PARP inhibition in gynecological cancers: recent insights
Poly(ADP-ribose) polymerase (PARP) inhibitors have been established as an important drug class for the treatment of advanced ovarian cancer (OC), which is a leading cause of cancer deaths in women. Olaparib and niraparib have been widely approved for maintenance treatment of OC patients who responded to platinum-based chemotherapy. Data presented at ESMO 2019 provide information on the use of PARP inhibition in earlier lines as well as in combination with other drug classes. Moreover, studies conducted with veliparib indicate combinability of this PARP inhibitor with chemotherapy in both ovarian and breast cancer.
BAROCCO: olaparib plus cediranib
Novel strategies are called for in the treatment of platinum-resistant OC, which represents a high unmet medical need. Weekly paclitaxel is regarded as the most effective chemotherapy regimen, although it has limited clinical activity. A new potential approach is the combined administration of the PARP inhibitor olaparib and the VEGFR tyrosine kinase inhibitor cediranib, which may have a synergistic effect. This combination was investigated by the randomized, three-arm BAROCCO trial that included patients with platinum-resistant OC who had any germline BRCA mutation status [1]. Another objective of BAROCCO was the question of whether an intermittent schedule of this combination would improve the gastrointestinal tolerability in terms of diarrhea severity. The experimental group consisted of two arms that either received the continuous schedule (cediranib 20 mg/day 7 days/week plus olaparib 300 mg twice daily 7 days/week) or the intermittent schedule (cediranib 20 mg/day 5 days/week plus olaparib 300 mg twice daily 7 days/week) in any line of treatment and any last line. Patients in the control arm were treated with paclitaxel 80 mg/m2 weekly. Each of the three arms included 41 women. Two independent primary comparisons were conducted in terms of PFS, which was defined as the primary endpoint, between each of the schedules and the comparator regimen.
Overall, this was a difficult-to-treat population 59 % of whom had already received ≥ 3 treatment lines, with a median platinum-free interval of < 3 months in all arms. The majority (89 %) had BRCA wildtype or unknown BRCA status. BAROCCO was the first trial to evaluate olaparib plus cediranib in platinum-resistant OC that contained a control arm.
No beneficial effect of intermittent administration
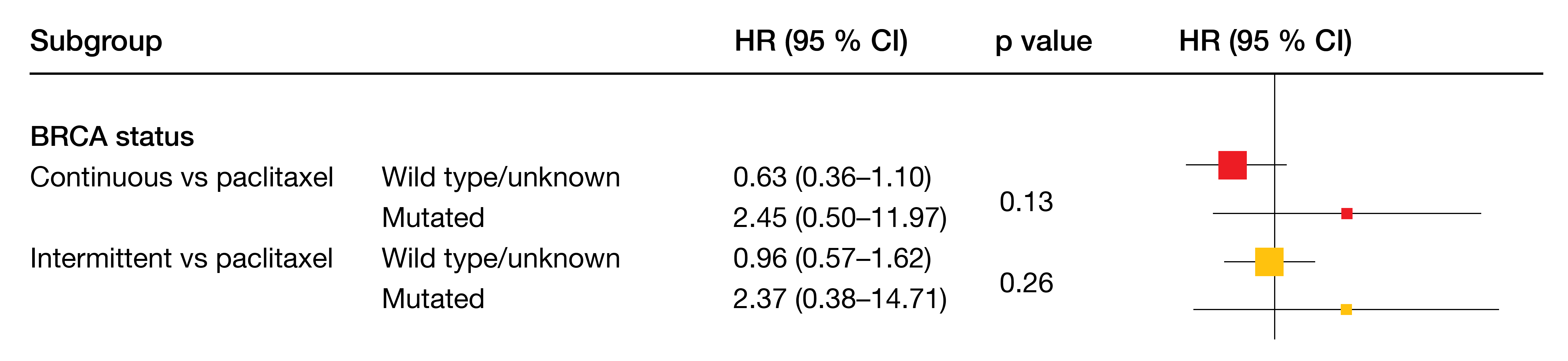
Only the continuous combination schedule was shown to be superior to chemotherapy, although not significantly so (median PFS, 5.7 vs. 3.1 months; HR, 0.76). For the intermittent schedule, PFS was 3.8 months (HR for the comparison with chemotherapy, 1.08). According to the subgroup analysis, the PFS benefit achieved with the continued administration was greatest in patients with BRCA wildtype or unknown BRCA status (5.8 vs. 2.1 months; HR, 0.63; Figure 1). With respect to response outcomes, clinical benefit was obtained with continuous treatment in 84.6 %, while this was 62.8 % and 54.1 % for the intermittent and chemotherapy groups, respectively. Correspondingly, duration of response was longest in the continuously treated arm (6.2 months compared to 2.7 and 4.4 months).
At the same time, the continuous regimen was well tolerated, with few severe side effects. Grade ≥ 3 diarrhea occurred only in 5 % of patients (intermittent schedule, 3 %). Moreover, the rates for any-grade and severe adverse events (AEs) in terms of anemia, fatigue and hypertension did not differ across the two schedules. The authors concluded that the continuous schedule showed a promising trend for improved PFS, particularly in patients with germline BRCA wildtype. While the interruption of cediranib administration for two days might have a detrimental effect on PFS with no toxicity benefit, the regimen of cediranib 20 mg daily and olaparib 300 mg twice daily represents an active, feasible, oral regimen that deserves further investigation. These results support ongoing trials investigating the same combination in the setting of platinum-resistant OC.
