Next-generation ALK inhibitors excel after crizotinib failure
ALK fusion-gene–positive lung cancer occurs in approximately 5 % of patients with advanced NSCLC [1]. The ALK inhibitor crizotinib demonstrates significant initial efficacy in patients with ALK-positive advanced NSCLC.
However, most patients eventually develop resistance, with the central nervous system (CNS) being one of the most common sites of first progression. Approximately half of these patients develop CNS metastases during crizotinib treatment. Next-generation ALK inhibitors represent efficacious options for patients who have progressed on crizotinib.
ASCEND-5
Ceritinib is a next-generation ALK inhibitor with 20-fold greater potency than crizotinib [2]. Anti-tumour effects of ceritinib in pre-treated patients were demonstrated in the ASCEND-1 and ASCEND-2 trials [3–5]. In ASCEND-2, ceritinib treatment promoted durable responses in an ALK-positive NSCLC population that had progressed on chemotherapy and crizotinib, including patients with brain metastases [5].
At the ESMO Congress, Scagliotti et al. presented the confirmatory phase III ASCEND-5 study, which compared ceritinib with second-line chemotherapy in the crizotinib-pretreated setting [6]. In this global, randomised, open-label trial, a total of 231 patients with locally advanced or metastatic ALK-positive NSCLC were randomised at 99 sites in 20 countries. Prior to study entry, they had received one or two chemotherapy regimens for advanced disease, as well as crizotinib (at any time). The treatment consisted of either ceritinib 750 mg/day or chemotherapy with pemetrexed or docetaxel. PFS was defined as the primary study endpoint. In each arm, more than half of the patients had metastatic brain disease, and radiotherapy had been administered to the CNS in one third of these cases.
Ample benefits with ceritinib treatment
This ALK inhibitor therapy proved highly efficient, promoting statistically significant and clinically meaningful improvement in PFS according to the blinded independent review committee (BIRC; 5.4 vs. 1.6 months; HR, 0.49; p < 0.001). This effect was robust and consistent across a number of subgroups. Clinical benefit was further supported by ORR (39.1 % vs. 6.9 %) and DCR (76.5 % vs. 36.2 %). OS data were immature at the data cut-off. The safety profile matched the observations in prior ceritinib studies, which featured primarily diarrhoea, nausea, vomiting, and transaminase elevation.
The analysis of patient-reported outcomes showed that compared with chemotherapy, ceritinib significantly improved lung-cancer specific symptoms and overall health status. While the majority of symptoms assessed with the QLQ-C30 questionnaire improved with ceritinib treatment, some deterioration was observed with the ALK inhibitor according to two scales for gastrointestinal symptoms (i.e., diarrhoea, nausea and vomiting). The authors concluded that these results establish ceritinib as a preferred treatment option in patients with crizotinib-resistant ALK-positive NSCLC.
Long-term follow-up of ASCEND-3: remarkable findings
Felip et al. presented the long-term follow-up of the global, phase II, single-arm, open-label ASCEND-3 study, which assessed ceritinib in 124 patients with metastatic ALK-positive NSCLC who had received no prior ALK inhibitor treatment [7]. They were either chemotherapy-naïve (although only two patients) or had been treated with up to three lines of chemotherapy and had experienced progression during or after the last chemotherapy regimen. Asymptomatic or neurologically stable brain metastases at baseline were allowed. Forty percent of patients had brain lesions at study entry; local radiotherapy had been applied in 53.1 %. The primary endpoint was ORR according to the investigator.
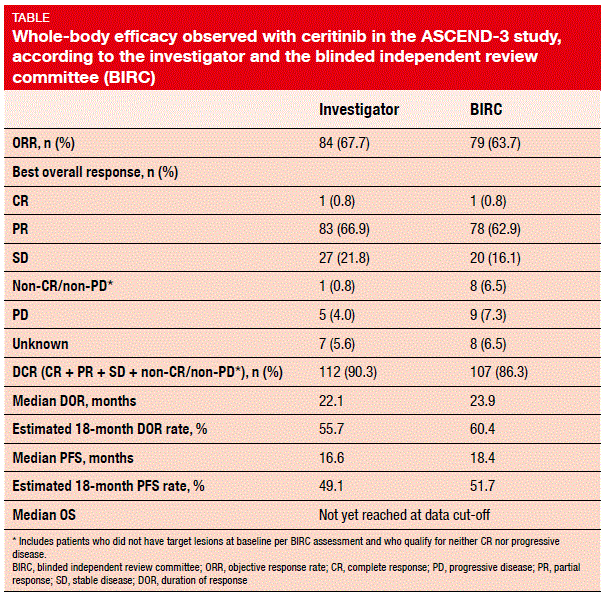
After a median follow-up of 25.9 months, 48.4 % of patients remained on treatment. With regard to whole-body efficacy, the analysis yielded robust ORR findings of 67.7 % according to the investigator, and 63.7 % according to the BIRC (Table). Decreases in tumour burden from baseline occurred in 94.7 % of patients. Disease control was obtained in 90.3 % and 86.3 % according to the investigator and the BIRC, respectively. The study revealed remarkable results with regard to median PFS (16.6 and 18.4 months according to the investigator and the BIRC, respectively) and OS: at 24 months, 67.5 % of the patients were alive, and median OS had not been reached yet. Ceritinib also showed activity in patients with brain metastases. Those with CNS lesions at baseline achieved a whole-body ORR of 57.1 % and a median PFS of 10.8 months. Overall intracranial responses were obtained in 61.5 %.
