Emerging treatments in ALK-positive NSCLC: new options, but also new challenges
Treatment with the ALK tyrosine kinase inhibitor (TKI) crizotinib has been established as a standard first-line option in patients with ALK-rearranged advanced NSCLC. Before the advent of crizotinib, a platinum–pemetrexed doublet followed by pemetrexed maintenance was standard of care in non-squamous NSCLC. However, after an initial response to crizotinib, acquired resistance invariably develops due to multiple mechanisms, which can include secondary mutations in the ALK tyrosine kinase domain.
A range of newer-generation ALK inhibitors are now available or are currently under development. These include ceritinib, alectinib, brigatinib, ensartinib, entrectinib and lorlatinib. All of these have increased ALK-inhibition potencies and activities against the mutations that confer resistance to crizotinib. Also, many of these show improved CNS penetration over crizotinib.
First-line ceritinib: ASCEND-4
Ceritinib demonstrated robust anti-tumour activity in crizotinib-naïve and crizotinib-refractory patients with ALK-positive advanced NSCLC in the single-arm phase I and II ASCEND trials (ASCEND-1–3) and in a randomised phase III study (ASCEND-5). The randomised, global, open-label, ASCEND-4 phase III study that was presented in the Presidential Symposium at the WCLC compared first-line ceritinib 750 mg/day with platinum and pemetrexed chemotherapy, followed by pemetrexed maintenance, in untreated patients [1]. PFS by blinded independent radiological review was the primary endpoint. A total of 376 patients were enrolled in the study, with 189 randomised to ceritinib and 187 to chemotherapy. Approximately one third in each arm had brain metastases. Prior brain radiotherapy had been administered in 40 % of these patients.
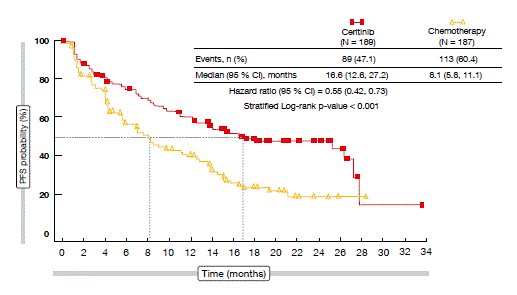
The study was positive with respect to its primary endpoint: PFS with ceritinib was significantly longer than with chemotherapy (16.6 vs. 8.1 months, respectively; HR, 0.55; p < 0.001; Figure). Most of the pre-defined subgroups derived greater PFS benefit from ceritinib than from chemotherapy. The differences in OS were not significant, but they trended in favour of ceritinib. At 24 months, OS rates were 70.6 % versus 58.2 %. The ceritinib ORR surpassed that obtained with chemotherapy by over 45 % (72.5 % vs. 26.7 %). Also, the patients responded more rapidly with ceritinib, as the median times to first response were 6.1 and 13.4 months, respectively. Median duration of response was 23.9 months versus 11.1 months, respectively. Patients with and without brain metastases benefited from ceritinib, with PFS improved in both groups (HRs, 0.70 and 0.48, respectively). The ALK inhibitor treatment gave rise to a superior intracranial response rate compared to chemotherapy (72.7 % vs. 27.3 %), and the ceritinib intracranial benefit was durable, with a median duration of response of 16.6 months.
Figure: Primary endpoint in ASCEND-4: PFS advantage with ceritinib over chemotherapy
According to patient-reported outcomes, the lung cancer symptom scores were significantly improved versus chemotherapy, and the time to definitive deterioration of lung-cancer-specific symptoms was prolonged. The safety profile of ceritinib was consistent with previous studies, with diarrhoea, nausea and liver enzyme elevations as the most common AEs. Management included dose adjustments or dose nterruptions/ delays, as well as concomitant medication.
J-ALEX: superiority of alectinib over crizotinib
In addition to ceritinib, alectinib is a standard option in the setting of progression on crizotinib treatment. The Japanese J-ALEX trial enrolled 207 patients who had received at most one prior line of chemotherapy. They were randomised to either first-line alectinib 300 mg BID (i.e., standard alectinib dose in Japan) or crizotinib 250 mg BID [2]. Patients with treated or asymptomatic brain metastases were also included.
The study was strongly positive. Alectinib proved superior to crizotinib with respect to the primary endpoint, which was independently reviewed PFS (not reached for alectinib vs. 10.2 months for crizotinib; HR, 0.34; p < 0.0001). Multiple stratified Cox regression analysis demonstrated consistent treatment effects that favoured alectinib over crizotinib, and this also extended to patients with brain metastases. As the CNS is a common site of disease progression in patients with ALK-positive NSCLC, the activity of new ALK-inhibiting compounds is of particular importance in this respect. There was an imbalance between the two treatment arms regarding the number of patients without and with CNS disease, as this was not a stratification factor in J-ALEX. More patients with untreated brain metastases enrolled on the alectinib arm.