Figure 1: BAROCCO: greatest reduction in the risk of progression or death with continuous cediranib plus olaparib vs. weekly paclitaxel in patients with BRCA wildtype or unknown status
Addition of olaparib to bevacizumab maintenance: PAOLA-1/ENGOT-ov25
PAOLA-1/ENGOT-ov25 is the first phase III study to evaluate maintenance therapy with a PARP inhibitor in patients with advanced OC regardless of BRCA mutation status who receive first-line standard-of-care treatment including the anti-VEGF antibody bevacizumab [2]. Newly diagnosed patients who had obtained complete response (CR), partial response (PR) or no evidence of disease with debulking or non-debulking, upfront or interval surgery and platinum-based chemotherapy plus at least 3 cycles of bevacizumab were randomized to maintenance treatment with either olaparib plus bevacizumab (n = 537) or placebo plus bevacizumab (n = 269) for 2 years. Tumor BRCA mutations were present in 30 % in each arm.
The trial met its primary objective, demonstrating a statistically significant PFS improvement with olaparib plus first-line standard-of-care bevacizumab maintenance therapy (22.1 vs. 16.6 months; HR, 0.59; p < 0.0001). Furthermore, the combination conferred a significant advantage regarding time to first subsequent treatment (24.8 vs. 18.5 months; HR, 0.59; p < 0.0001). For OS, the data were still immature.
The safety profile of the combination was generally consistent with previous trials of each drug. Dose interruptions due to AEs became necessary in 54 % and 24 %, respectively, and treatment was discontinued due to AEs in 20 % vs. 6 %. The tolerability of bevacizumab was not reduced by the addition of olaparib. Among AEs of special interest for olaparib, the rate of new primary malignancies was shown to be comparable for the two arms (1.3 % and 1.1 %, respectively). Pneumonitis occurred in 1.1 % and 0 %, respectively. Health-related quality of life (HRQoL) was not reduced by the addition of olaparib.
Findings according to BRCA and HRD status
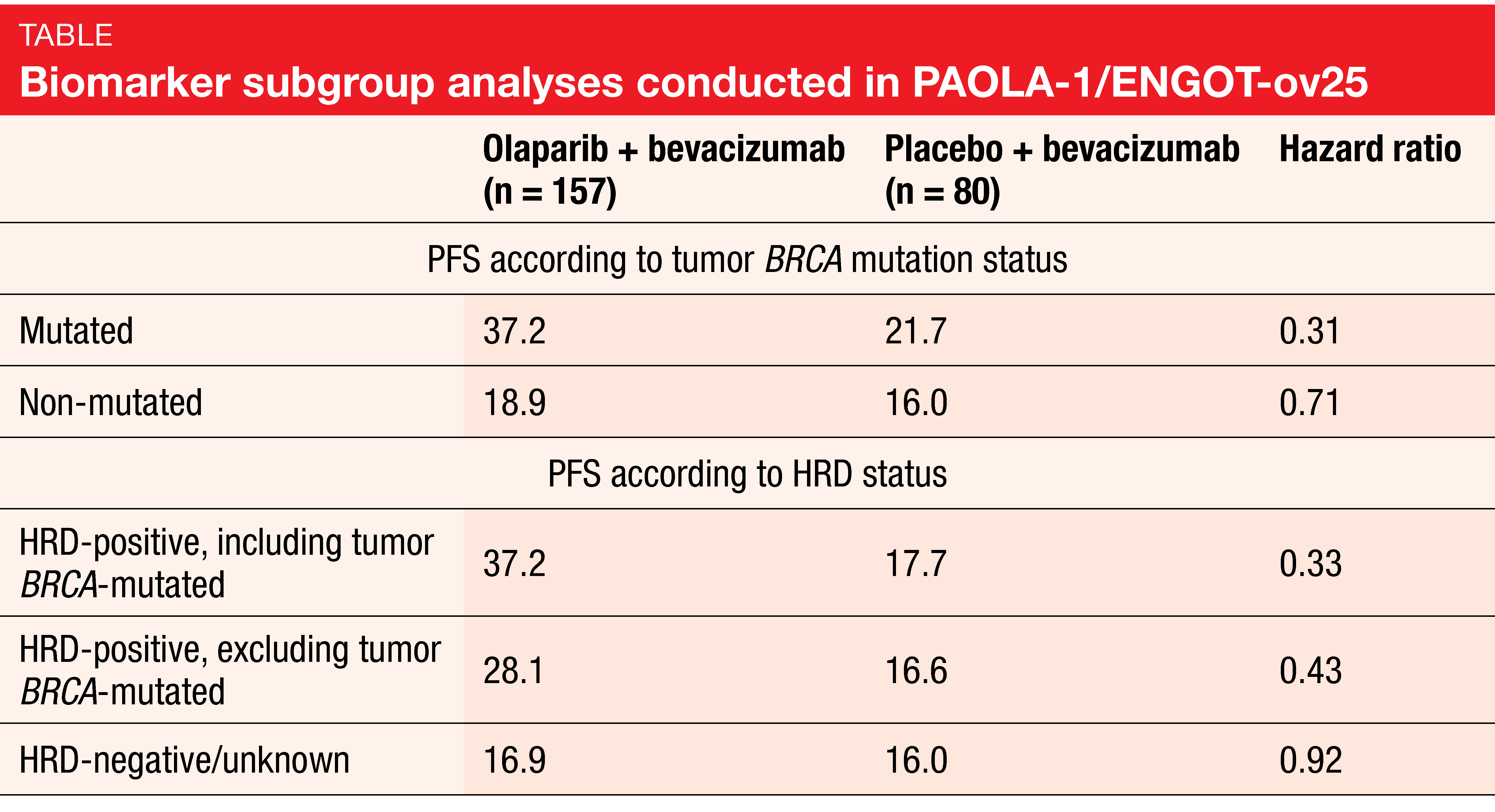
Prespecified subgroup analyses showed that patients with tumor BRCA mutations and those with a positive homologous recombination deficiency (HRD) status derived the greatest PFS benefits (Table). In the BRCA-positive group, PFS was 37.2 vs. 21.7 with olaparib plus bevacizumab compared to bevacizumab only (HR, 0.31), whereas the non-BRCA-mutant population benefited to a markedly lesser extent from the addition of olaparib (18.9 vs. 16.0 months; HR, 0.71). With respect to HRD status, several populations were assessed. Those with HRD positivity including tumor BRCA mutations demonstrated a 67 % risk reduction (median PFS, 37.2 vs. 17.7 months; HR, 0.33). For the HRD-positive population excluding tumor BRCA mutations, this was 57 % (28.1 vs. 16.6 months; HR, 0.43). The combined analysis of patients with HRD negativity and those with unknown HRD status showed no difference with regard to PFS (16.9 vs. 16.0 months; HR, 0.92), although when viewed separately, the HRD-unknown subgroup derived PFS benefit (HR, 0.71), whereas HRD-negative patients did not (HR, 1.00).
The authors pointed out that these results reveal a patient population beyond tumor BRCA-mutant patients who are HRD-positive and experience substantial benefit from maintenance treatment with olaparib and bevacizumab. The activity of the combination was apparent in a broad front-line population not restricted by surgical outcome or BRCA mutation status.
