Disease progression on EGFR TKI therapy: what to do after erlotinib, gefitinib and afatinib?
First-line treatment for patients with EGFR-mutation-positive NSCLC includes the first-generation reversible EGFR TKI inhibitors erlotinib and gefitinib, and the second-generation ErbB family blocker, afatinib. These drugs can elicit dramatic responses, but acquired resistance generally limits the successful long-term treatment. In the majority of patients, tumour progression eventually develops, due to resistance.
T790M: culprit number 1
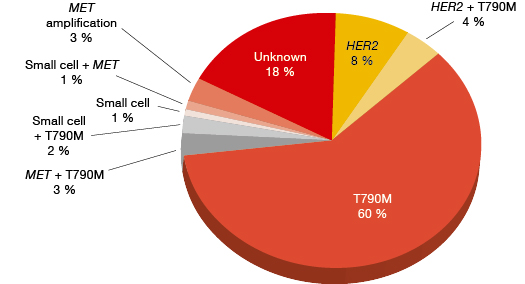
The most common mechanism of acquired resistance is the T790M mutation within exon 20 of the EGFR gene (Figure 1) [1]. Indolent slow tumour growth typically occurs when this mutation emerges [2]. “Repeated imaging may be necessary to identify progression,” explained Pasi A. Jänne, MD, PhD, Lowe Center for Thoracic Oncology, Dana Farber Cancer Institute, Boston, USA, during a symposium at the ESMO Asia Congress.
T790M works through an unusual resistance mechanism, in that it changes the affinity of the EGFR receptor for its natural substrate, ATP. While drugs like gefitinib and erlotinib are competitive inhibitors that outcompete the ATP binding at the binding site, the receptor shows greater affinity for ATP in the presence of this mutation. The original publication that described the T790M mutation in 2005 suggested the need for the development of covalent EGFR inhibitors as a potential solution with which to overcome the competitive advantage of T790M [3]. This led to the evaluation of covalent EGFRinhibitors. Two randomised phase III clinical trials assessed afatinib and dacomitinib in patients who had developed resistance to gefitinib and erlotinib [4, 5]. “However, the beneficial effects in terms of response rates, PFS or OS, as compared to placebo, were negligible” Dr. Janne said.
Other types of aberrations can also mediate resistance, such as MET amplification or small-cell transformation. Small-cell transformation has completely different effects from the T790M mutation, and it gives rise to rapid progression. These tumours can show the appearance of small-cell lung cancer (SCLC) and respond to SCLC treatments.
Figure 1: Relative frequencies of mechanisms of acquired resistance in EGFR-mutation-positive tumours
General principles of management
As Dr. Jänne explained, the first question that needs to be answered when progression occurs on EGFR TKI therapy is whether this progression is clinically significant in the individual patient. Tumour growth according to RECIST does not always necessitate a change of strategy, particularly if the patient is tolerating the treatment well. “Many patients receive post-progression TKI therapy.” This especially applies to patients experiencing longer PFS, a better response, and better performance status, according to the ASPIRATION trial [6].
One of the aspects that has been gaining attention is the use of local therapies, such as stereotactic radiation for brain metastases, isolated lung metastases, or bone metastases, while systemic anti-EGFR treatment is maintained. “This can be an alternative to discontinuation, if the patient shows only a single site of growth,” Dr. Jänne noted. The data provided by Weickhardt et al. suggested potential benefits by such a combined approach, especially in patients with CNS metastases as the first site of progression [7]. However, patients need to be selected carefully. The treatment decisions depend on the pattern of progression and the availability of local therapies. Finally, it should be established whether re-biopsy is feasible and whether a switch to chemotherapy should be considered.
Continuation of EGFR TKI treatment after the switch to chemotherapy used to be very common in clinical practice. Practice-changing results were generated by the IMPRESS trial, however [8]. Here, continuation of gefitinib together with chemotherapy did not provide any benefit compared to chemotherapy alone for PFS or response.