According to separate PFS analyses for patients without and with CNS disease at baseline, both groups experienced highly significant benefits with alectinib compared to crizotinib. The risk reductions amounted to 63 % in those without brain lesions (median PFS, 20.3 vs. 10.0 months; HR, 0.37; p = 0.0001) and 91 % in those with pre-existing CNS metastases (not reached vs. 10.2 months; HR, 0.09; p = 0.0062). Alectinib-treated patients in the brain lesion group also fared significantly better with regard to time to progression of CNS disease (HR, 0.16; p = 0.0492). Similarly, for those without brain metastases at baseline, time to appearance of CNS disease was significantly longer with alectinib (HR, 0.17; p = 0.0019). Overall, alectinib showed greater activity for existing CNS disease, and greater potential to prevent the development of new CNS lesions.
Updated analysis on CNS results obtained with alectinib
These data are supported by a pooled analysis of two phase II trials. The pivotal NP28761 and NP28673 studies investigated alectinib 600 mg BID after progression on crizotinib treatment. NP28761 was conducted in North America and NP28673 globally. The results demonstrated high response rates and durable responses [3, 4]. A pooled analysis of these two trials performed with the data cut-off on 27 April, 2015, yielded a CNS ORR of 64.0 % and a duration of CNS response of 10.8 months in patients with measurable CNS disease at baseline [5].
At the WCLC, Ou et al. presented updated pooled data using 2016 data cut-offs to further evaluate the CNS efficacy of alectinib in these two trials [6]. According to this analysis, CNS ORR was 64.0 % in patients with measurable CNS disease and 44.1 % in those with measurable and non-measurable disease combined. Complete responses were achieved in 22.0 % and 28.7 %, respectively, with CNS disease control in 90.0 % and 86.0 %, respectively. These CNS responses were also durable, as they lasted for 11.1 and 13.8 months, respectively.
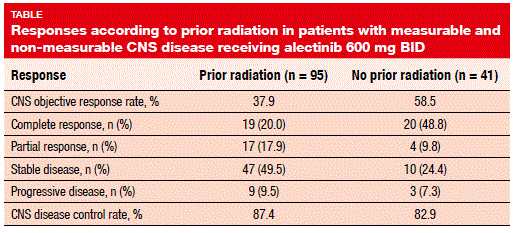
Moreover, the pooled data show that alectinib is active in the CNS regardless of prior radiation. Seventy percent of patients with measureable and non-measurable CNS disease had received prior radiotherapy; here, CNS ORR and CNS DCR were 37.9 % and 87.4 %, respectively. For those without prior radiotherapy, these were 58.5 % and 82.9 %, respectively, and complete responses occurred in 48.8 % (Table).
Brigatinib & lorlatinib
Likewise, the investigational next-generation ALK inhibitors brigatinib and lorlatinib have been shown to have pronounced activities, particularly in the CNS. An update from the pivotal randomised ALTA phase II trial that evaluated brigatinib at two doses (90 mg and 180 mg OD) in crizotinib-refractory patients demonstrated substantial efficacy and an acceptable safety profile in both arms [7]. At brigatinib 180 mg, ORR was 54 % according to the Independent Review Committee, and OS probability at 1 year was 82 %. Median PFS obtained with 180 mg surpassed PFS in the 90 mg arm considerably (15.6 vs. 9.2 months, respectively; HR, 0.58). When treated with brigatinib 180 mg, patients with measurable brain metastases experienced an intracranial ORR of 67 %.
A separate analysis of an ongoing phase I/II trial and the ALTA study assessed brigatinib activity in patients with intracranial CNS metastases, which yielded the high intracranial response rates of 53 % and 67 % (at brigatinib 180 mg) in patients with measurable metastases in the two trials [8]. Also, the median intracranial PFS findings were robust, at 14.6 and 18.4 months.
For lorlatinib, a phase I dose-finding study demonstrated significant clinical activity in patients with both ALK-positive and ROS1-positive NSCLC, most of whom had brain metastases and had received at least one prior ALK TKI [9]. In the ALK-positive group, ORR was 46 %, and median PFS was 9.6 months. Patients with brain metastases and target lesions achieved intracranial responses in 42 %. Durable responses were noted in patients who had received two or more prior ALK TKIs. Lorlatinib was generally well tolerated, with the most frequent treatment-related toxicity of hypercholesterolaemia, which was manageable with statin therapy. The phase II portion of this trial is ongoing at 57 centres worldwide.
