Enhancing the profile of KRAS-mutant lung cancer
Characteristics and outcomes
KRAS mutations constitute the largest subset of oncogene-driven lung adenocarcinomas, at approximately 30 %. Patients with KRAS-mutant metastatic lung cancer have heterogeneous clinical outcomes depending on the mutation subtype and associated co-mutations. El Osta et al. analysed the Lung Cancer Mutation Consortium (LCMC) database to evaluate the characteristics of these patients and the effect of KRAS mutation features on their outcomes [1]. In all, data from 1,655 patients who consented to participate in LCMC between 2009 and 2015 were analysed for baseline characteristics, mutations status/ subtypes/ co-mutations, OS (calculated from date of distant metastasis to death), and association of patient KRAS data with OS. Median follow-up was 2.15 years.
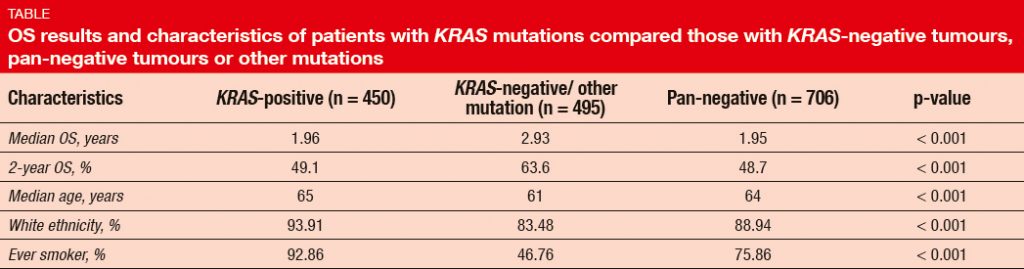
In this population, the incidence of KRAS mutations was 27 %. The presence of KRAS mutation predicted short OS (Table). Compared to patients with other mutations, average patient age was slightly higher in the KRAS-positive cohort, and there was a greater proportion of ever smokers. OS did not differ across KRAS mutations subtypes (i.e., KRAS G12C, G12D, and G12V), with 2-year OS rates of 46.5 %, 47.4 %, and 51.4 %, respectively. However, never smokers were more likely to have KRAS mutant subtype G12D. TP53 mutation occurred as the most common co-mutation (52 %), followed by STK-11 alterations (18 %), MET amplification (4 %), and PIK3CA mutation (3 %). With respect to outcome, STK- 11 co-mutation was shown to be associated with shorter OS.
ntrinsic primary resistance to immunotherapy
Skoulidis et al. retrospectively assessed clinical responses to PD-1/ PD-L1 therapy in co-mutation-defined subsets of KRAS-mutant NSCLC patients [2]. The rationale for this was the fact that the identification of molecular predictors of response to immunotherapy is deemed critical in order to maximise the therapeutic potential of immune checkpoint inhibitors. Previously, patients with KRAS-mutant lung cancer and co-occurring genetic events in STK11/LKB1 or TP53 had been defined as subgroups that showed marked differences in immune contexture. The present cohort included 162 patients harbouring metastatic KRAS-mutant NSCLC who received at least one cycle of PD-1/ PD-L1 therapy (i.e., nivolumab, pembrolizumab, durvalumab, anti-PD-1 plus anti-CTLA-4 therapy) and had available molecular profiling.