MEDIOLA: olaparib plus durvalumab
The rationale for combining PARP-targeted agents with immune checkpoint inhibitors is based on the observation that PARP inhibition may upregulate PD-L1 expression [3]. Therefore, the phase I/II, open-label, multicenter MEDIOLA trial assessed olaparib in combination with the PD-L1 inhibitor durvalumab in patients with advanced solid tumors including germline BRCA-mutant, platinum-sensitive relapsed OC after ≥ 1 platinum-based chemotherapy. Following an initial 4-week treatment phase with olaparib monotherapy, patients received olaparib plus durvalumab until disease progression. Preliminary data from this study demonstrated an objective response rate (ORR) of 72 % [4]. At ESMO 2019, Drew et al. presented updated findings from the germline BRCA-mutant, platinum-sensitive relapsed OC cohort of MEDIOLA after an additional follow-up of one year (n = 34) [5].
Disease control rate at 12 weeks, which constituted the primary endpoint, was 81.3 %. At 28 weeks, this still amounted to 65.6 %. Median PFS was 11.1 months, while median OS had not been reached yet. At the time of data cutoff, the ORR was 71.9 (n = 23), with CRs observed in 25.0 % (n = 8). While ORR was consistent with the previous report, CR had increased with longer follow-up. Median duration of response was 10.2 months. Patients after only one or two lines of previous therapy fared better than those who had received at least 3 lines. The less pretreated group showed longer PFS (15.4 and 12.0 months after 1 and 2 lines of treatment, respectively, vs. 8.3 months after ≥ 3 lines) and contained seven of eight complete responders.
PD-L1 expression on tumor cells was analyzed in 31 patients. This showed that positive (≥ 1 %) baseline PD-L1 expression correlated with longer PFS (13.6 vs. 10.3 months for PD-L1–negative patients). The combination continued to be well tolerated. Ever since the latest analysis one year previously, only one patient had discontinued treatment due to an AE. In all, the findings indicated that the combination of olaparib and durvalumab is most effective in early-line patients; over time, the addition of durvalumab might drive deeper responses. Further analysis in a larger patient population is warranted to assess the role of PD-L1 expression as a predictor for treatment benefit. According to the authors, it remains to be determined whether this regimen has the potential to replace chemotherapy in patients with germline BRCA-mutant, platinum-sensitive relapsed OC. An expansion cohort in the early-line setting is underway.
Secondary analyses of the SOLO1 trial
The randomized, double-blind, international phase III SOLO1 trial evaluated maintenance therapy with olaparib (n = 260) versus placebo (n = 131) after first-line chemotherapy in patients with newly diagnosed, advanced OC who had germline or somatic BRCA1 or BRCA2 mutations. Olaparib treatment led to a substantial PFS benefit compared to placebo (HR, 0.30; p < 0.001) [6] and thus was widely approved for the maintenance treatment of patients with BRCA-mutated advanced OC who achieved CR or PR to first-line platinum-based chemotherapy.