Treatment selection – the current perspective
The growing armamentarium in the field of ALK-targeted agents raises several questions with respect to patient selection and selection of ALK TKIs. “The observation that many crizotinib-resistant tumours remain ALK-dependent over time provides the rationale for sequential therapy,” noted Benjamin Solomon, MBBD, PhD, Peter MacCallum Cancer Centre, Melbourne, Australia [10]. Retrospective analyses have suggested survival benefits with sequential ALK inhibitor therapies in ALK-positive NSCLC patients [11, 12].
In the light of the recent clinical trials, the optimal first-line treatment of advanced ALK-positive NSCLC remains to be established, although it appears likely that next-generation inhibitors will be used from the beginning, instead of crizotinib. “The ongoing phase III studies will provide us with much more data to definitively address this question soon,” Dr. Solomon pointed out. For now, following first-line crizotinib therapy, second-generation ALK inhibitors can be used, such as ceritinib, alectinib and brigatinib. When progression occurs with these agents, the choice of the third-line drug depends on factors such as CNS activity and the prevailing mutations at the time of progression. “One type of ALK inhibitor may be more effective in the CNS than another,” Dr. Solomon explained. Activities also vary with regard to mutations. For instance, the I1171T mutation confers resistance to alectinib, but not to ceritinib. The spectra of mutations against the newer ALK inhibitors differ from the mutation spectrum found in crizotinib-resistant specimens [13]. Among the currently available ALK inhibitors, lorlatinib has the widest range of mutation coverage, which includes the G1202R mutation that confers resistance to crizotinib, ceritinib and alectinib. Third-line agents such as lorlatinib can therefore be an option in cases of second-generation ALK TKI resistance mutations and/ or CNS disease.
As Dr. Solomon stated, assessment of ALK mutations using strategies such as liquid biopsy may eventually guide the choice of the ALK TKI therapy. Combination strategies may be required to overcome off-target mechanisms of resistance. “This potentially includes combinations with immunotherapy.”
REFERENCES
- De Castro G et al., First-line ceritinib versus chemotherapy in patients with ALK-rearranged (ALK+) NSCLC: a randomized, phase 3 study (ASCEND-4). WCLC 2016, PL03.07
- Kim YH et al., Alectinib (ALC) versus crizotinib (CRZ) in ALK-positive non-small cell lung cancer (ALK+ NSCLC): primary results from phase III
study (J-ALEX) – focus on CNS efficacy. WCLC 2016, MA07.03 - Shaw AT et al., Alectinib in ALK-positive, crizotinib-resistant, non-small-cell lung cancer: a single-group, multicentre, phase 2 trial. Lancet Oncol
2016; 17(2): 234-242 - Ou SH et al., Alectinib in crizotinib-refractory ALK-rearranged non-small-cell lung cancer: a phase II global study. J Clin Oncol 2016; 34(7): 661-668
- Gadgeel S et al., Pooled analysis of CNS response to alectinib in two studies of pretreated patients with ALK-positive non-small-cell lung cancer. J Clin Oncol 2016; 34(34): 4079-4085
- Ou SH et al., Updated pooled analysis of CNS endpoints in two phase II studies of alectinib in ALK+ NSCLC. WCLC 2016, MA07.01
- Camidge DR et al., Brigatinib in crizotinib-refractory ALK+ NSCLC: Central assessment and updates from ALTA, a pivotal randomized phase 2 trial. WCLC 2016, P3.02a-013
- Gettinger SN et al., Brigatinib activity in patients with ALK+ NSCLC and intracranial CNS metastases in two clinical trials. WCLC 2016, OA08.06
- Felip E et al., Safety and efficacy of lorlatinib (PF-06463922) in patients with advanced ALK+ or ROS1+ non-small-cell lung cancer (NSCLC). WCLC 2016, MA07.11
- Solomon B, Optimal application and sequence of ALK inhibition therapy. WCLC 2016, SC11
- Gainor JF et al., Progression-free and overall survival in ALK-positive NSCLC patients treated with sequential crizotinib and ceritinib. Clin Cancer
Res 2015; 21(12): 2745-2752 - Watanabe S et al., Progression-free and overall survival of patients with ALK rearrangementpositive non-small cell lung cancer treated sequentially with crizotinib and alectinib. Clin Lung Cancer 2016; 17(6): 528-534
- Gainor JF et al., Molecular mechanisms of resistance to first- and second-generation ALK inhibitors in ALK-rearranged lung cancer. Cancer Discov 2016; 6(10): 1118-1133