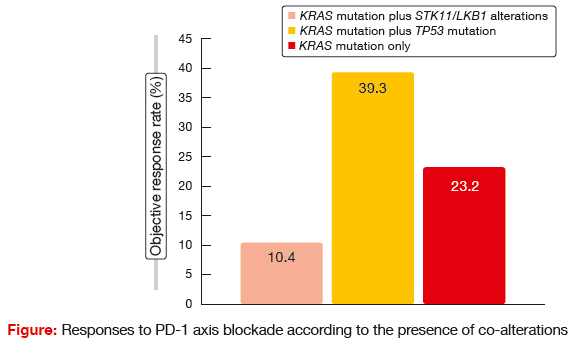
STK11 genetic alterations were demonstrated to be associated with poor response to PD-1 axis blockade. Patients with this co-mutation experienced significantly shorter PFS and OS following PD- 1/ PD-L1 therapy than those with TP53 mutation and KRAS mutation only. There were also significant differences with regard to response rates (Figure). The authors concluded that genetic alterations in the STK11/LKB1 tumour suppressor gene represent a novel, prevalent, tumour- cell-intrinsic mediator of primary resistance to PD-1/ PD-L1 blockade in KRAS-mutant NSCLC. Therefore, in addition to PD-L1 expression and tumour mutational burden, personalised immunotherapy approaches should take the co-mutation status of individual tumours into consideration.
References:
- El Osta B et al., Characteristics and outcomes of patients with metastatic KRAS mutant lung adenocarcinomas: Lung Cancer Mutation Consortium (LCMC) database. ASCO 2017, abstract 9021
- Skoulidis F et al., STK11/LKB1 co-mutations to predict for de novo resistance to PD-1/PD-L1 axis blockade in KRAS-mutant lung adenocarcinoma. ASCO 2017, abstract 9016
More posts
Anti-angiogenic and immunotherapeutic approaches in mesothelioma
Malignant pleural mesothelioma (MPM) is a rare tumour that is often diagnosed at an advanced stage. Limited efficacy of the available therapies contributes to the generally poor prognosis for MPM patients. Since 2003, the only approved regimen for MPM treatment has been chemotherapy with pemetrexed and cisplatin, with median survival of approximately 12 months.
Interview: Lung cancer in China: hurdles and progress
Lung cancer is a considerable issue in China. Every year, we have 700,000 new cases. There is a need to perform clinical trials and to launch innovative drugs. With regard to the introduction of targeted therapies, China lags 3 to 4 years behind when compared to the western countries. Two months ago, the EGFR TKI afatinib was launched, offering Chinese patients with EGFR-mutant lung cancer an effective treatment option.
Real-world utility of ctDNA NGS to identify matched targeted therapy
Liquid biopsy for plasma circulating tumour DNA (ctDNA) next generation sequencing (NGS) is a rapidly evolving science. Plasma ctDNA assays are now commercially available, and are increasingly adopted in the community with a paucity of evidence-based guidance on timing and value of this test. Sabari et al. sought to determine the feasibility and utility of plasma ctDNA NGS to identify matched targeted therapy in a real-world clinical setting.
Further defining the optimal use of immune checkpoint inhibitors
As the anti-PD-1 antibody nivolumab is known to induce deep and durable responses in a subset of lung cancer patients, this agent was investigated in the neoadjuvant setting, which is an area of unmet need. There have been no advances in systemic treatment of resectable lung cancer since 2004. Chaft et al. hypothesised that neoadjuvant nivolumab treatment might induce immunity against micrometastases.
Established targeted agents taking root in the HER2-positive setting
HER2 aberrations in lung cancer are being increasingly identified due to the use of sensitive testing procedures, such as multiplexed testing and next-generation sequencing. Mutations of the HER2 gene need to be distinguished from HER2 amplifications and HER2 protein overexpression. In contrast to breast and gastric cancer, HER2 overexpression in NSCLC does not always occur with HER2 amplification, while amplifications and HER2 mutations are generally mutually exclusive.
Diagnostics of EGFR-mutant disease: biomarkers with significant clinical implications
The clinical relevance of additional genetic alterations in advanced EGFR-mutant NSCLC is not clear. Blakely et al. hypothesised that co-occurring genomic alterations in cancer-related genes can cooperate with the mutant EGFR to drive de-novo resistance to EGFR TKI treatments. The investigators performed targeted exome sequencing of plasma cell-free DNA (cfDNA) in 86 samples collected from 81 patients with known clinical history.