ALK-positive disease: pushing the borders of treatability
CNS and non-CNS efficacy of alectinib confirmed in ALEX
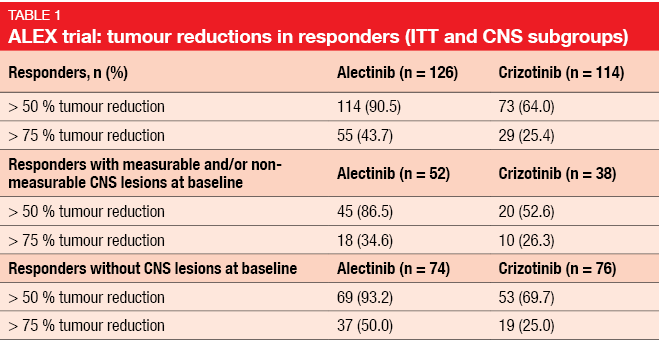
Standard treatment for patients with ALK-positive, advanced NSCLC includes the first-generation ALK inhibitor crizotinib and, more recently, second-generation ALK TKIs such as ceritinib and alectinib. The global, phase III ALEX trial tested the highly selective, CNS-active ALK inhibitor alectinib as first-line agent compared to crizotinib in patients with stage IIIB/IV ALK-positive NSCLC. Asymptomatic brain metastases were allowed in this study. According to the primary analysis, alectinib treatment gave rise to improved investigator-assessed PFS (not estimable vs. 11.1 months; HR, 0.47; p < 0.0001) [1]. PFS was also superior when estimated by the independent review committee (IRC; 25.7 vs. 10.4 months). Moreover, the patients in the experimental arm experienced prolonged DOR (not estimable vs. 11.1 months; HR, 0.36) and an improved safety profile. The updated efficacy and safety analysis of ALEX presented at the ASCO 2018 Congress confirmed the superiority of alectinib over crizotinib with respect to investigator-assessed PFS (34.8 vs. 10.9 months; HR, 0.43) [2]. PFS benefits occurred irrespective of the presence of baseline CNS metastases. Median PFS was 27.7 vs. 7.4 months (HR, 0.35) in patients with brain lesions and 34.8 vs. 14.7 months (HR, 0.47) in those without. In spite of similar ORRs across the two arms (82.9 % vs. 75.5 %), alectinib-treated patients with and without CNS metastases showed longer duration and greater depth of response. Overall, responses lasted 33.1 vs. 11.1 months with alectinib and crizotinib, respectively. In the ITT population, 43.7 % vs. 25.4 % of responders treated with alectinib and crizotinib, respectively, demonstrated tumour reductions > 75 % (Table 1). Both patients with and without CNS lesions experienced more pronounced tumour reductions in the experimental arm.
Alectinib showed consistently better tolerability compared to crizotinib despite longer treatment duration. OS data are still immature. These data consolidate alectinib as the standard of care for the first-line treatment of patients with ALK-positive NSCLC.
ALTA: update on brigatinib
However, the emergence of resistance and progression despite the use of second-generation agents necessitated the development of further agents. The next-generation ALK inhibitor brigatinib is being tested in the ongoing, randomised, phase II ALTA trial that includes 222 patients with advanced ALK-positive NSCLC who have experienced disease progression on crizotinib. Patients are receiving either brigatinib 90 mg QD (Arm A) or brigatinib 90 mg followed by 180 mg QD (Arm B). Updated data and exploratory analyses of the ALTA study presented at the ASCO 2018 Congress highlighted the continued efficacy of this treatment [3].
Confirmed ORRs as assessed by the investigator, which constituted the primary endpoint, were 46 % and 56 % in Arms A and B. Disease control occurred in 81 % and 86 %, respectively. In patients with ≥ 1 baseline CNS target lesion, ORRs were 43 % and 61 %. Systemic PFS according to IRC amounted to 9.2 and 16.7 months. At one year, 45 % and 61 % of patients were progression-free and alive. For OS, the analysis yielded median results of 29.5 and 34.1 months. Two-year OS probability was 55 % and 66 %.
The treatment induced similar depth of response in target lesions both in the CNS and outside of the CNS. Confirmed intracranial responses per IRC in patients with measurable baseline brain metastases occurred in 50 % and 67 %, and intracranial disease control rates were 85 % and 83 %. Intracranial responses lasted for 9.4 and 16.6 months. In patients with any brain metastases at baseline, intracranial PFS was 12.8 and 18.4 months.
The authors noted that the median PFS of 16.7 months in Arm B is highly comparable to the 16.3-month PFS seen in patients with crizotinib-treated, ALK-positive NSCLC who received the same regimen in the phase I/II trial of brigatinib [4]. Although cross-trial comparisons are limited by differing patient characteristics and assessment methods, this remains the longest median PFS in the post-crizotinib setting for any next-generation ALK inhibitor reported to date. The randomised phase III ALTA-1L trial that is evaluating brigatinib 180 mg (with lead-in) as compared to crizotinib in patients with ALK-inhibitor-naïve, advanced, ALK-positive NSCLC has already completed accrual.