At ESMO 2019, Oaknin et al. reported time from randomization to second disease progression or death (PFS2) and time from randomization to second subsequent therapy from the SOLO1 trial [7]. According to this analysis, maintenance olaparib provided benefit beyond first progression, increasing both PFS2 (not reached vs. 41.9 months; HR, 0.50; p = 0.0002) and time to second subsequent therapy (not reached yet vs. 40.7 months; HR, 0.45) compared to placebo. These benefits were clinically meaningful and suggested that olaparib does not diminish the patient ability to receive subsequent therapy and respond to it.
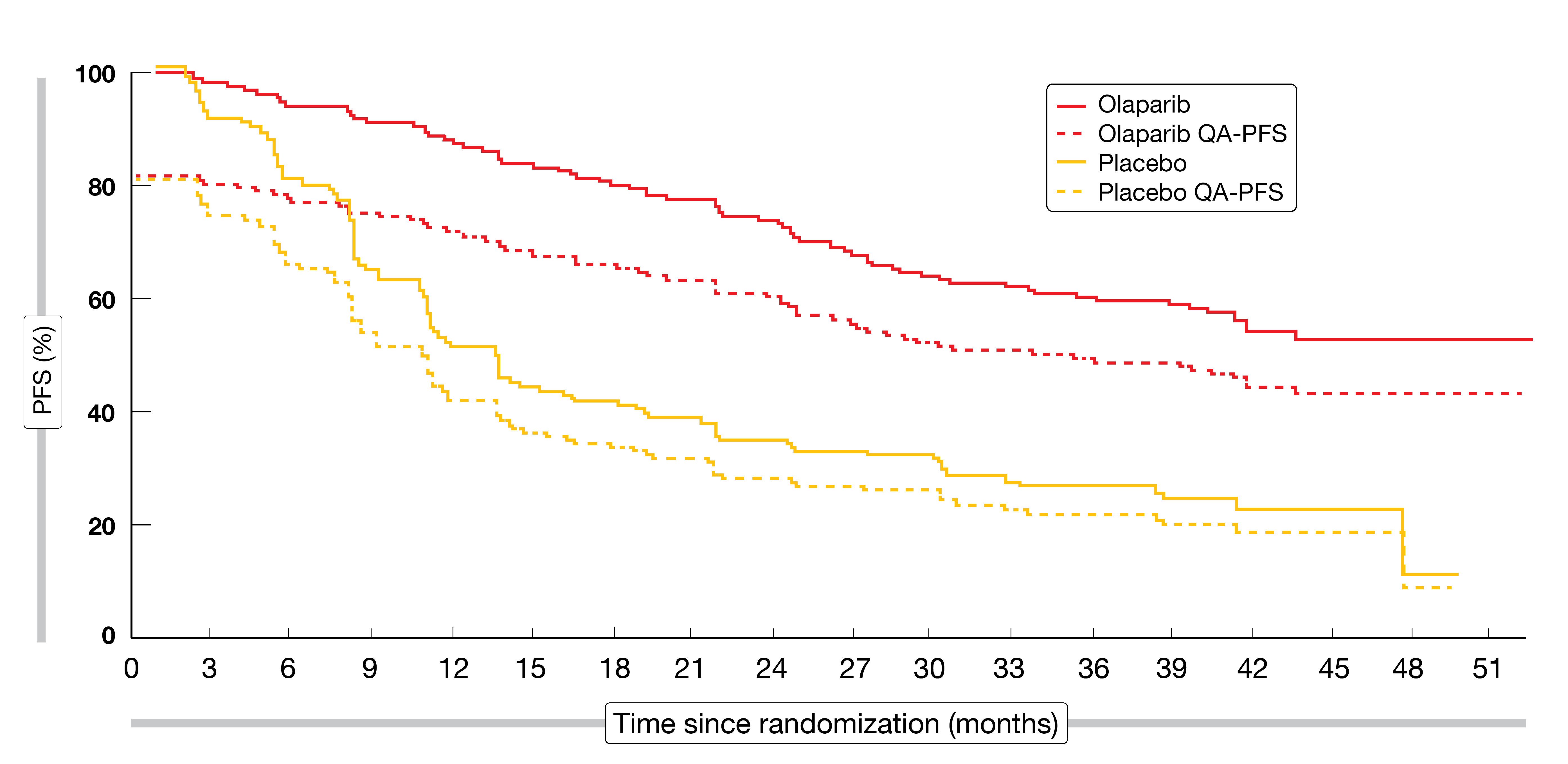
Considering the importance of high tolerability of maintenance therapy in a setting where most patients do not usually have OC-related symptoms, Friedlander et al. evaluated the patient-centered outcomes of quality-adjusted PFS and time without symptoms of disease or toxicity [8]. For both outcomes, olaparib maintenance, as compared to placebo, showed patient-centered benefits. Mean quality-adjusted PFS improved to a clinically meaningful extent, with a highly significant difference of 12.17 months between groups (Figure 2). Likewise, time without symptoms of disease or toxicity increased in a clinically meaningful manner with olaparib versus placebo; here, the between-group difference was 12.92 months and highly significant. These results provided further support indicating that prolongation of PFS did not take place at the expense of reduced HRQoL due to toxicity.