A TKI penetrating the brain: osimertinib
Progression is frequently observed in the same or similar locations where the initial tumour was identified. Sometimes it is confined to the CNS, with brain metastases or leptomeningeal disease emerging. “Many of the current EGFR inhibitors show poor penetration into the CNS, which poses problems in clinical practice,” Dr. Jänne said.
An exception to this is the third-generation EGFR TKI osimertinib (AZD9291) that has been developed to inhibit the T790M mutation rather than any of the other types of resistance mechanisms. It effectively penetrates the CNS. Osimertinib is approved for the treatment of T790M-mutation-positive NSCLC in the United States and Europe.
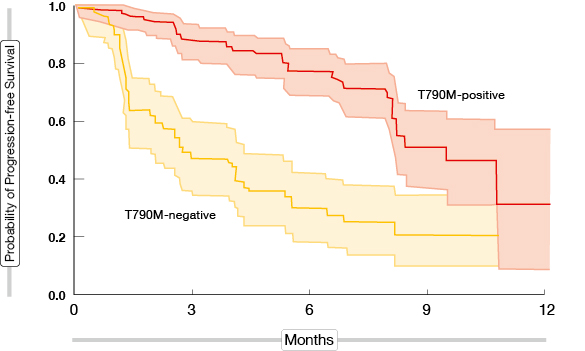
In the trial by Lee et al., a significant proportion of patients with leptomeningeal disease experienced responses to osimertinib treatment that were accompanied by significant clinical improvement [9]. Changes in the EGFR mutant copy number were obtained in the cerebrospinal fluid. A phase I study demonstrated greater efficacy of osimertinib in patients harbouring the T790M mutation than in those without it; the Kaplan-Meier plot showed instant separation of the PFS curves between these two populations (Figure 2) [10]. The AURA3 phase III trial is currently testing osimertinib compared to platinum-based doublet chemotherapy for advanced or metastatic T790M-mutation-positive NSCLC.
Figure 2: Greater PFS benefit of osimertinib in T790M-mutation-positive patients than in the T790M-mutation-negative cohort
BI 1482694
Another third-generation EGFR-mutant-specific drug is BI 1482694, which is active against mutant EGFR isoforms including T790M, while sparing wild-type EGFR. An open-label, multicentre phase I/II trial conducted in Korean patients assessed the safety and tolerability of BI 1482694, as well as its clinical activity at the recommended phase II dose [11]. Lee et al. presented findings at the maximum tolerated dose and at the recommended phase II dose of 800 mg once daily, in 76 patients with T790M-positive NSCLC who had previously been treated with an EGFR TKI [12].
In the 69 patients who were evaluable for response by independent assessment, the objective response rate was 62 %. Responses were observed early on. Eighty-four percent of confirmed objective responses were achieved by week 6. Median duration of response had not yet been reached at the time of the analysis. The ORR was similar whether or not the last treatment prior to study entry had been an EGFR TKI or chemotherapy. Disease control was obtained in 91 %. The most common treatment-related AEs included mild-to-moderate gastrointestinal symptoms (e. g., diarrhoea, nausea) and dermatological toxicity (e. g., rash, pruritus). There was no obvious effect on the QTc interval or glucose metabolism at the dose of 800 mg daily.
The ELUXA programme will assess the efficacy and safety of BI 1482694 in various settings of EGFR-mutated NSCLC. Findings in the T790M-positive patients are currently being generated in the phase II ELUXA 1 trial. Further third-generation EGFR TKIs, such as rocelitinib, ASP8273 and EGF816, are under clinical evaluation.