Updated patient-reported outcomes at a follow-up of up to 29 cycles were consistent with those previously reported. The patients showed improvements in symptoms burden from baseline with a mean change in the overall Lung Cancer Symptom Scale score that ranged from -3.39 to -14.83. Quality of life was maintained on treatment.
Alectinib: update on pivotal data
The highly selective and potent oral ALK inhibitor alectinib has been approved by the FDA for the treatment of patients who have progressed on, or are intolerant to, crizotinib. Two pivotal phase II studies, the global NP28673 trial and the North American NP28761 trial, formed the basis of this approval [8–11]. They enrolled a total of 225 previously treated patients with locally advanced or metastatic ALK-positive NSCLC, who had progressed after prior crizotinib treatment. All patients received alectinib 600 mg orally twice daily.
The updated safety and efficacy analysis of the NP28673 study was presented at ESMO 2016, and these demonstrated robust efficacy and good tolerability of alectinib, both systemically and in the CNS [12]. ORR in response-evaluable patients was 50.8 % according to the independent review committee. Chemotherapy- naïve patients benefited to a greater extent than those who had received prior chemotherapy (ORR, 73.1 % and 44.8 %, respectively). DCR was 78.7 % in response-evaluable individuals. Median PFS was 8.9 months in the intention-to-treat population, and median OS was 26.0 months. The patients with measurable CNS disease at baseline had a CNS ORR of 58.8 %.
An exploratory analysis assessed the time to response in both NP28673 and NP28761 [13]. Determination of how rapidly patients can benefit from alectinib was rated as important for both symptomatic patients and patients at the point of developing symptoms, particularly within the CNS. In addition, rapidity of response is of relevance for those with active CNS disease, as an area of high unmet medical need. The findings showed that alectinib therapy can achieve a rapid response. Most patients in all populations showed a RECIST response by the first assessment (8 weeks in NP28673, and 6 weeks in NP28761). This also applied to time to CNS response in patients with measurable and/or non-measurable CNS disease at baseline, irrespective of prior radiotherapy. Further investigation into the early clinical benefit (<6 weeks) is warranted, to evaluate alectinib for initial treatment of CNS metastases, with the potential for sparing radiation therapy.
Substantial anti-tumour activity of brigatinib
An ongoing phase I/II, single-arm, multicenter dose-escalation, dose-expansion trial is evaluating the investigational next-generation ALK inhibitor brigatinib in patients with advanced malignancies, which includes 79 ALK-positive NSCLC patients. Ninety percent of them had received prior crizotinib therapy.
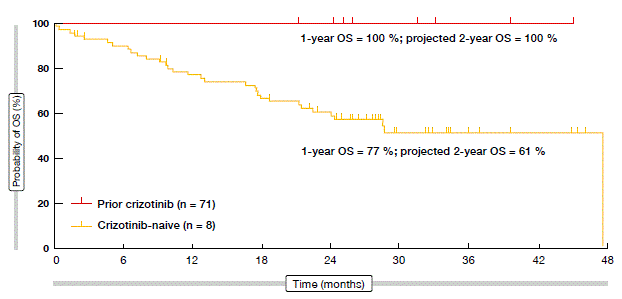
Updated data on the activity and safety of brigatinib in ALK-positive NSCLC patients after a median time on treatment of 20 months showed that tumour reductions with brigatinib were obtained in almost all of the cases [14]. Thirty-three percent of 72 evaluable patients had a 100 % decrease in target lesions. The confirmed ORR amounted to 62 % with all doses of brigatinib. One-hundred percent of crizotinib-naïve patients achieved confirmed objective responses, including three complete remissions. Disease control was achieved in 87 %. Median PFS had not yet been reached in the crizotinib-naïve population, and was 12.9 months in patients after prior crizotinib. For OS, the 1-year rates were 100 % and 77 % for these two groups, respectively (Figure). According to the analysis of patients with brain metastases (n = 50), brigatinib was highly active in the CNS. The intracranial ORR was 67 % in patients with measurable baseline disease. Most common treatment-emergent adverse events were nausea, fatigue, diarrhoea, headache and cough.