Using tumor samples from 341 patients included in the SOLO1 trial, Gourley et al. found that BRCA-gene–specific loss of heterozygosity (LOH) and genome-wide LOH scores are no feasible biomarkers in newly diagnosed, BRCA-mutated, advanced OC as they do not discriminate the extent of olaparib benefit [9]. Significant benefit was observed with olaparib in patients with both high and low genome-wide LOH scores. The utility of this marker in BRCA wildtype patients who receive first-line treatment for OC requires further investigation.
Figure 2: Significant improvement of mean quality-adjusted progression-free survival (QA-PFS) with olaparib maintenance vs. placebo
Niraparib in newly diagnosed OC
Niraparib was the first oral PARP inhibitor to be approved as maintenance therapy for all patients with recurrent OC that is both BRCA-mutant and BRCA-wildtype. Considering the high unmet need for many patients with newly diagnosed advanced OC after platinum-based chemotherapy, the PRIMA/ENGOT-OV26/GOG-3012 trial assessed niraparib in this setting [10]. Overall, 733 patients were randomized in a 2:1 fashion to either niraparib or placebo after response to first-line platinum-based chemotherapy. The population included patients at high risk of relapse. Stage IV disease was present in 35 % of cases, and almost 100 % of those with stage III OC had residual disease after primary debulking surgery. In 67 %, neoadjuvant chemotherapy had been administered. First-line chemotherapy had given rise to CR and PR in 69 % and 31 %, respectively. In 30 %, BRCA mutations were identified. Fifty-one and 34 % of patients had homologous recombination (HR)-deficient and HR-proficient tumors, respectively. PFS was defined as the primary endpoint.
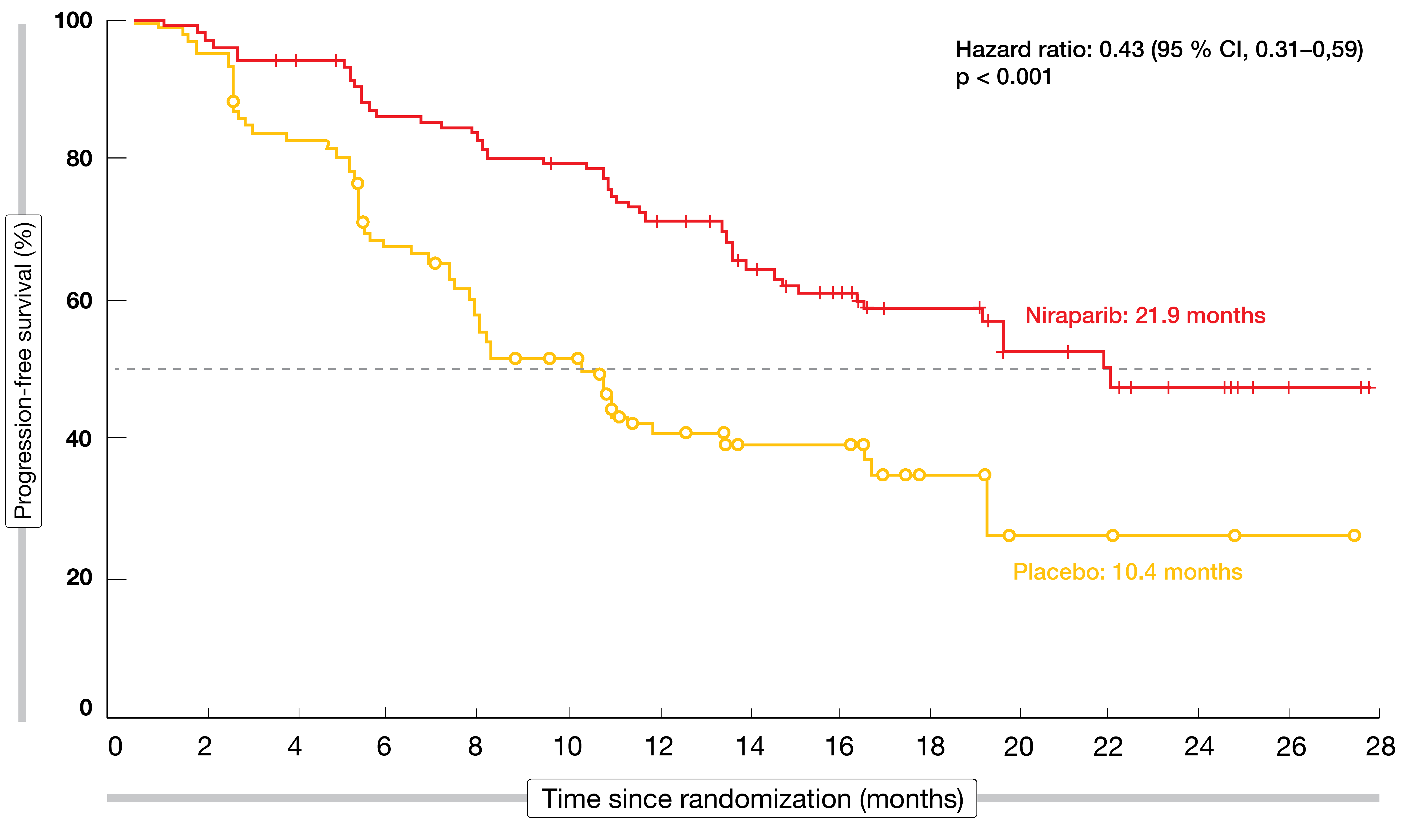
Niraparib provided a clinically significant PFS improvement after first-line chemotherapy in all patients. In the overall population, PFS was 13.8 vs. 8.2 months (HR, 0.62; p < 0.001). Patients with HR-deficient tumors benefited to an even greater extent from treatment, deriving a 57 % reduction in the risk of relapse or death (21.9 vs. 10.4 months; HR, 0.43; p < 0.001; Figure 3). Subgroup analyses according to BRCA mutation status showed that within the HR-deficient population, the PFS benefit obtained with niraparib was similar for both BRCA-mutant and -wildtype patients (HRs, 0.40 and 0.50, respectively). The HR-proficient subgroup experienced a 32 % risk reduction (HR, 0.68).