The future of EGFR inhibition
According to Dr. Jänne, physicians are finally in a position to begin to answer the question of the best sequence to use for EGFR inhibitors. Even allowing for the limited number of drugs available to date, there is the choice between starting a patient on a first-generation or second-generation EGFR inhibitor, followed by a third-generation inhibitor, or using third-generation inhibitors from the very start of the treatment. “The latter is rapidly becoming a more interesting option, even though more data are required to find out if this is a suitable approach,” Dr. Jänne explained. Initial data from the phase I clinical trial conducted with osimertinib indicates that first-line results might be superior to those that can be expected with first-generation EGFR inhibitors [13]. The study contained a cohort of EGFR-TKI-naïve individuals; these patients achieved an objective response rate of 75 %, and their median PFS had not yet been reached at the time of the analysis. “Although this was only a small cohort, the results are certainly encouraging,” Dr. Jänne emphasised. Also, the ongoing FLAURA trial, which is comparing osimertinib with gefitinib or erlotinib, will provide further insights on the use of this class of agents as an initial treatment.
Long-term success will ultimately require combination therapies, as Dr. Jänne pointed out. Combination studies were not feasible with older EGFR inhibitors due to overlapping toxicities, but they can be conducted with these new drugs. Various combination trials are ongoing. Common strategies include MEK and MET inhibition combinations as well as immunotherapy combinations. “The opportunity for all of us lies in figuring out which combination therapy to use and to decide whether this should be the initial treatment, an option after first-generation drugs, or the therapy of choice after the failure of successive EGFR TKI treatment,” Dr. Jänne summarised. “Preclinical trials will hopefully be able to provide answers to this.”
REFERENCES
- Yu HA et al., Analysis of tumor specimens at the time of acquired resistance to EGFR-TKI therapy in 155 patients with EGFR-mutant lung cancers. Clin Cancer Res 2013; 19: 2240-2247
- Sacher AG et al., Management of acquired resistance to epidermal growth factor receptor kinase inhibitors in patients with advanced non-small cell lung cancer. Cancer 2014; 120(15): 2289-2298
- Kobayashi S et al., EGFR mutation and resistance of non-small-cell lung cancer to gefitinib. N Engl J Med 2005; 352(8): 786-792
- Miller VA et al., Afatinib versus placebo for patients with advanced, metastatic non-small-cell lung cancer after failure of erlotinib, gefitinib, or both, and one or two lines of chemotherapy (LUX-Lung 1): a phase 2b/3 randomised trial. Lancet Oncol 2012; 13: 528-538
- Ellis PM et al., Dacomitinib compared with placebo in pretreated patients with advanced or metastatic non-small-cell lung cancer (NCIC CTG BR.26): a double-blind, randomised, phase 3 trial. Lancet Oncol 2014; 15(12): 1379-1388
- Park K et al., ASPIRATION: first-line erlotinib (E) until and beyond RECIST progression (PD) in Asian patients (pts) with EGFR mutation-positive (mut+) NSCLC. Ann Oncol 2014; 25(suppl_4): iv426-iv470
- Weickhardt AJ et al., Local ablative therapy of oligoprogressive disease prolongs disease control by tyrosine kinase inhibitors in oncogene-addicted non-small-cell lung cancer. J Thorac Oncol 2012; 7(12): 1807-1814
- Soria JC et al., Gefitinib plus chemotherapy versus placebo plus chemotherapy in EGFR-mutation-positive non-small-cell lung cancer after progression on first-line gefitinib (IMPRESS): a phase 3 randomised trial. Lancet Oncol 2015; 16(8): 990-998
- Lee DH et al., AACR-NCI-EORTC 2015, abstract PRO7
- Jänne PA et al., AZD9291 in EGFR inhibitor-resistant non-small-cell lung cancer. N Engl J Med 2015; 372(18): 1689-1699
- Park K et al., Updated safety and efficacy results from phase I/II study of HM61713 in patients (pts) with EGFR mutation positive non-small cell lung cancer (NSCLC) who failed previous EGFR-tyrosine kinase inhibitor (TKI). J Clin Oncol 2015; 33(suppl; abstr 8084)
- Lee J-S et al., Clinical activity and safety of the EGFR mutant-specific inhibitor, BI 1482694 (HM61713) in patients with T790M-positive NSCLC. ESMO Asia 2015, abstract 425PD
- Ramalingam SS et al., IASLC 2015