The results from this phase I/II study support further evaluation of brigatinib at 90 mg/day and 180 mg/day. The international, randomised, dose-evaluation ALTA trial showed that brigantinib has potential as a new treatment option in the crizotinib-resistant setting [15]. A randomised phase III trial of brigatinib versus crizotinib has been initiated in ALK inhibitor-naïve patients with advanced ALK-positive NSCLC (ALTA-1L; NCT02737501).
Figure: Overall survival obtained with brigatinib (ITT population)
REFERENCES
- Dearden S et al., Mutation incidence and coincidence in non small-cell lung cancer: metaanalyses by ethnicitiy and histology (mutMap).
Ann Oncol 2013; 24(9): 2371-2376 - Friboulet L et al., The ALK inhibitor certinib overcomes crizotinib resistance in non-small lung cancer. Cancer Discov 2014; 4: 662-673
- Shaw AT et al., Ceritinib in ALK-rearranged non-small-cell lung cancer. Engl J Med 2014; 370: 1189-1197
- Kim DW et al., Activity and safety of ceritinib in patients with ALK-rearranged non-small-cell lung cancer (ASCEND-1): updated results from the multicentre, open-label, phase 1 trial. Lancet Oncol 2016; 17: 452-463
- Crinò L et al., Multicenter Phase II Study of Whole-Body and Intracranial Activity With Ceritinib in Patients With ALK-Rearranged Non-
Small-Cell Lung Cancer Previously Treated With Chemotherapy and Crizotinib: Results From ASCEND-2. J Clin Oncol 2016; 34: 2866-2873 - Scagliotti G et al., Ceritinib versus chemotherapy in patients with advanced ALK+ NSCLC previously treated with chemotherapy and crizotinib: results from the confirmatory phase III ASCEND-5 study. ESMO 2016, abstract LBA_42
- Felip E et al., Phase II study of ceritinib in previously treated ALKi-naïve patients with ALK+NSCLC: whole-body efficacy in all patients and
in patients with baseline brain metastases. ESMO 2015, abstract 1208O - Ou SH et al., Alectinib in crizotinib-refractory ALK-rearranged non-small-cell lung cancer: A phase II global study. J Clin Oncol 2016; 34:
661-668 - Barlesi F et al., Updated efficacy and safety results from a global phase 2, open-label, single-arm study (NP28673) of alectinib in crizotinib-
refractory ALK+ non-small-cell lung cancer (NSCLC). Eur J Cancer 2015; 51(Suppl. 3): abstr 3101 - Shaw AT et al., Alectinib in ALK-positive, crizotinib-resistant, non-small-cell lung cancer: a single-group, multicentre, phase 2 trial. Lancet Oncol 2016; 17: 234-242
- Shaw AT et al., J Thorac Oncol 2015; 10 (Suppl. 2): abstr 1261
- Barlesi F et al., Updated efficacy and safety from the global phase II NP28673 study of alectinib in patients with previously treated ALK+
non-small-cell lung cancer (NSCLC). ESMO 2016, abstract 1263P - Gandhi L et al., Time to response in patients with ALK+ NSCLC receiving alectinib in the phase II NP28673 and NP28761 studies. ESMO
2016, abstract 1209PD - Bazhenova LA et al., Brigatinib in patients with anaplastic lymphoma kinase-positive nonsmall cell lung cancer in a phase 1/2 trial. ESMO 2016, abstract 1207PD
- Kim D-W et al., Brigatinib in patients with crizotinib-refractory ALK+ non-small cell lung cancer: first report of efficacy and safety from a pivotal
randomized phase 2 trial (ALTA). J Clin Oncol 34, 2016 (suppl; abstr 9007)
More posts
Next-generation ALK inhibitors excel after crizotinib failure
Next-generation ALK inhibitors excel after crizotinib failure ALK fusion-gene–p
Targeting angiogenesis can prolong life
Targeting angiogenesis can prolong life Anders Mellemgaard, MD, PhD, Clinical A
EGFR-targeted therapy: at the right time in the right patient
EGFR-targeted therapy: at the right time in the right patient Approximately 11
No phase III benefit with selumetinib in KRAS-mutant NSCLC
No phase III benefit with selumetinib in KRAS-mutant NSCLC Oncogenic mutations
SCLC: genomic alterations pave the way to targeted approaches
SCLC: genomic alterations pave the way to targeted approaches Rapid growth and
Rare driver mutations: encouraging results in small patient populations
Rare driver mutations: encouraging results in small patient populations As well