A pre-planned interim analysis of OS, which represented a key secondary endpoint, numerically favored niraparib over placebo. At 2 years, 91 % vs. 85 % of patients with HR-deficient tumors were alive; in the HR-proficient group, this was 81 % vs. 59 %. No new safety signals were observed, with reversible myelosuppression being the most common treatment-emergent AE. Evaluation of the FACT Ovarian Symptom Index Adjusted Health Utility Index Score showed that quality of life was maintained on both niraparib and placebo throughout the trial.
Based on these findings, the authors noted that niraparib is the first PARP inhibitor to demonstrate benefit in patients across biomarker subgroups after platinum-based chemotherapy in frontline. Significant activity was observed in patients at the highest risk of early disease progression.
Figure 3: Niraparib vs. placebo: progression-free survival in the HR-deficient population
Integration of veliparib in frontline and maintenance
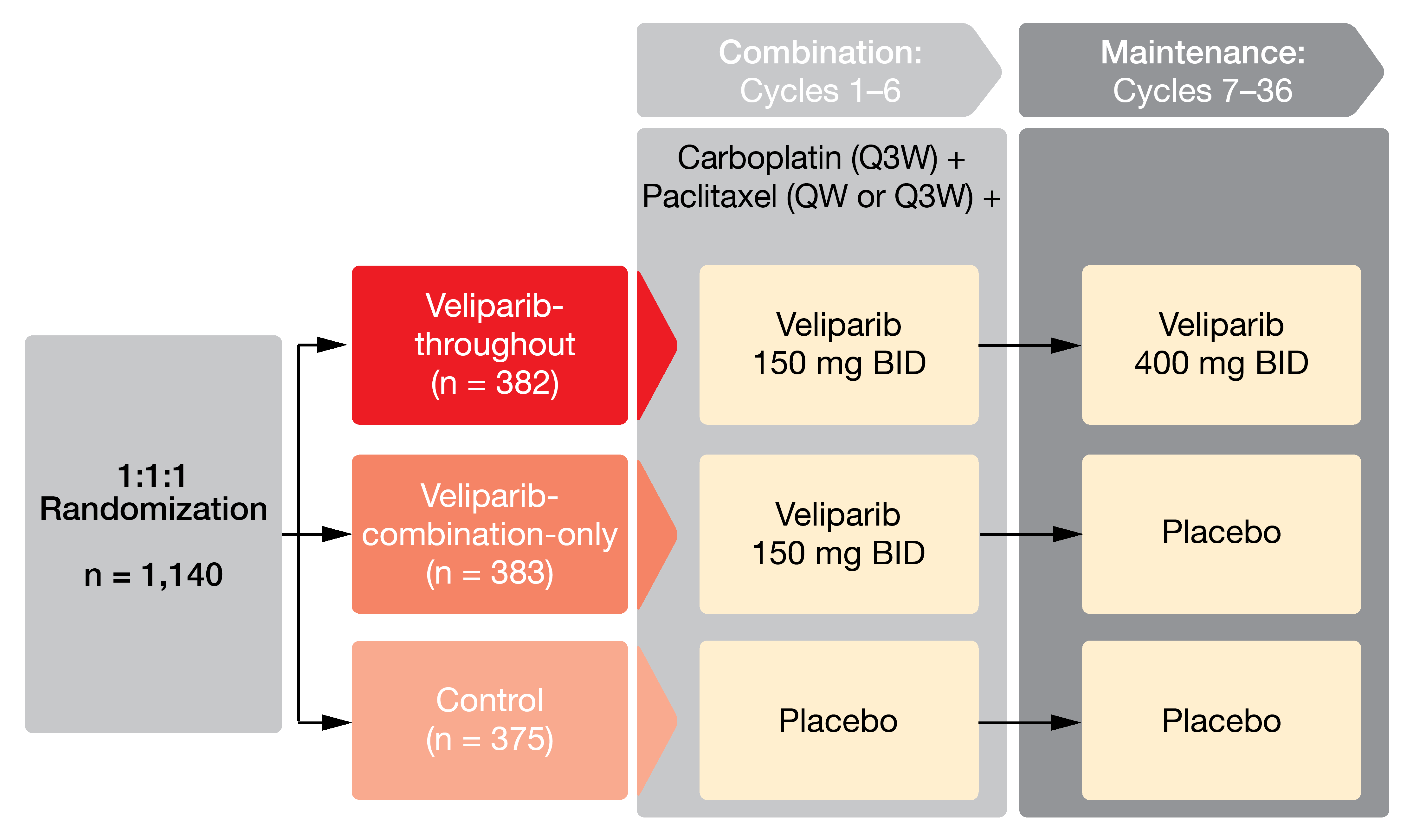
Combinations of PARP inhibitors with chemotherapy have been historically challenging due to hematologic toxicity. However, the specific binding characteristics of veliparib, primarily increased PARylation and decreased PARP trapping, were assumed to allow for use together with chemotherapy [11, 12]. The placebo-controlled, phase III VELIA/GOG-3005 trial assessed the integration of veliparib with front-line chemotherapy and maintenance in women with high-grade serous epithelial OC. This is the first randomized study designed to enroll all previously untreated patients with advanced-stage high-grade serous cancer regardless of BRCA status, surgical management, or response to treatment. Overall, 1,140 patients were randomized to one of three arms (Figure 4). In the veliparib throughout arm, veliparib 150 mg twice daily was added to carboplatin and paclitaxel for six cycles and followed by veliparib 400 mg twice daily as maintenance during cycles 7 to 36 (n = 382). The veliparib-combination-only arm received veliparib 150 mg twice daily plus chemotherapy, while placebo was administered during maintenance (n = 383). Patients in the control arm were treated with chemotherapy plus placebo followed by placebo (n = 375).
PFS for the veliparib-throughout regimen compared to controls was defined as the primary endpoint, with PFS including both the combination and maintenance phases. At ESMO 2019, Coleman et al. reported the results for this outcome [13]. BRCA mutations were present in the two arms in approximately 30 %, and HRD positivity was observed in 63 % each.
Figure 4: Design of the VELIA/GOG-3005 trial
Benefits regardless of biomarker status