Lorlatinib: deep and durable benefits in the phase II
An ongoing, multicentre, open-label, single-arm phase I/II study is testing the third-generation ALK and ROS1 TKI lorlatinib in patients with metastatic, ALK– or ROS1-positive NSCLC. Lorlatinib is CNS-active and shows broad-spectrum potency against most known ALK resistance mutations that develop during treatment with first- and second-generations TKIs. Asymptomatic untreated or treated CNS metastases at baseline are permitted in this trial. In the phase I part, lorlatinib showed clinically meaningful and durable responses (ORR, 46 %; DOR, 12.4 months) among patients with ALK-positive disease, many of whom had CNS lesions and disease progression after previous ALK TKI therapy [5].
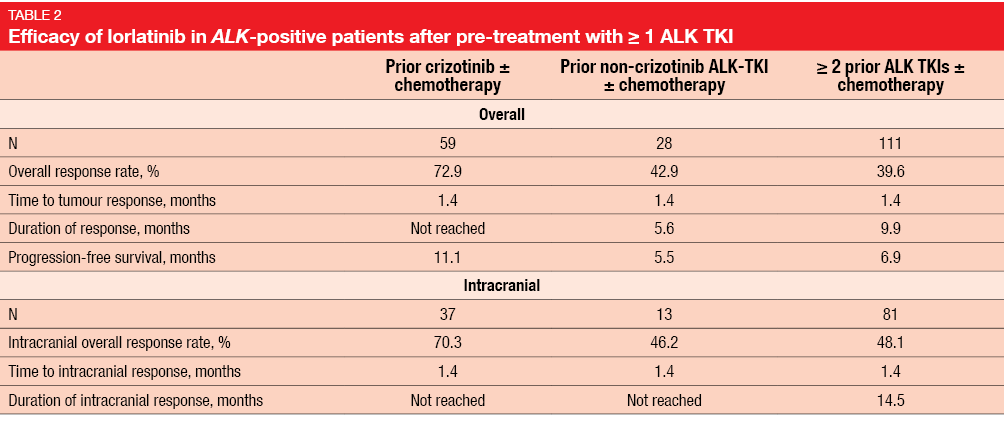
Besse et al. reported updated efficacy findings from the phase II portion that investigated lorlatinib among patients with ALK-positive NSCLC who had been pre-treated with ≥ 1 ALK TKI (n = 198) [6]. Of these 198 patients, 131 (66 %) showed CNS involvement. Safety data were presented for all treated phase II patients, i.e., those with both ALK-positive and ROS1-positive disease; these were both pre-treated and treatment-naïve (n = 275).
Lorlatinib gave rise to clinically meaningful benefit in pre-treated patients, including those who had received prior crizotinib, only 1 second-generation ALK TKI, or up to 3 prior ALK TKIs (Table 2). Rapid, deep and durable systemic and intracranial responses were observed. Translational analyses demonstrated that 45 patients had ≥ 1 detectable ALK kinase domain mutation. Lorlatinib showed anti-tumour activity across a variety of ALK kinase domain resistance mutations, including the G1202R/del mutation that was most frequently found. Responses also occurred in patients without detectable ALK kinase domain mutations. The treatment generally proved tolerable, with a low incidence of permanent discontinuations due to toxicity. AEs were manageable through dose modifications and/or supportive therapy.
REFERENCES
- Peters S et al., Alectinib versus crizotinib in untreated ALK-positive non-small-cell lung cancer. N Engl J Med 2017; 377: 829-838
- Camidge DR et al., Updated efficacy and safety data from the global phase III ALEX study of alectinib (AL) versus crizotinib (CZ) in untreated advanced ALK+ NSCLC. J Clin Oncol 36, 2018 (suppl; abstr 9043)
- Huber RM et al., Brigatinib in crizotinib-refractory ALK+ non-small cell lung cancer: efficacy updates and exploratory analysis of CNS ORR and overall ORR by baseline brain lesion status. J Clin Oncol 36, 2018 (suppl; abstr 9061)
- Bazhenova L et al., Brigatinib in anaplastic lymphoma kinase (ALK)-positive non-small cell lung cancer: long-term efficacy and safety results from a phase 1/2 trial. Ann Oncol 2017; 28 (suppl_5): v460-v496
- Shaw AT et al., Lorlatinib in non-small-cell lung cancer with ALK or ROS1 rearrangement: an international, multicentre, open-label, single-arm first-in-man phase 1 trial. Lancet Oncol 2017; 18: 1590-1599
- Besse B et al., Lorlatinib in patients with previously treated ALK+ advanced non-small cell lung cancer (NSCLC): updates efficacy and safety. J Clin Oncol 36, 2018 (suppl; abstr 9032)
More posts
Comprehensive sequencing of plasma cell-free DNA permits non-invasive cancer detection
Comprehensive sequencing of plasma cell-free DNA permits non-invasive cancer detect
Recent benchmarks in the management of small-cell tumours
Recent benchmarks in the management of small-cell tumours KEYNOTE-158 Extensive
ALK-positive disease: pushing the borders of treatability
ALK-positive disease: pushing the borders of treatability CNS and non-CNS effic
Interview with Barbara Melosky: “The sequencing question remains”
Interview with Barbara Melosky: “The sequencing question remains” Interview: Ba
New data on EGFR-directed TKIs across 3 generations
New data on EGFR-directed TKIs across 3 generations Erlotinib plus bevacizumab
Immune checkpoint blockade: determinants of treatment success
Immune checkpoint blockade: determinants of treatment success Various clinical