %). The treatment was discontinued in three patients due to AEs. No treatment-related deaths occurred.
The ORR observed with fluzoparib was 8.1 % for ovarian cancer and 7.7 % for breast cancer. No responses occurred in colorectal and gastric cancer or other tumor types. For the 11 ovarian cancer patients with BRCA mutation, median PFS was 8.5 months. Among patients treated with fluzoparib ≥ 120 mg/day, median PFS was 10.2 months in the platinum-sensitive cohort. In their conclusion, the authors noted that fluzoparib was well tolerated at a dose of 150 mg twice daily in advanced solid malignancies. This selective PARP1 inhibitor demonstrated single-agent antitumor activity in breast and ovarian cancer, particularly in BRCA-mutated and platinum-sensitive ovarian cancer.
REFERENCES
- Rathkopf DE et al., Safety and antitumor activity of apalutamide (ARN-509) in metastatic castration-resistant prostate cancer with and without prior abiraterone acetate and prednisone. Clin Cancer Res 2017; 23(14): 3544-3551
- Robinson D et al., Integrative clinical genomics of advanced prostate cancer. Cell 2015; 161(5): 1215-1228
- Pritchard CC et al., Inherited DNA-repair gene mutations in men with metastatic prostate cancer. N Engl J Med 2016; 375: 443-453
- Abida W et al. Prospective genomic profiling of prostate cancer across disease states reveals germline and somatic alterations that may affect clinical decision making. JCO Precis Oncol 2017. doi: 10.1200/PO.17.00029
- Mateo J et al., DNA-repair defects and olaparib in metastatic prostate cancer. N Engl J Med 2015; 373(18): 1697-1708
- Smith MR et al., Phase II study of niraparib in patients with metastatic castration-resistant prostate cancer (mCRPC) and biallelic DNA-repair gene defects (DRD): Preliminary results of GALAHAD. J Clin Oncol 37, 2019 (suppl 7S; abstr 202)
- Abida W et al., Preliminary results from TRITON2: a phase 2 study of rucaparib in patients (pts) with metastatic castration-resistant prostate cancer (mCRPC) associated with homologous recombination repair (HRR) gene alterations. ESMO 2018, abstract 793PD
- Mateo J et al., TOPARP-B: A phase II randomized trial of the poly(ADP)-ribose polymerase (PARP) inhibitor olaparib for metastatic castration resistant prostate cancers (mCRPC) with DNA damage repair (DDR) alterations. J Clin Oncol 37, 2019 (suppl; abstr 5005)
- Hussain M et al., PROfound: Phase III study of olaparib versus enzalutamide or abiraterone for metastatic castration-resistant prostate cancer (mCRPC) with homologous recombination repair (HRR) gene alterations. ESMO 2019, abstract LBA12_PR
- Smith MR et al., Pre-specified interim analysis of GALAHAD: A phase 2 study of niraparib in patients (pts) with metastatic castration-resistant prostate cancer (mCRPC) and biallelic DNA-repair gene defects (DRD). ESMO 2019, abstract LBA50
- Abida et al., Preliminary results from TRITON2: a phase 2 study of rucaparib in patients (pts) with metastatic castration-resistant prostate cancer (mCRPC) associated with homologous recombination repair (HRR) gene alterations. ESMO 2018, abstract 793PD
- Abida W et al., Preliminary results of the TRITON2 study of rucaparib in patients with DNA damage repair (DDR)-deficient metastatic castration resistant prostate cancer (mCRPC): updates analyses. ESMO 2019, abstract 846PD
- Kristeleit R et al., A Phase I-II study of the oral PARP inhibitor rucaparib in patients with germline BRCA1/2-mutated ovarian carcinoma or other solid tumors. Clin Cancer Res 2017; 23(15): 4095-4106
- Swisher EM et al., Rucaparib in relapsed, platinum-sensitive high-grade ovarian carcinoma (ARIEL2 Part 1): an international, multicentre, open-label, phase 2 trial. Lancet Oncol 2017; 18(1): 75-87
- Coleman RL et al., Rucaparib maintenance treatment for recurrent ovarian carcinoma after response to platinum therapy (ARIEL3): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet 2017; 390(10106): 1949-1961
- Golan T et al., Maintenance olaparib for germline BRCA-mutated metastatic pancreatic cancer. N Engl J Med 2019; 381: 317-327
- Van Cutsem et al., POLO: Time to treatment discontinuation and subsequent therapies following maintenance olaparib for patients with a germline BRCA mutation and metastatic pancreatic cancer. ESMO 2019, abstract 693P
- Hammel P et al., Health-related quality of life in patients with a germline BRCA mutation and metastatic pancreatic cancer receiving maintenance olaparib. Ann Oncol 2019 Sep 28. pii: mdz406. doi: 10.1093/annonc/mdz406. [Epub ahead of print]
- Lickliter J et al., Dose escalation/expansion study to investigate the safety, pharmacokinetics, food effect, and antitumor activity of BGB-290 in patients with advanced solid tumors. ESMO 2017, abstract 368PD
- Voskoboynik M et al., Safety, antitumor activity, and pharmacokinetics of pamiparib (BGB-290), a PARP1/2 inhibitor, in patients with advanced solid tumors: Updated phase 1 dose-escalation/expansion results. ESMO 2019, abstract 452PD
- Johnson M et al., Preliminary results of pamiparib (BGB-290), a PARP1/2 inhibitor, in combination with temozolomide in patients with locally advanced or metastatic solid tumors. ESMO 2018, abstract 421P
- Stradella A et al., Updated results of the PARP1/2 inhibitor pamiparib in combination with low-dose temozolomide in patients with locally advanced or metastatic solid tumors. ESMO 2019, abstract 451PD
- Wang L et al., Pharmacologic characterization of fluzoparib, a novel poly(ADP-ribose) polymerase inhibitor undergoing clinical trials. Cancer Sci 2019; 110(3): 1064-1075
- Li H et al., Phase I dose-escalation and expansion study of the PARP inhibitor, fluzoparib (SHR3162), in patients with advanced solid tumors. ESMO 2019, abstract 345P