The analysis showed that the addition of veliparib to chemotherapy and continuation as maintenance significantly extended PFS in all patient cohorts regardless of biomarker, choice of surgery, or paclitaxel regimen. Median PFS in the ITT population was 23.5 vs. 17.3 months for veliparib throughout vs. controls (HR, 0.68; p < 0.001). The BRCA-mutant population derived a 56 % risk reduction for disease progression and death (34.7 vs. 22.0 months; HR, 0.44; p < 0.001). In the HRD population, this was 43 % (31.9 vs. 20.5 months; HR, 0.57; p < 0.001). Smaller PFS benefits were observed for the non-HRD population (HR, 0.81), the BRCA wildtype/HRD group (HR, 0.74), and the BRCA wildtype patients (HR, 0.80). Moreover, the choice of surgery (primary vs. interval surgery) did not affect outcomes, which also applied to the paclitaxel regimen (weekly vs. every 3 weeks).
The importance of maintenance treatment is demonstrated by an analysis comparing the veliparib-combination-only arm with the control arm; here, no PFS benefit was gained (HR, 1.07). This finding was similar across the BRCA-mutant, HRD, and ITT populations. However, at the end of the combination phase, both veliparib-containing arms showed numerically higher ORRs than the control arm (84 % and 79 % vs. 74 %).
Veliparib could be safely administered together with carboplatin and paclitaxel. AEs observed with veliparib were consistent with chemotherapy during the combination phase, with cytopenia constituting the majority of grade 3 or 4 AEs. During maintenance phase, the AEs were in keeping with the known safety profile. HRQoL was assessed using the disease-related symptom – physical score category of the NCCN-FACT ovarian symptom index-18. Here, differences in mean change from baseline between arms and within all subgroups were small and not considered clinically significant. In their conclusion, the authors noted that veliparib plus chemotherapy should be considered a new treatment option for women with newly diagnosed, advanced-stage serous OC.
Veliparib plus chemotherapy in breast cancer: BROCADE3
The use of veliparib plus chemotherapy in patients with advanced HER2-negative breast cancer and germline BRCA1 or 2 mutation after ≤ 2 prior lines of cytotoxic therapy for metastatic disease was tested in the randomized, placebo-controlled phase III BROCADE3 study [14]. Veliparib plus carboplatin/paclitaxel (n = 337) was compared to placebo plus carboplatin/paclitaxel (n = 172). A maximum of 1 prior line of platinum was permitted; also, progression must have occurred at least 12 months after completion of chemotherapy. Most of the patients (81 % in each arm) had had no prior chemotherapy for metastatic disease. Prior (neo-)adjuvant chemotherapy had been administered in approximately 70 % in each arm. Investigator-assessed PFS constituted the primary endpoint.
Patients treated with veliparib plus chemotherapy experienced a statistically significant and clinically meaningful PFS benefit (14.5 vs. 12.6 months; HR, 0.705; p = 0.002). At 24 and 36 months, 34 % and 26 %, respectively, were alive and progression-free. These results were confirmed by independent central review, with median PFS of 19.3 vs. 13.5 months (HR, 0.695; p = 0.005) as well as 24- and 36-month rates of 44 % and 37 %. The durable benefit of the combination was also demonstrated by the longer duration of response (14.7 vs. 11.0 months), although ORRs were similar across treatment arms (75.8 % vs. 74.1 %), as were clinical benefit rates at 24 weeks (90.7 % vs. 93.2 %). Moreover, patients treated in the experimental arm experienced a significant benefit with respect to PFS2, i.e. the time from randomization until disease progression on subsequent therapy or death (21.3 vs. 17.4 months; HR, 0.760; p = 0.020). An interim OS analysis revealed non-significant superiority of the veliparib treatment (median, 33.5 vs. 28.2 months; HR, 0.945) after a substantial crossover. Among patients treated with chemotherapy only, 44 % elected to receive open-label veliparib as their first subsequent treatment.
Veliparib plus chemotherapy was well tolerated, with less than 10 % of patients discontinuing treatment due to AEs. The addition of veliparib did not substantially alter the toxicity profile of carboplatin/paclitaxel. For select AEs of special interest, it was shown that the rates of infections within 14 days of neutropenia did not differ across treatment arms; this was also true for hemorrhages within 14 days of thrombocytopenia, and myelodysplastic syndromes. In their conclusion, the authors noted that BROCADE3 is the first phase III trial to evaluate a PARP inhibitor with highly active platinum chemotherapy in patients with advanced breast cancer and a germline BRCA mutation.
REFERENCES
- Colombo N et al., Italian multicenter randomized phase II study of weekly paclitaxel vs. cediranib-olaparib with continuous schedule vs. cediranib-olaparib with intermittent schedule in patients with platinum resistant high-grade epithelial ovarian, fallopian tube, or primary peritoneal cancer. ESMO 2019, abstract LBA58
- Ray-Coquard I et al., Phase III PAOLA-1/ENGOT-ov25: maintenance olaparib with bevacizumab in patients with newly diagnosed, advanced OC treated with platinum-based chemotherapy and bevacizumab as standard of care. ESMO 2019, abstract LBA2_PR
- Jiao S et al., PARP inhibitor upregulates PD-L1 expression and enhances cancer-associated immunosuppression. Clin Cancer Res 2017; 23(14): 3711-3720
- Drew Y et al., 2018. Available at: https://www.sgo.org/wpcontent/uploads/2018/03/Late-Breaking-Abstracts-2018-SGOAnnual-Meeting-on-Womens-Cancer.pdf. Last accessed 2 August 2019
- Drew Y et al., Phase II study of olaparib + durvalumab (MEDIOLA): updated results in the germline BRCA-mutated (BRCAm) platinum-sensitive relapsed (PSR) ovarian cancer (OC) cohort. ESMO 2019, abstract 1190PD
- Moore K et al., Maintenance olaparib in patients with newly diagnosed advanced ovarian cancer. N Engl J Med 2018; 379(26): 2495-2505
- Oaknin A et al., Time to second progression and second subsequent therapy for patients with newly diagnosed, advanced ovarian cancer and a BRCA mutation treated with maintenance olaparib – phase III SOLO1 trial. ESMO 2019, abstract 998PD
- Friedlander M et al., Patient-centred outcomes with maintenance olaparib in newly diagnosed patients with advanced OC and BRCA mutations in the phase III SOLO1 trial to support the clinical benefit of prolongation of progression-free survival. ESMO 2019, abstract 996PD
- Gourley C et al., Analysis of tumour samples from SOLO1: frequency of BRCA-specific loss of heterozygosity (LOH) and progression-free survival (PFS) according to homologous recombination repair (HRR) deficiency LOH score. ESMO 2019, abstract 998PD
- González-Martin A et al., Niraparib therapy in patients with newly diagnosed advanced ovarian cancer (PRIMA/ENGOT-OV26/GOG-3012). ESMO 2019, abstract LBA1
- Donawho CK et al., ABT-888, an orally active poly(ADP-ribose) polymerase inhibitor that potentiates DNA-damaging agents in preclinical tumor models. Clin Cancer Res 2007; 13(9): 2728-2737
- Gray HJ et al., Phase I combination study of the PARP inhibitor veliparib plus carboplatin and gemcitabine in patients with advanced ovarian cancer and other solid malignancies. Gynecol Oncol 2018; 148(3): 507-514
- Coleman RL et al., VELIA/GOG-3005: Integration of veliparib with front-line chemotherapy and maintenance in women with high-grade serous carcinoma of ovarian, fallopian tube, or primary peritoneal origin. ESMO 2019, abstract LBA3
- Diéras V et al., Phase 3 study of veliparib with carboplatin and paclitaxel in HER2-negative advanced/metastatic gBRCA-associated breast cancer: BROCADE3. ESMO 2019, abstract LBA9